Although most cases of acute rhinosinusitis are caused by viruses, acute bacterial rhinosinusitis is a fairly common complication. Even though most patients with acute rhinosinusitis recover promptly without it, antibiotic therapy should be considered in patients with prolonged or more severe symptoms. To avoid the emergence and spread of antibiotic-resistant bacteria, narrow-spectrum antibiotics such as amoxicillin should be used for 10 to 14 days. In patients with mild disease who have beta-lactam allergy, trimethoprim/sulfamethoxazole or doxycycline are options. Second-line antibiotics should be considered if the patient has moderate disease, recent antibiotic use (past six weeks), or no response to treatment within 72 hours. Amoxicillin-clavulanate potassium and fluoroquinolones have the best coverage for Haemophilus influenzae and Streptococcus pneumoniae. In patients with beta-lactam hypersensitivity who have moderate disease, a fluoroquinolone should be prescribed. The evidence supporting the use of ancillary treatments is limited. Decongestants often are recommended, and there is some evidence to support their use, although topical decongestants should not be used for more than three days to avoid rebound congestion. Topical ipratropium and the sedating antihistamines have anticholinergic effects that may be beneficial, but there are no clinical studies supporting this possibility. Nasal irrigation with hypertonic and normal saline has been beneficial in chronic sinusitis and has no serious adverse effects. Nasal corticosteroids also may be beneficial in treating chronic sinusitis. Mist, zinc salt lozenges, echinacea extract, and vitamin C have no proven benefit in the treatment of acute bacterial rhinosinusitis.
In this article, the evidence supporting different treatments for acute bacterial rhinosinusitis (ABRS) is reviewed. In part one1 of this two-part article, clinical criteria for evaluating ABRS are described.
Strength of Recommendations
| Key clinical recommendation | Label | References |
|---|---|---|
| Amoxicillin for 10 to 14 days is a reasonable first-line agent. | A | 4,5,10 |
| In patients with mild disease who have beta-lactam hypersensitivity, trimethoprim-sulfamethoxazole (Bactrim, Septra) or doxycycline (Vibramycin) are reasonable, cost-effective, first-line options. | A | 16 |
| In patients with moderate disease, recent antibiotic use, or lack of treatment response within 72 hours, amoxicillin-clavulanate potassium (Augmentin) or a fluoroquinolone should be prescribed. | C | 16 |
| Ancillary treatments such as decongestants, topical anticholinergics, guaifenesin (Hytuss), saline nasal irrigation, and nasal corticosteroids may be beneficial. | C | 27,28,31,3–36,38,39,44,45 |
| Mist, zinc salt lozenges, echinacea, and vitamin C have no proven benefit. | A | 46,47 |
| Patients with complications or treatment failure after extended antibiotic therapy should be referred to an otolaryngologist. Patients with frequent recurrences of acute bacterial rhinosinusitis and inadequately controlled allergic rhinitis should be referred to an allergist for consideration of immunotherapy. | C | 6,48 |
Antibiotics
About two thirds of patients with ABRS improve without antibiotic treatment, and most patients with viral upper respiratory infection (URI) improve within seven days.2 Antibiotic therapy should be reserved for patients who have had symptoms for more than seven days and who present with two or more clinical criteria for ABRS (purulent nasal discharge, maxillary tooth or facial pain [especially unilateral], unilateral maxillary sinus tenderness, or worsening symptoms after initial improvement), or for those with severe symptoms3 (Figure 1).
Figure 1 Diagnosis and Management of Acute Bacterial Rhinosinusitis in Immunocompetent Adults
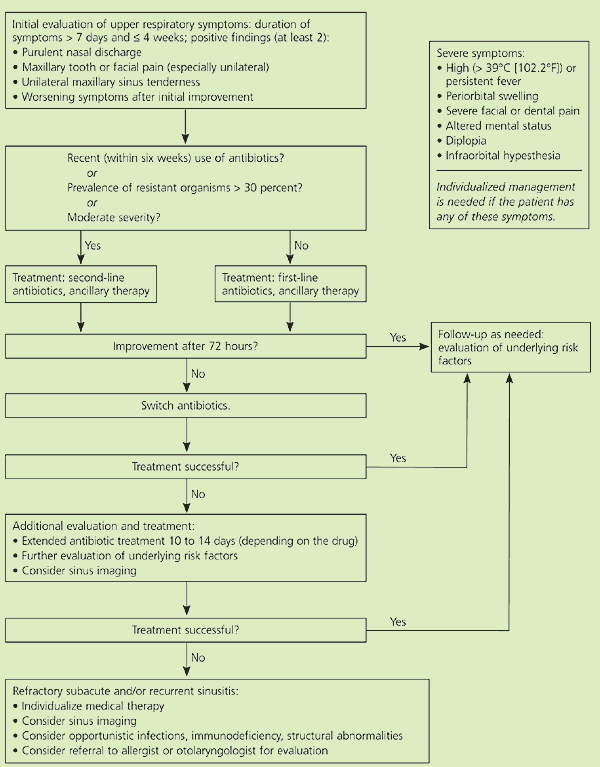
Algorithm for diagnosis and management of acute bacterial rhinosinusitis in immunocompetent adults.
RESULTS OF CLINICAL TRIALS
There have been no randomized controlled trials (RCTs) of antibiotic treatment for ABRS using sinus aspirate cultures before and after treatment, although nonrandomized trials have demonstrated bacteriologic cures. Five RCTs and two meta-analyses have compared antibiotics, usually amoxicillin and trimethoprim-sulfamethoxazole (TMP-SMX; Bactrim, Septra), with placebo, with clinical improvement as the outcome, which is the more clinically relevant patient-oriented outcome.4,5 About 47 percent of patients treated with antibiotics and 32 percent of the control group were cured at 10 to 14 days. Eighty-one percent of patients treated with antibiotics and 66 percent of the control group were cured or improved, meaning one patient benefited for every seven treated with antibiotics. The treatment effect in these trials may have been underestimated because the lack of specificity of diagnosis diluted the effect of treatment.
Amoxicillin-clavulanate potassium (Augmentin), cephalosporins (cefuroxime [Ceftin] and cefixime [Suprax]), and macrolides (azithromycin [Zithromax] and clarithromycin [Biaxin]), have been studied extensively.6,7 All have demonstrated similar clinical success rates—generally above 85 percent. The use of fluoroquinolones for ABRS is relatively new. Ciprofloxacin (Cipro) and cefuroxime had 90 percent resolution rates when administered to patients in a primary care setting.8 In an open-label RCT, levofloxacin (Levaquin) and clarithromycin had 96 and 93 percent clinical success rates, respectively.9
Four meta-analyses published within the past seven years concluded that newer broad-spectrum antibiotics are no more effective than narrow-spectrum antibiotics.4,5,10,11 In most of these studies, amoxicillin was compared with a cephalosporin, a fluoroquinolone, or a macrolide. The rapid emergence of antibiotic-resistant organisms associated with ABRS has made choosing an antibiotic more difficult. Surveillance studies have shown an increasing prevalence of antibiotic-resistant Streptococcus pneumoniae.12,13 Up to 25 percent of these bacteria are penicillin resistant, and 15 percent are penicillin intermediate. Resistance to macrolides, doxycycline (Vibramycin), and TMP-SMX is common.12 The prevalence of beta-lactamase–producing Haemophilus influenzae is about 30 percent, and resistance to TMP-SMX is common.12 Nearly all Mycobacterium catarrhalis isolates produce beta-lactamase.
SELECTING AN ANTIBIOTIC
To integrate current antibiotic resistance surveillance data into antibiotic recommendations, the Sinus and Allergy Health Partnership (SAHP) used the Poole Therapeutic Outcomes Model, a mathematical model that predicts clinical efficacy for each of the antibiotics commonly prescribed for ABRS (Table 1).13–16 The model incorporates assumptions about the probability of bacterial infection, pathogen distribution, spontaneous resolution rates, and in vitro activity of antibiotics.15
TABLE 1 Oral Antibiotics Used in the Treatment of Acute Bacterial Rhinosinusitis
| Antibiotic | Dosage/frequency | Calculated clinical efficacy (%)* | Cost† | |
|---|---|---|---|---|
| Mild disease and no recent antibiotic use | ||||
| Amoxicillin-clavulanate potassium (Augmentin)‡ | 500 mg every 8 hours, 875 mg every 12 hours§ | 91 | $83.96 to 112.08 | |
| High dose (Augmentin XR) | 2,000 mg every 12 hours§ | — | 112.08 | |
| Amoxicillin (Amoxil) | 500 mg every 8 hours, 875 mg every 12 hours | 88 | 7.35 to 8.77 (8.25) | |
| High dose | 1,000 mg every 8 hours | — | 14.70 to 17.54 (16.50) | |
| Cefpodoxime (Vantin)‡ | 200 mg every 12 hours | 87 | 118.48 | |
| Cefuroxime (Ceftin)‡ | 250 mg or 500 mg every 12 hours | 85 | 108.53, 197.75 | |
| Cefdinir (Omnicef)‡ | 300 mg every 24 hours | 83 | 44.66 | |
| If beta-lactam allergic: | ||||
| TMP-SMX DS (Bactrim DS, Septra) | 160 to 800 mg every 12 hours | 83 | 6.64 to 27.76 (38.85) | |
| Doxycycline (Vibramycin) | 100 mg every 12 hours | 81 | 5.00 to 27.36 (97.13) | |
| Azithromycin (Zithromax) | 500 mg on day 1, 250 mg on days 2 through 5 | 77 | 47.44 | |
| Clarithromycin (Biaxin) | 250 mg or 500 mg every 12 hours | 77 | 90.22, 90.22 | |
| Telithromycin (Ketek) | 800 mg every 24 hours | 77 | — | |
| Moderate disease or recent antibiotic use | ||||
| Gatifloxacin (Tequin) | 400 mg every 24 hours | 92 | 95.68 | |
| Levofloxacin (Levaquin) | 500 mg every 24 hours | 92 | 101.47 | |
| Moxifloxacin (Avelox) | 400 mg every 24 hours | 92 | 101.92 | |
| Amoxicillin-clavulanate (high dose) | 2,000 mg every 12 hours§ | 91 | 112.09 | |
| Ceftriaxone (Rocephin) | 1 g every 24 hours | 91 | 255.80∥ | |
| Combination therapy¶ | — | — | — | |
| If beta-lactam allergic: | ||||
| Gatifloxacin, levofloxacin, moxifloxacin | As above | As above | As above | |
| Clindamycin (Cleocin) plus rifampin (Rifadin)** | 150 to 450 mg every 6 hours, 300 mg every 12 hours | — | 76.49 to 183.19 (154.21 to 370.38) | |
TMP-SMX = trimethoprim-sulfamethoxazole.
*— Clinical efficacy based on calculation from the Poole Therapeutic Outcomes Model.15
†—Estimated cost to the pharmacist based on average wholesale prices in Red book. Montvale, N.J.: Medical Economics Data, 2004. Cost to the patient will be higher, depending on prescription filling fee. Cost is for 10 days of therapy, unless stated otherwise.
‡—Any benefit of these agents as initial therapy must be balanced against their much higher cost and concerns about increasing antibiotic resistance.
§—Based on amoxicillin component.
∥—Cost is for five days of therapy, including injection fee.
¶—Combination therapies include high-dose amoxicillin or clindamycin plus cefixime or high-dose amoxicillin or clindamycin plus rifampin.16
**—Provides coverage for Streptococcus pneumoniae but has no activity against Haemophilus influenzae.
When choosing antibiotic therapy for ABRS, physicians should consider recent antibiotic use, efficacy, and cost. The SAHP guidelines classify patients with ABRS into two groups to determine initial treatment: (1) those with mild symptoms who have not received antibiotics within six weeks and (2) those who have moderately severe disease or have received antibiotics within six weeks.16 Patients with moderate disease are considered less likely to have spontaneous resolution and thus have a higher rate of treatment failure. The guidelines offer no criteria for severity. The categorization of moderate or mild severity is left to the physician’s clinical judgment, but an example was offered with earlier recommendations that emphasized the inflammatory signs of fever and tenderness.17
Although lacking complete H. influenzae coverage, amoxicillin is still a good choice for a first-line antibiotic in community-acquired ABRS because many infections with resistant organisms improve anyway,18 and because it is well tolerated and inexpensive (Table 1).13–16 Higher daily doses of amoxicillin (3 to 4 g per day) may be necessary in areas with a high prevalence of penicillin-resistant S. pneumoniae. TMP-SMX and doxycycline are alternatives for use in patients who are allergic to beta lactams, but they have limited coverage for H. influenzae and S. pneumoniae, and failure rates of up to 25 percent are possible.16 Erythromycin, second-generation cephalosporins with less activity against H. influenzae (e.g., cefaclor [Ceclor], cefprozil [Cefzil], loracarbef [Lorabid]), and tetracycline should not be used to treat ABRS.19
Although the cephalosporins (cefpodoxime [Vantin], cefuroxime, cefdinir [Omnicef], ceftriaxone [Rocephin]) and amoxicillin/clavulanate potassium also have been recommended for initial treatment,16 any benefit of these agents as initial therapy must be balanced against their much higher cost and concerns about increasing antibiotic resistance in the community. A retrospective cohort study of a pharmaceutical database of 29,000 adults with ABRS showed equivalent success rates with the use of older, inexpensive antibiotics at one half the cost.20 A cost-effectiveness analysis showed that even if more expensive agents were 23 percent more effective than amoxicillin, using them empirically would be cost effective only if the prevalence of true bacterial sinusitis in treated patients was greater than 80 percent.21
Second-line antibiotics should be considered when the patient has moderate disease, has used antibiotics in the past six weeks, or has no response to treatment within 72 hours. Amoxicillin-clavulanate potassium and fluoroquinolones (gatifloxacin [Tequin], levofloxacin, and moxifloxacin [Avelox]) currently have the best coverage for H. influenzae and S. pneumoniae. Other choices include intramuscular ceftriaxone or combination therapies including high-dose amoxicillin; clindamycin (Cleocin) plus cefixime; or high-dose amoxicillin or clindamycin plus rifampin (Rifadin).16 In patients with a history of beta-lactam allergies, the use of fluoroquinolones or combination therapy with clindamycin and rifampin has been recommended.16
DURATION OF TREATMENT
Most clinical trials have used 10- to 14-day courses of antibiotic therapy. Sinus puncture studies have shown eradicated pathogens in at least 95 percent of patients after a 10-day course of antibiotics.22 Results of one study showed no differences in clinical or radiographic improvement between patients receiving three- or 10-day courses of TMP-SMX.23 However, this study was conducted before 1995, and microbial resistance patterns have changed since then. More recently, five-day treatment courses with azithromycin and telithromycin (Ketek) were found to be effective.24,25
TREATMENT FAILURE
When a patient fails to respond to therapy, additional history, physical examination, cultures, or imaging may be necessary. If a change in antibiotic therapy is made, the limitations in coverage of the initial antibiotic should be considered. A switch to a fluoroquinolone is recommended after failure of amoxicillin or doxycycline.16 Combination therapy may be advantageous, particularly in patients previously treated with cefdinir or macrolides.
Ancillary Treatment
The evidence supporting the use of ancillary treatment for ABRS is relatively weak (Table 2).26–48 Some studies show improvement in symptoms, but no treatments have been shown to affect the duration of illness.26–28 Oral decongestants can be used until symptoms resolve. In patients with stable hypertension, decongestants have not been shown to seriously increase blood pressure.29 Decongestants should be used with caution in patients with ischemic heart disease, glaucoma, or prostatic hypertrophy.
TABLE 2 Ancillary Treatment for Acute Bacterial Rhinosinusitis
| Agent | Dosage | |
|---|---|---|
| Likely to be effective | ||
| Oral decongestants | ||
| Pseudoephedrine (Sudafed) | 60 mg every 6 hours or 120 mg every 12 hours | |
| Topical decongestants* | ||
| Oxymetazoline (Afrin) [ | 2 sprays every 12 hours | |
| Xylometazoline (Otrivin) | 2 sprays every 8 hours | |
| Phenylephrine (Neo-Synephrine) [ | 2 sprays every 4 hours | |
| Possibly effective | ||
| Topical anticholinergics | ||
| Ipratropium (Atrovent) 0.06 percent | 2 sprays every 6 hours | |
| Antihistamines† | ||
| Brompheniramine (Dimetapp) | 8 to 12 mg every 12 hours | |
| Chlorpheniramine (Chlor-Trimeton) | 8 to 12 mg every 12 hours | |
| Diphenhydramine (Benadryl) | 25 to 50 mg every 6 hours | |
| Guaifenesin (Hytuss)† | 600 mg every 12 hours | |
| Nasal corticosteroids | Dosage varies‡ | |
| Hypertonic and normal saline nasal irrigation | Regimen varies | |
| No proven benefit | ||
| Saline spray | ||
| Mist | ||
| Less sedating antihistamines | ||
| Zinc salt lozenges | ||
| Echinacea extract | ||
| Vitamin C | ||
*—Although topical decongestants are effective, use must be limited to three days to avoid rebound congestion.
†—Often combined with an oral decongestant.
‡—Dosage varies by drug.
Although topical decongestants have been advocated in the past, their use is more controversial. Symptoms are improved in some patients, but reduced mucosal blood flow may increase inflammation.30 Topical decongestants should not be used longer than three days to avoid rebound vasodilation.
No clinical studies support the use of antihistamines for treatment of patients with ABRS. Even though histamine does not play a role in this infectious condition except, possibly, in patients who also have a predisposing allergic rhinitis, these drugs have some anti-inflammatory effects that may be beneficial.31 However, the anticholinergic effects of first-generation antihistamines could impair clearance by thickening mucus.31 Newer second-generation antihistamines have little or no anticholinergic effect and may have a role in treatment of patients with allergy and chronic sinusitis, but are not recommended for ABRS.48
The topical anticholinergic agent ipratropium bromide (Atrovent) has been used to decrease rhinorrhea in patients with the common cold,32 but there are no studies in patients with ABRS. Theoretically, ipratropium may increase the viscosity of mucus and impair its clearance, but this effect appears to be less prominent with ipratropium than with antihistamines.31
Most studies of intranasal steroids in patients with ABRS have not shown an effect on clinical outcomes. These studies often were underpowered and included patients who had chronic sinusitis and nasal polyposis, as well as ABRS.33–36 A recent RCT,37 limited to patients with a history of previous recurrent or chronic sinusitis, compared fluticasone (Flovent) with placebo in the treatment of patients with ABRS. Both groups received cefuroxime and topical xylometazoline (Otrivin) for three days. Patients who received fluticasone showed more rapid improvement (6.0 versus 9.5 days) than patients who received placebo.
Nasal saline spray, nasal irrigation, and misthumidificationhavebeenrecommended in the past to promote mucociliary clearance by decreasing congestion, moistening the nasal cavity, and removing crusty mucus.31 Most trials have been small, many were not controlled, and methods varied, so evidence supporting their use is only fair.27,28,38 Saline irrigation is safe, and there are no documented serious adverse effects.38 Hypertonic saline irrigation improved symptoms and decreased medication use in patients with chronic sinusitis.39,40 Saline sprays have been shown to reduce symptoms of rhinitis,41 but there are no studies in patients with ABRS. Controlled studies of mist use in URI have not shown a benefit.42,43
Guaifenesin (Hytuss), a mucolytic agent, should improve mucus clearance by thinning secretions, but there is no evidence of clinical benefit. An RCT showed no effect on mucociliary clearance in healthy subjects.44 However, guaifenesin did reduce nasal congestion in an RCT of patients infected with human immunodeficiency virus who had acute or chronic rhinosinusitis.45
There is insufficient evidence to recommend the use of vitamin C, zinc salt lozenges, or echinacea in patients with ABRS. Using the outcome of cold symptoms after seven days, a meta-analysis of eight clinical trials of zinc salt–lozenge treatment for the common cold did not find a significant benefit (odds ratio, 0.50; 95 percent confidence interval, 0.19 to 1.29).46 Several trials of echinacea extract in the treatment of the common cold reported a mild benefit, but each trial had serious methodologic flaws. A recent RCT of echinacea in college students with common colds showed no benefit.47
Complications and Referral
Patients with complications or treatment failure after extended antibiotic therapy should be referred to an otolaryngologist (Table 3).6,48 Patients who are referred to otolaryngologists usually are evaluated with nasal endoscopy and a sinus computed tomographic scan. Patients with frequent recurrences of ABRS and inadequately controlled allergic rhinitis should be referred to an allergist for consideration of immunotherapy.
TABLE 3 Indications for Referral in Patients with Bacterial Rhinosinusitis
| Findings of severe acute bacterial rhinosinusitis | |
| Complications | |
| Periorbital cellulitis | |
| Intracranial abscess | |
| Meningitis | |
| Cavernous sinus thrombosis | |
| Pott’s puffy tumor (infectious erosion of the ethmoid or frontal sinus) | |
| Anatomic defects causing obstruction | |
| Treatment failure after extended course of antibiotics | |
| Frequent recurrences (more than three episodes per year) | |
| Nosocomial infections | |
| Immunocompromised host | |
| Biopsy to rule out granulomatous disease, neoplasms, or fungal infections | |
| Evaluation for immunotherapy of allergic rhinitis | |

