There is a common misconception that symptomatic tendon injuries are inflammatory; because of this, these injuries often are mislabeled as “tendonitis.” Acute inflammatory tendinopathies exist, but most patients seen in primary care will have chronic symptoms suggesting a degenerative condition that should be labeled as “tendinosus” or “tendinopathy.” Accurate diagnosis requires physicians to recognize the historical features, anatomy, and useful physical examination maneuvers for these common tendon problems. The natural history is gradually increasing load-related localized pain coinciding with increased activity. The most common overuse tendinopathies involve the rotator cuff, medial and lateral elbow epicondyles, patellar tendon, and Achilles tendon. Examination should include thorough inspection to assess for swelling, asymmetry, and erythema of involved tendons; range-of-motion testing; palpation for tenderness; and examination maneuvers that simulate tendon loading and reproduce pain. Plain radiography, ultrasonography, and magnetic resonance imaging can be helpful if the diagnosis remains unclear. Most patients with overuse tendinopathies (about 80 percent) fully recover within three to six months, and outpatient treatment should consist of relative rest of the affected area, icing, and eccentric strengthening exercises. Although topical and systemic nonsteroidal anti-inflammatory drugs are effective for acute pain relief, these cannot be recommended in favor of other analgesics. Injected corticosteroids also can relieve pain, but these drugs should be used with caution. Ultrasonography, shock wave therapy, orthotics, massage, and technique modification are treatment options, but few data exist to support their use at this time. Surgery is an effective treatment that should be reserved for patients who have failed conservative therapy.
Tendon injury may be secondary to acute trauma (e.g., rupture, laceration) or repetitive loading (e.g., overuse injury). The latter is the focus of this article and is the most common type of tendon problem seen by family physicians. Overuse injuries, including tendinopathies, account for nearly 7 percent of all injury-related physician office visits in the United States.1 Hundreds of thousands of employees are affected by overuse tendinopathies each year, causing significant loss of work time.2 Understanding the pathophysiology, diagnostic criteria, and treatment of tendinopathies can help reduce pain, hasten return to function, and reduce the economic impact of lost productivity in the workforce.
SORT: KEY RECOMMENDATIONS FOR PRACTICE
| Clinical recommendation | Evidence rating | References |
|---|---|---|
| Relative rest and reduced activity prevent further damage and promote healing and pain relief. There are no clear recommendations for the duration of rest and avoidance of activity. | C | * |
| Cryotherapy provides acute relief of tendinopathy pain and its use is widely accepted. Repeated applications of melting ice water through a wet towel for 10-minute periods are most effective. | B | 14 |
| Eccentric strengthening is an effective treatment of tendinopathy and may reverse degenerative changes. | B | 15–17 |
| NSAIDs are recommended for short-term pain relief but have no effect on long-term outcomes. Topical NSAIDs are effective and may have fewer systemic side effects. It is unclear whether NSAIDs are better than other analgesics. | B | 18–20 |
| Locally injected corticosteroids may be more effective than oral NSAIDs in acute-phase pain relief but do not alter long-term outcomes. | B | 21–23 |
| No conclusive recommendations can be made for the use of orthotics and braces in patellar tendinopathy or elbow tendinopathy. Clinical experience and patient preference should guide therapy. | B | 24,25 |
| Therapeutic ultrasonography, corticosteroid iontophoresis, and phonophoresis are of uncertain benefit for tendinopathy. | B | 27,30 |
| Extracorporeal shock wave therapy appears to be a safe, noninvasive, effective but expensive means of pain relief for a number of chronic tendinopathies. | B | 28,29 |
| Surgery is an effective option in carefully selected patients who have failed three to six months of conservative therapy. | B | 31,32 |
NSAIDs = nonsteroidal anti-inflammatory drugs.
*—Generally accepted practice.
A = consistent, good-quality, patient-oriented evidence; B = inconsistent or limited-quality, patient-oriented evidence; C = consensus, disease-oriented evidence, usual practice, expert opinion, or case series. For information about the SORT evidence rating system, see page 736 orhttps://www.aafp.org/afpsort.xml.
Pathophysiology
Tendon is composed of densely arranged collagen fibers, elastin, proteoglycans, and lipids. It is sheathed by the epitenon, which contains the tendon’s neurovascular supply. Muscular force is transmitted to the skeleton at the point where the tendon inserts into bone. This osteotendinous junction is the most common site of overuse tendon injury, but problems can occur throughout.
Tendons are relatively hypovascular proximal to the tendon insertion.3 This hypovascularity may predispose the tendon to hypoxic tendon degeneration and has been implicated in the etiology of tendinopathies.
Tendinopathy classification remains problematic and encompasses a variety of histopathologic entities. One classification scheme is as follows: (1) acute tendonitis alone, (2) chronic tendinosus with acute tendonitis, and (3) chronic tendinosus alone4 (Table 15). Although true inflammatory tendinopathies exist, most patients have prolonged symptoms before presentation to a family physician, by which time acute inflammation probably has subsided and been supplanted by degeneration of the normally highly arranged collagen fiber structure.
TABLE 1 Comparison of Overuse Tendinosus and Overuse Tendonitis
| Comparison factors | Tendinosus | Tendonitis |
|---|---|---|
| Prevalence | Common | Uncommon |
| Time for full recovery (initial) | Two to three months | Two to three days |
| Time for full recovery (chronic) | Three to six months | Four to six weeks |
| Likelihood of full recovery | About 80 percent | 99 percent |
| Focus of conservative therapy | Encourage collagen synthesis, maturation, and strength | Anti-inflammatory therapy and drugs |
| Role of surgery | Excise abnormal tissue | Not known |
| Prognosis of surgery | 70 to 85 percent | 95 percent |
| Surgical recovery | Four to six months | Three to four weeks |
Adapted with permission from Khan K, Cook J. The painful nonruptured tendon: clinical aspects. Clin Sports Med 2003;22:715.
Histologic descriptions of tendinopathies have demonstrated disordered collagen arrangement together with increased proteoglycan ground substance and neovascularization (Figures 1 and 2).4,6 It is unclear if these chronic degenerative changes are preceded consistently by an acute inflammatory response; therefore, the designation of tendon pain as “tendonitis” may be a misnomer. The terms “tendinopathy” and “tendinosus” are more appropriate and should be used to describe these clinical entities in the absence of biopsy-proven histopathologic evidence of acute inflammation, particularly in patients who have had symptoms for more than a few weeks.
Figure 1.
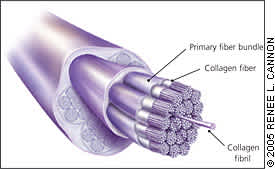
Histology of a normal tendon.
Figure 2.
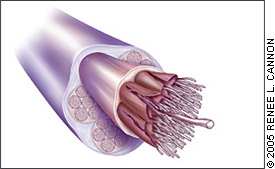
Histology of a damaged tendon. Note the collagen disorientation and fiber separation.
General Diagnostic Approach
HISTORY
The usual presentation of a tendinopathy is insidious onset of load-related localized pain coinciding with increased activity. Most, but not all, patients with tendinopathy will report engaging in a new activity or increasing the intensity of a current activity before the onset of their symptoms. In the early stages, pain is present during activity but may subside after a warm-up period. Pain gradually increases in intensity and duration and may be present at rest in the later stages of tendinopathy. Patients describe their pain as “sharp” or “stabbing” during aggravating activities, but often they describe a “dull” ache immediately after activity and at rest.
PHYSICAL EXAMINATION
Examination includes inspection for muscle atrophy, asymmetry, swelling, erythema, and joint effusions. Atrophy often is present with chronic conditions and is an important clue to the duration of the tendinopathy. Swelling, erythema, and asymmetry are commonly noted when examining pathologic tendons. Joint effusions are uncommon with tendinopathy and suggest the possibility of intra-articular pathology. Range-of-motion testing often is limited on the symptomatic side.
Palpation tends to elicit well-localized tenderness that is similar in quality and location to the pain experienced during activity. Palpation is limited occasionally by the anatomic location of the tendon (e.g., supraspinatus and rotator cuff tendinopathy). The diagnosis of tendinosus is supported by physical maneuvers that simulate tendon loading and predictably reproduce the patient’s pain. The presence of multiple symptomatic tendons should alert the physician to consider evaluation for rheumatic disease or rheumatologic referral.
IMAGING
Plain-film radiography usually is unable to demonstrate the soft-tissue changes of tendinopathy but may reveal bony abnormalities, such as loose bodies or osteoarthritis, that can cause symptoms. Further imaging studies should be reserved for when the diagnosis remains unclear after a thorough history and physical examination, in the case of recalcitrant pain despite adequate conservative management, or for preoperative evaluation. Ultrasonography and magnetic resonance imaging (MRI) are options that can show characteristic changes of tendinosus (Table 2).7–15 Tendon morphology seen on ultrasonography and MRI does not necessarily correlate with clinical symptoms.7
General Approach to Conservative Treatment
It may take up to six months for symptoms of tendinopathy to improve, but most patients eventually respond to conservative treatment. The goal of treatment is to reduce pain and return function. Unfortunately, few methodologically adequate randomized controlled trials (RCTs) have investigated the variety of treatments recommended for patients with tendinosus. Relative rest of the affected area, stretching, ice, and analgesics are all appropriate in the acute phase of tendinopathy pain.
RELATIVE REST
Patients should be encouraged to reduce activity to decrease repetitive loading of the damaged tendon. Relative rest prevents ongoing damage, reduces pain, and may promote tendon healing. There are no clear recommendations for the duration of rest, and different regimens have not been studied in RCTs. Most physicians allow patients to continue activities that do not worsen pain. Complete immobilization should be avoided to prevent muscular atrophy and deconditioning. Tensile loading of the tendon stimulates collagen production and guides normal alignment of newly formed collagen fibers.
ICE
Cryotherapy is effective for short-term pain relief. Icing may slow the release of blood and proteins from the surrounding vasculature by reducing tissue metabolism. Ice may be effective for reducing swelling and pain in cases of acute inflammatory tendinopathies by blunting the inflammatory response. Although specifics about cryotherapy are not well studied, the authors of a recent systematic review16 of cryotherapy for soft-tissue injuries concluded that applications of ice through a wet towel for 10-minute periods are most effective.
STRENGTHENING AND STRETCHING
Once pain has subsided, strengthening and stretching exercises should be considered. Eccentric strength training (Figures 3a and 3b) is particularly effective in treating tendinopathies and helps promote the formation of new collagen.17,18 Eccentric contraction involves the lengthening of muscle fibers as the muscle contracts, preferentially loading the tendon. Eccentric exercise has proved beneficial in Achilles tendinosus17,18 and patellar tendinosus,19 and may be helpful in other tendinopathies. Stretching exercises generally are thought to be helpful and are widely accepted.
Figure 3.
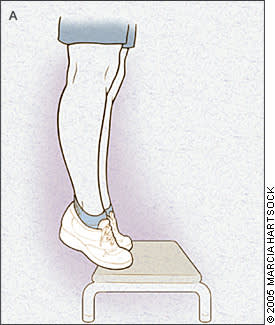
Eccentric exercise (lengthening contraction) for Achilles tendinopathy. (A) A toe raise is performed with both legs. (B) Once the feet are plantar flexed, the uninvolved leg is lifted and the body is slowly lowered into dorsiflexion.
ANALGESICS
Nonsteroidal anti-inflammatory drugs (NSAIDs) effectively relieve tendinopathy pain and may offer additional benefit in acute inflammatory tendonitis because of their anti-inflammatory properties. Topical NSAIDs also reduce tendon pain and eliminate the increased risk of gastrointestinal hemorrhage associated with systemic NSAIDs.20 However, because the majority of chronic tendinopathies are not inflammatory, few data exist to support the use of NSAIDs over analgesics without anti-inflammatory effects.21 In one systematic review,22 investigators found insufficient evidence to recommend or discourage the use of oral NSAIDs in the short-term treatment of tendinopathies.
CORTICOSTEROIDS
Locally injected corticosteroids are a topic of debate, and more research in this area is needed. The optimal drugs, dosages, techniques, intervals, and post-injection care remain unknown. For unclear reasons, injected corticosteroids may be more effective than oral NSAIDs for relief in the acute phase of tendon pain, but they do not tend to alter long-term outcomes.23,24 Unfortunately, no evidence-based guidelines support the use of local corticosteroid injections in tendinopathy, and there may be deleterious effects on the tendon when they are injected into the tendon substance.
The effects of peritendinous corticosteroid injections are unknown, but they should be used with some caution. Because the role of inflammation in tendinopathies is unclear, corticosteroids may serve only to inhibit healing and reduce the tensile strength of the tissue, predisposing to spontaneous rupture.25
ORTHOTICS AND BRACES
Orthotics and braces (e.g., tennis elbow bands, shoe orthotics) are used to reinforce, unload, and protect tendons during activity and are valuable adjuncts to the above therapies. Although few data support definitive conclusions regarding their effectiveness,26,27 orthotics and braces are safe, widely used, and often helpful in correcting biomechanical problems, such as excessive foot pronation or pes planus deformity.
OTHER MODALITIES
Tendinopathies can be treated with ultrasonography, extracorporeal shock wave therapy (ESWT), iontophoresis and phonophoresis, and technique modification. Therapeutic ultrasonography produces high frequency vibrations that generate heat as they penetrate superficial tissues. Ultrasonography may decrease pain and increase the rate of collagen synthesis, but evidence for a consistent benefit in tendinopathies is weak.28,29
ESWT uses acoustic shock waves to impart structural and neurochemical alterations that may reduce pain and promote tendon healing. ESWT appears to be safe and effective, but further research is needed to clarify the mechanisms of action and optimal treatment strategies.30,31
Iontophoresis and phonophoresis use electric and ultrasonographic impulses to deliver topical NSAIDs and corticosteroids to symptomatic subcutaneous tissues. Both are widely used and anecdotally effective, but well-designed RCTs are lacking to permit reliable recommendations.29,32
Technique modification for athletes and manual laborers aims to minimize the repetitive stresses placed on tendons to eliminate pain and promote healing.
SURGERY
Although the mechanism of surgical healing promotion is not entirely understood, surgery is an effective treatment option in carefully selected patients.33,34 If pain persists despite a well-managed conservative treatment trial, referral to an orthopedic surgeon for surgical evaluation may be warranted. Surgical techniques vary by tendinopathy location but typically include excision of abnormal tendon tissue and performance of longitudinal tenotomies to release areas of scarring and fibrosis. Consideration for surgical referral typically occurs after three to six months of failed conservative treatment.
Rotator Cuff Tendinopathy
Rotator cuff tendinopathy affects one in 50 adults35 and is particularly common in athletes who throw repetitively and in laborers who have to work with their arms overhead. The cuff is composed of the subscapularis anteriorly, the supraspinatus superiorly, and the teres minor and infraspinatus posteriorly. Improper functioning and muscle weakness permit the humeral head to migrate superiorly, resulting in supraspinatus tendon impingement. The supraspinatus complex occupies a narrow space, with light contact between the supraspinatus and the coracoacromial arch during normal abduction. Repeated impingement of the coracoacromial arch onto the supraspinatus tendon has been implicated as the likely mechanism of tendon injury. Hypovascularity in the region proximal to the insertion of the supraspinatus tendon also may have implications in rotator cuff tendinopathy.36
Supraspinatus tendon impingement is most apparent with the humerus simultaneously abducted and internally rotated. The Hawkins’ test (Figure 4) elicits pain with forcible internal rotation with the patient’s arm passively flexed forward at 90 degrees. It is sensitive (92 percent) but not specific (25 percent) for impingement. The Neer’s test (Figure 5) elicits pain with full forward flexion between 70 and 120 degrees. It too is sensitive (88 percent), but not specific (33 percent).37,38 See Table 27–15 for a summary of diagnostic imaging for rotator cuff tendinopathy.
Figure 4.
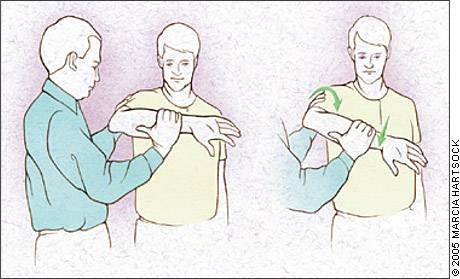
The Hawkins’ test for subacromial impingement or rotator cuff tendinosus. The arm is forward, elevated to 90 degrees, then forcibly internally rotated.
Figure 5.
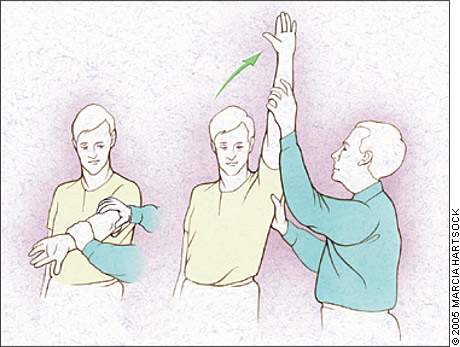
The Neer’s test for impingement of the rotator cuff tendons under the coracoacromial arch. The arm is fully pronated and placed in forced flexion.
Elbow Tendinopathy
Lateral epicondylosis is seven to 10 times more common than medial epicondylosis; it involves the dominant arm 75 percent of the time.39 Elbow tendinopathies affect men and women equally and are most common after 40 years of age. Activities that require repetitive wrist extension contribute to the development of lateral epicondylosis. The condition commonly occurs in persons who play racquet sports (“tennis elbow”) and often is a problem in occupations requiring repetitive wrist flexion and extension. Medial epicondylosis is associated with repetitive wrist flexion, which is common among athletes who throw repetitively, golfers (“golfer’s elbow”), and manual laborers.
Patients with lateral epicondylosis often report insidious lateral elbow pain that is exacerbated by wrist extension activities. Palpation produces point tenderness over or just distal to the lateral epicondyle where the extensor tendons insert. Pain may be reproducible with resisted wrist extension. In the case of medial epicondylosis, patients will have medial elbow pain exacerbated with wrist flexion and pronation. Palpation produces tenderness over the medial epicondyle near the insertion of the wrist flexor tendons. Pain is reproducible with resisted wrist flexion and pronation. Table 319, 24–26, 30, 40 provides a summary of the evidence for treatments of elbow tendinopathy.
TABLE 3 Treatment Recommendations for Specific Tendinopathies
| Elbow tendinopathy |
| Corticosteroid injections for lateral epicondylopathy are more effective than NSAIDs in the acute phase of epicondylosis but do not change long-term outcomes of pain. |
| Studies of extracorporeal shock wave therapy have demonstrated mixed benefit for lateral elbow pain. |
| Orthotics commonly are used, although no definitive conclusions concerning their effectiveness can be drawn from existing clinical trials. |
| Patellar tendinopathy |
| Eccentric exercise is recommended to reduce symptoms, increase strength, and promote tendon healing. |
| Deep transverse friction massage is recommended to reduce pain. |
| Corticosteroid iontophoresis is effective for treatment of patellar tendinosus pain and function. |
NSAIDs = nonsteroidal anti-inflammatory drugs.
Patellar Tendinopathy
Patellar tendinopathy causes activity-related anterior knee pain and occurs in athletes and nonathletes alike. Repetitive loading of the patellar tendon places jumping athletes at greatest risk. Pain that is exacerbated by navigating stairs and prolonged sitting is common. Physical examination may elicit pain at the inferior pole of the patella with the leg fully extended using resisted leg extension. A decline squat test (Figure 6) places increased load on the patellar tendon and often reproduces a patient’s pain. See Table 27–15 for a summary of diagnostic imaging for patellar tendinopathy and Table 319,24–26,30,40 for a summary of the evidence for treatment of patellar tendinopathy.
Figure 6
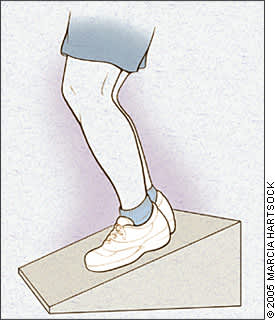
Decline squat test for patellar tendinopathy. This test places greater load on the patellar tendon than does a squat performed on level ground.
Achilles Tendinopathy
Achilles tendinopathy is common among adult runners, with a higher incidence as age increases. The etiology and pathogenesis of Achilles tendinosus tend to be multifactorial (e.g., improper technique, anatomic misalignment, weakness, uneven training surfaces, improperly fitted footwear). Most patients have posterior heel pain proximal to the tendon insertion. Physical examination often reveals thickening and tender, palpable nodules. Special attention should be paid to anatomic deformities such as forefoot and heel varus and excessive pes planus or foot pronation, which often are associated with this problem. Imaging rationale is similar to that of previously discussed tendinopathies.
Treatment should be conservative. Eccentric strength training is a particularly popular and effective option.17,18,41 Stretching and deep friction massage of the gastrocnemius-soleus complex generally are considered helpful for Achilles tendinopathy. Anatomic misalignment can be treated with shoe orthotics to correct overpronation or pes planus problems. Many patients will benefit from heel lift orthotics to unload the tendon for pain relief.

