Bariatric surgery leads to sustainable long-term weight loss and may be curative for such obesity-related comorbidities as diabetes and obstructive sleep apnea in severely obese patients. The Rouxen-Y gastric bypass has become the most common procedure for patients undergoing bariatric surgery. The procedure carries a mortality risk of up to 1 percent and a serious complication risk of up to 10 percent. Indications include body mass index of 40 kg per m2 or greater, or 35 kg per m2 or greater with serious obesity-related comorbidities (e.g., diabetes, obstructive sleep apnea, coronary artery disease, debilitating arthritis). Pulmonary emboli, anastomotic leaks, and respiratory failure account for 80 percent of all deaths 30 days after bariatric surgery; therefore, appropriate prophylaxis for venous thromboembolism (including, in most cases, low-molecular-weight heparin) and awareness of the symptoms of common complications are important. Some of the common short-term complications of bariatric surgery are wound infection, stomal stenosis, marginal ulceration, and constipation. Symptomatic cholelithiasis, dumping syndrome, persistent vomiting, and nutritional deficiencies may present as long-term complications.
Sixty-seven percent of Americans are obese or overweight, resulting in combined direct and indirect health care costs estimated to be $117 billion per year.1,2 Common obesity-related comorbidities include coronary artery disease, diabetes, obstructive sleep apnea, osteoarthritis, chronic infections, and psychological disorders (Table 1). The combination of highly available, energy-dense food and the reduction of required daily energy expenditure from the use of modern-day conveniences (e.g., automobiles, elevators, remote controls) most likely contributes to the obesity epidemic.
SORT: KEY RECOMMENDATIONS FOR PRACTICE
| Clinical recommendation | Evidencerating | Reference |
|---|---|---|
| Bariatric surgery leads to sustainable long-term weight loss and may reduce obesity-related comorbidities such as diabetes mellitus and obstructive sleep apnea. | A | 5 |
| Patients should undergo multispecialty preoperative evaluation by a team with medical, surgical, psychological, and nutritional experience before bariatric surgery. | C | 7 |
| Prophylaxis to prevent venous thromboembolism using low-molecular-weight heparin is indicated for most patients. | A | 17 |
A = consistent, good-quality patient-oriented evidence; B = inconsistent or limited-quality patient-oriented evidence; C = consensus, disease-oriented evidence, usual practice, expert opinion, or case series. For information about the SORT evidence rating system, see page 1313 orhttps://www.aafp.org/afpsort.xml.
TABLE 1 Obesity-Related Comorbidities
| System | Comorbidities |
|---|---|
| Cardiovascular | Hypertension, hyperlipidemia, cardiomyopathy, long QT syndrome, atrial fibrillation |
| Pulmonary | Reactive airway disease, obstructive sleep apnea, restrictive lung disease |
| Musculoskeletal | Debilitating osteoarthritis, chronic low back pain, immobility |
| Psychological | Binge-eating disorder, depression, body dysmorphic disorder |
| Dermatologic | Intertrigo, venous stasis, decubitus ulcer, acanthosis nigricans |
| Endocrine | Diabetes mellitus, hypoandrogenemia, metabolic syndrome, polycystic ovarian syndrome |
| Genitourinary | Ovarian cancer, uterine cancer, breast cancer, renal cell cancer, dysfunctional uterine bleeding |
| Gastrointestinal | Irritable bowel syndrome, nonalcoholic fatty liver disease, gastroesophageal reflux, esophageal cancer, colon cancer |
| Neurologic | Ischemic stroke, meralgia paresthetica |
The number of bariatric surgeries performed in the United States has recently increased 10-fold, from 14,000 in 1993 to an estimated 140,000 in 2004.3 A number of studies4–6 have demonstrated that bariatric surgery leads to sustainable long-term weight loss and, in many patients, may be curative for obesity-related comorbidities such as diabetes and obstructive sleep apnea.
As more patients have bariatric surgery, it is important for the family physician to be knowledgeable about the risks and benefits of the procedure and to understand the complexities of the lifelong medical surveillance that these patients require.
The Roux-en-Y gastric bypass (RYGB) has become the most commonly performed procedure for patients undergoing bariatric surgery.5–7 It has the advantages of providing a restrictive and a malabsorptive component to induce weight loss.
Indications for Bariatric Surgery
In 1991, the National Institutes of Health (NIH) convened a multidisciplinary panel to review the role of surgery for severe obesity and help establish guidelines for patient selection (Table 2).8 Based on available evidence, the panel essentially sanctioned two procedures: RYGB and vertical banded gastroplasty (VBG).
RYGB involves partitioning the stomach into a functional 20-mL pouch with an outlet to an anastomosed loop of small intestine (Figure 1). This creates a restrictive (small stomach pouch) and malabsorptive (bypassed section of small intestine) component for weight loss.
Figure 1.
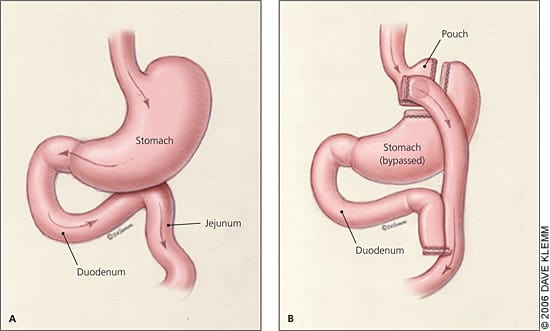
(A) Stomach before bariatric surgery. (B) Stomach after Roux-en-Y gastric bypass procedure; food is redirected to the middle portion of the small intestine, limiting absorption of calories.
VBG creates a small stomach pouch with an outlet that lies along the lesser curvature of the stomach, resulting in only a restrictive component for weight loss (Figure 2). Newly developed procedures include the adjustable gastric band (LapBand) (Figure 3), which is potentially reversible; and biliopancreatic diversion, which is highly effective in super-obese patients (i.e., body mass index [BMI] higher than 50 kg per m2). These procedures were not addressed by the NIH panel in 1991.
Figure 2.
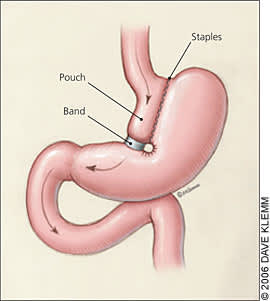
Vertical banded gastroplasty divides the stomach using staples and directs the food into a small pouch, limiting caloric intake.
Figure 3.
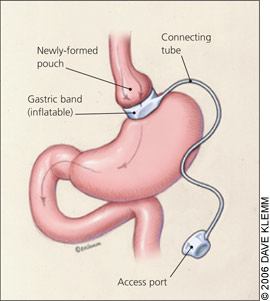
Adjustable gastric banding involves inserting an inflatable ring, usually laparoscopically, which can be adjusted via a subcutaneous access point.
Weight loss varies depending on the type of procedure, initial BMI, and compliance with recommended postoperative behavior modification. Restrictive procedures such as VBG and adjustable gastric banding result in long-term weight loss of approximately 40 percent of excess body weight, whereas RYGB and biliopancreatic diversion result in an average long-term weight loss of 60 percent.5
It is recommended that, before bariatric surgery, potential patients be evaluated by a team with medical, surgical, psychological, and nutritional expertise.8 Potential candidates for bariatric surgery should be selected carefully based on the criteria in Table 28 and only after a thorough multidisciplinary evaluation. It is important that family physicians be aware of which patients may be candidates for the surgery, understand basic risks and benefits of the surgery, and be able to answer questions and serve as advisors for their patients.
TABLE 2 Indications for Bariatric Surgery from the National Institutes of Health
| BMI of 40 kg per m2 or higher, or BMI of 35 kg per m2 or higher with serious obesity-related comorbidities (e.g., diabetes, obstructive sleep apnea, coronary artery disease, debilitating arthritis) |
| Previous failed weight loss attempts involving an integrated nonsurgical weight loss program including dietary modification, behavioral support, and appropriate exercise |
| Possession of appropriate motivation and psychological stability to understand risks and benefits of the procedure as well as the commitment to lifelong postoperative lifestyle changes and medical surveillance |
note: A good candidate for bariatric surgery will meet all three indications.
BMI = body mass index.
Information from reference8.
Risks of Surgery
RYGB is a major intra-abdominal surgery that involves anatomic changes to the stomach and small intestine. The procedure carries a mortality risk of up to 1 percent and a serious complication risk of up to 10 percent (Table 39–14)15. Factors including preexisting comorbidities, experience of the surgeon, and hospital volume of procedures act as independent predictors of complications.16
TABLE 3 Complications of Roux-en-Y Gastric Bypass Surgery
| Complication | Incidence (%) |
|---|---|
| Stomal stenosis | 9 to 20* |
| Incisional hernia | 2 to 20 |
| Small bowel obstruction | 2 to 10* |
| Marginal ulcer | 1 to 16 |
| Wound infection | 1 to 15 † |
| Bleeding | 5 |
| Anastomotic leak | 2 |
| Deep venous thrombosis/pulmonary embolus | 2 |
| Operative mortality | 1 |
*—Incidence higher in laparoscopic procedure.
†—Incidence higher in open procedure.
LIFE-THREATENING COMPLICATIONS
The International Bariatric Surgery Registry was founded in 1986 and provides data on 35,000 patients.9 According to this registry, the leading cause of death following bariatric surgery is pulmonary embolism. Risk factors for pulmonary embolism include a BMI of 60 kg per m2 or higher, chronic lower extremity edema, obstructive sleep apnea, and previous pulmonary embolism.10 Based on guidelines from the American College of Chest Physicians, low-molecular-weight heparin and compression stockings should be used to minimize risk of pulmonary embolism for most patients.17 Patients at moderate risk of pulmonary embolism should receive prophylacticdoses of low-molecular-weight heparin (up to 3,400 U per day), whereas high-risk patients should be considered for therapeutic doses (more than 3,400 U per day).17
Anastomotic leaks and respiratory failure are other possible causes of death in patients who have had bariatric surgery. Signs of anas tomotic leak are sustained tachycardia, severe abdominal pain, fever, rigors, and hypotension. Work-up should include an upper gastrointestinal series or computed tomography scan with contrast and a prompt surgical consultation. Exploratory surgery may be the only way to determine if there is an anastomotic leak in patients with equivocal symptoms or diagnostic imaging studies.
Together, pulmonary emboli, anastomtic leaks, and respiratory failure account for 80 percent of all deaths in the first 30 days following bariatric surgery. Family physicians can help prevent deaths from these major complications through appropriate prophylaxis for thromboembolic events, identifying symptoms early, and ensuring that patients are educated about and understand the importance of early reporting of such symptoms.
SHORT-TERM COMPLICATIONS
Some common short-term complications of bariatric surgery are wound infections, stomal stenosis (i.e., narrowing of the gastrojejunostomy), marginal ulceration, and constipation. Wound infections are more common in open than in laparoscopic procedures and may occur in up to 20 percent of patients.11,12 An appropriate history and physical examination nation should be conducted to rule out early signs of wound infection, which may include fever, pain, erythema, and purulence around the surgical site. Wound infection may present up to three weeks after surgery in obese patients and should be treated aggressively with drainage and broad-spectrum antibiotics to optimize wound healing. Early wound infections also are associated with a higher risk of subsequent incisional hernias.11
Vomiting and intolerance to liquid meals are abnormal after RYGB and may suggest an organic cause other than overeating. Stomal stenosis occurs in 9 to 20 percent of patients after RYGB surgery and results in persistent vomiting.13 Stomal stenosis can be diagnosed with an upper gastrointestinal series and often is correctable by endoscopic dilation.
Marginal ulceration along the surgical anastomotic site occurs in 1 to 16 percent of patients.14 Hematemesis, melena, and orthostatic hypotension may be early signs of a bleeding ulcer. The complex anatomy of RYGB makes it difficult to access the excluded stomach; however, an endoscopic examination of the gastric pouch should be done. Long-term avoidance of nonsteroidal anti-inflammatory drugs may reduce the incidence of recurrent marginal ulcer formation in these patients.
Constipation is a common short-term consequence of bariatric surgery. It often is caused by pain medicine or dehydration from poor fluid intake and malabsorption and should be treated accordingly. Granular bulking agents should be avoided because they have been reported to lead to esophageal obstruction in these patients.18
LONG-TERM COMPLICATIONS
In addition to the above-mentioned complications, which also can occur in the long term, special attention should be given to symptomatic cholelithiasis, dumping syndrome, persistent vomiting, and nutritional deficiencies. Cholelithiasis is a common consequence of rapid weight loss in the postoperative period and occurs in up to 50 percent of patients.19 Weight loss is most rapid in the first six months after surgery but usually continues at a slower pace for up to two years. Prophylactic cholecystectomy during the surgery or the use of bile salt therapy remain the mainstays of treatment. Evidence-based guidelines on patient selection for prophylactic cholecystectomy are lacking, and this decision is based on patient risk stratification made by the bariatric surgeon.
Dumping syndrome is a constellation of procholinergic symptoms resulting from an influx of undigested carbohydrates into the jejunum. It is a side effect of malabsorptive bariatric procedures such as RYGB and biliopancreatic diversion. Symptoms include nausea, vomiting, diarrhea, tachycardia, salivation, and dizziness. Dumping syndrome results from poor dietary compliance and can serve as a motivational tool for patients to avoid nonpermissible foods.20 It is self-limited and usually subsides one to two hours after consuming sweet foods or those high in simple carbohydrates.
It is important that patients who have had bariatric surgery adhere to specific dietary recommendations. Failure to do so may result in episodes of vomiting caused by pouch distention. Patients require lifelong adjustment in eating behavior, including cutting food into small portions, chewing food thoroughly before swallowing, eating slowly, and waiting one hour after a meal before drinking anything.21 Recurrent vomiting is more common with purely restrictive procedures such as VBG and adjustable laparoscopic banding. Pain medication, vitamin supplements, and dehydration are other causes of postoperative nausea and vomiting.
Patients undergoing malabsorptive procedures (i.e., RYGB and biliopancreatic diversion) are at higher risk of nutritional deficiencies compared with restrictive procedures (i.e., VBG and adjustable laparoscopic banding).22 Adherence to an appropriate diet high in protein and vitamin supplementation is an effective way to avoid this complication.23 Patients require lifelong supplementation with a high-potency multivitamin with iron, vitamin B12 (1,000 mcg intramuscularly monthly or 100 mcg orally daily), and calcium (1,200 mg daily).22–24 Menstruating women may develop anemia refractory to oral iron supplementation and may require parenteral iron infusions. The family physician plays an important role in ensuring long-term compliance with nutritional supplementation, which often is neglected by patients in the years after their surgery, and in ensuring that patients receive appropriate lifelong routine monitoring (Table 4). The family physician may be the only medical professional involved years after the surgery is performed who can ensure that patients meet their overall nutritional needs.
TABLE 4 Common Monitoring Parameters for Bariatric Surgery Patients
| Follow-up period | Laboratory tests |
|---|---|
| Every three months for first year | Complete blood count, glucose, creatinine |
| Every six months for first year | Liver function tests, protein and albumin, iron, total iron-binding capacity, ferritin, vitamin B12, folic acid, calcium, parathyroid hormone (if hypercalcemic) |
| Every year after the first year | All of the above laboratory tests |
Pregnancy is contraindicated for at least 18 months after surgery because of the rapid weight loss and nutritional requirements. The family physician plays an additional role for women of childbearing age in developing an appropriate contraception strategy.
A small subset of patients will develop protein-calorie malnutrition months to years after bariatric surgery because of anastomotic strictures or food phobias. Typically, these patients have had repeated episodes of nausea and daily vomiting and multiple hospitalizations for dehydration, renal insufficiency, and liver failure. Whereas the reflexive nature of inexperienced providers is to recommend reversal of the bariatric procedure, the first and most important step is to reverse the protein-calorie malnutrition by aggressive total parenteral nutrition, dilation of strictures, and later institution of enteral nutrition until all the physical and biochemical manifestations of malnutrition have resolved.
Panniculitis (i.e., severe infection of the excess abdominal wall skin) is common after bariatric surgery and should be treated with antibiotics and skin hygiene. The most effective treatment for recurrent panniculitis is surgical excision of the excess skin, which sometimes weighs 22 to 33 lb (10 to 15 kg).

