A 73-year-old woman presented with a three-week history of exertional dyspnea, nonproductive cough, and intermittent subjective fevers. Her medical history included rheumatoid arthritis with stable pleuropulmonary involvement, which was treated initially with corticosteroids. Eight months before presentation, the patient started taking methotrexate in addition to the corticosteroid regimen because of poor clinical response to and side effects from the original treatment. Methotrexate therapy improved joint symptoms and allowed for tapering of the steroids. The patient also had a history of breast cancer that was treated with curative mastectomy 17 years earlier; no radiation or chemotherapy had been used. She denied tobacco use or occupational pulmonary risk factors.
On presentation, the patient was afebrile and had tachypnea, fine bibasilar inspiratory rales that were more prominent on the left side than the right, and a left pleuritic rub. Cardiac impulse was displaced to the left with an accentuated second heart sound. She had mild joint thickening with early ulnar deviation, but no clubbing. Her laboratory results were significant for a room-air partial pressure of oxygen of 55 mm Hg and a white blood cell count of 6,900 per mm3 (6.9 × 109 per L).
Chest radiography from 10 months earlier showed minimal reticulonodular prominence in the bibasilar segments. The results of pulmonary function testing during admission showed a severe restrictive ventilatory defect with a diffusion capacity of carbon monoxide at 38 percent of predicted value for age. Her chest radiograph at the time of admission is shown in Figure 1.
Figure 1.
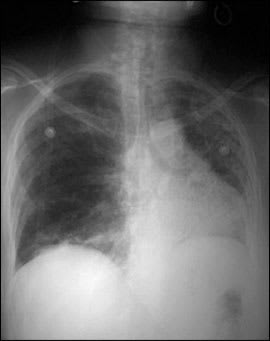
Question
Based on the patient's history and physical examination, which one of the following is the correct diagnosis?
A. Progressive pleuropulmonary rheumatoid arthritis.
B. Pulmonary embolism.
C. Pleuropulmonary methotrexate lung toxicity.
D. Opportunistic pneumonia.
E. Reactivated pulmonary tuberculosis.
Discussion
The answer is C: pleuropulmonary methotrexate lung toxicity. Methotrexate toxicity can manifest in several organ systems, including the lungs, central nervous system, skin, and liver. Each year, pulmonary involvement occurs in 2 to 3 percent of patients with rheumatoid arthritis who are receiving methotrexate.1,2 Pleuropulmonary signs and symptoms include acute to subacute onset of nonproductive cough and dyspnea. Intermittent fevers also are common.
Risk factors include age older than 60, pre-existing rheumatoid pleuropulmonary disease, low serum albumin levels, diabetes mellitus, previous use of disease-modifying antirheumatic drugs (e.g., sulfasalazine [Azulfidine], gold, d-penicillamine), non-sedentary occupations (women), and smoking (men). There is also a direct relationship between the number of extra-articular features of rheumatoid arthritis and risk of methotrexate lung toxicity.1,3
Chronic methotrexate toxicity may be difficult to distinguish from pleuropulmonary rheumatoid arthritis.1 Revised diagnostic criteria for methotrexate lung disease have been proposed (Table 1).1,4,5 The diagnosis can be established in patients with either hypersensitivity pneumonitis histopathology found on a lung biopsy specimen (major criterion 1) or a combination of the other two major criteria plus three of the minor criteria.
Table 1 Revised Diagnostic Criteria For Methotrexate Lung Disease
| Major criteria |
| Hypersensitivity pneumonitis on histopathologic examination; no pathologic organisms |
| Radiographic evidence of pulmonary interstitial or alveolar infiltrates |
| Negative blood cultures (if afebrile) and negative sputum culture |
| Minor criteria |
| Dyspnea for less than eight weeks |
| Nonproductive cough |
| Room-air oxygen saturation of 90 percent or less on initial evaluation |
| Diffusion lung capacity of carbon monoxide of 70 percent or less of predicted value |
| White blood cell count of 15,000 per mm3 (15 × 109 per L) or less |
note:Patient must meet major criterion 1 or major criteria 2 and 3 plus three of the five minor criteria.
Results of chest radiography at the time of admission showed pleural thickening, a loss of volume on the left side with ipsilateral mediastinal shift, and bibasilar reticular pattern with “honeycomb” fibrotic changes (Figure 2). These changes were a progression from radiography performed 10 months earlier (Figure 3).
Figure 2
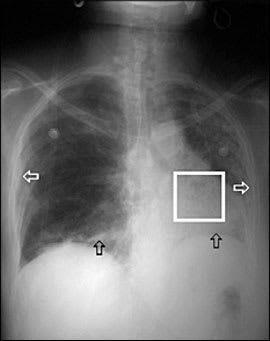
Chest radiograph showing bibasilar reticular inflammation (black arrows), left hemidiaphragm elevation secondary to loss of volume, bilateral pleural thickening (white arrows), and left lower lobe honeycomb fibrosis (white box).
Figure 3.
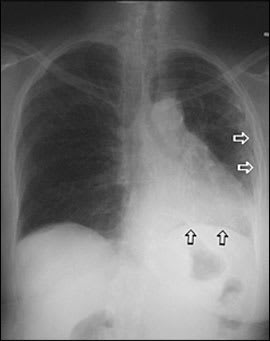
Chest radiograph from 10 months earlier showing unilateral basilar infiltrates and volume loss (black arrows) and mild left-sided pleural thickening (white arrows).
The minimal radiographic changes, an absence of pleuropulmonary symptoms before starting methotrexate therapy, and the onset of symptoms in close proximity to the initiation of methotrexate therapy favor the diagnosis of pleuropulmonary methotrexate lung toxicity over progressive pleuro-pulmonary rheumatoid arthritis.4
The clinical presentation of acute pulmonary embolism includes acute onset of dyspnea, pleuritic chest pain, hemoptysis, and hypoxia. History may reveal risk factors such as recent immobility or a hypercoagulable state.
Acute pulmonary infection would be expected if symptoms included fever and productive cough. Chest radiography often shows focal or diffuse infiltrates, and symptoms improve with appropriate antibiotic treatment. Opportunistic infections may occur in patients with impaired immune systems or those taking immunosuppressive medications.
Reactivated tuberculosis would be unlikely in this patient given the lack of chronic cough, unintentional weight loss, or night sweats. However, chronic steroid use places her at risk. Findings on chest radiography would be expected to show scarring with inflammation and formation of cavities in an apical distribution. A summary of the selected differential diagnosis is provided in Table 2.
TABLE 2 Selected Differential Diagnosis of Progressive Dyspnea and Cough in a Patient with Rheumatoid Arthritis
| Condition | Characteristics |
|---|---|
| Progressive pleuropulmonary rheumatoid arthritis | Insidious onset of dyspnea and nonproductive cough, and fine rales; bibasilar reticular pattern with “honeycombing” on chest radiography; restrictive ventilatory defect on pulmonary function testing |
| Pulmonary embolism | Acute onset of dyspnea, pleuritic pain, cough, and hemoptysis; hypoxia, tachypnea, and tachycardia; deep venous thrombosis and/or risk factors for venous thrombosis |
| Pleuropulmonary methotrexate lung toxicity | Acute to subacute dyspnea, dry cough, and hypoxia; interstitial and alveolar infiltrates on chest radiography; no evidence of infection; improves when medication is discontinued |
| Pulmonary infection | Acute to subacute onset of dyspnea, fever, and cough; tachypnea, rhonchi, and egophony on examination; focal or diffuse infiltrates on chest radiography |
| Tuberculosis | Insidious onset of dyspnea, cough, unintentional weight loss, and night sweats; commonly, radiographic scarring with cavitation and areas of inflammation in apical regions |

