Guideline source: American Academy of Pediatrics
Literature search described? No
Evidence rating system used? No
Published source: Pediatrics, June 2006
Family physicians play a major role in the early recognition and referral of children with developmental delays or mental retardation. Once the physician has recognized a possible developmental problem, she or he can help in determining whether neurologic, audiologic, or ophthalmologic evaluations or rehabilitative services are needed. The American Academy of Pediatrics (AAP) has published guidelines on the clinical evaluation of children with developmental delays or mental retardation. The guidelines focus on the diagnostic importance of the clinical history, family history, dysmorphologic examination, neurologic examination, cytogenic studies, submicroscopic subtelomeric rearrangements, molecular genetic testing, magnetic resonance imaging and computed tomography, and metabolic studies.
Developmental delay is defined as delay in the attainment of developmental milestones at the expected ages; the term usually is reserved for children younger than five years. Mental retardation is defined as a lifelong disability and cannot be diagnosed until a child is five years or older. Identifying possible developmental delay and mental retardation is the role of the family physician.
Once developmental delay is suspected, the family physician has the role of helping the family to understand the results of diagnostic testing and to plan for the future. When a child is diagnosed with developmental delay or mental retardation, the family often experiences feelings of loss of control, and a specific diagnosis can help restore a feeling of control. A comprehensive evaluation includes the clinical history, including prenatal and birth histories; the three-generation family history; physical and neurologic examinations to detect a specific recognizable syndrome or diagnosis (see Table); and sometimes a genetic evaluation (Figure 1).
Selected Clinical Findings and Laboratory Abnormalities Suggesting a Metabolic Disorder
| Abnormal sexual differentiation |
| Arachnodactyly |
| Ataxia |
| Bone abnormalities (e.g., dysostosis, occipital horns, punctuate calcifications) |
| “Coarse” appearance |
| Eye abnormalities (e.g., cataracts, ophthalmoplegia, corneal clouding, abnormal retina) |
| Failure of appropriate growth |
| Hepatosplenomegaly |
| Hyperammonemia |
| Hyperuricemia |
| Hypotonia |
| Loss of psychomotor skills |
| Low cholesterol |
| Metabolic/lactic acidosis |
| Recurrent somnolence/coma |
| Recurrent unexplained illness |
| Seizures |
| Skin abnormalities (e.g., angiokeratoma, “orange-peel” skin, ichthyosis) |
| Structural hair abnormalities |
| Unexplained deafness |
Adapted with permission from Moeschler JB, Shevell M, for the American Academy of Pediatrics Committee on Genetics. Clinical genetic evaluation of the child with mental retardation or developmental delays. Pediatrics 2006;117:2308.
Figure 1. Clinical Genetics evaluation for Developmental Delay and Mental Retardation
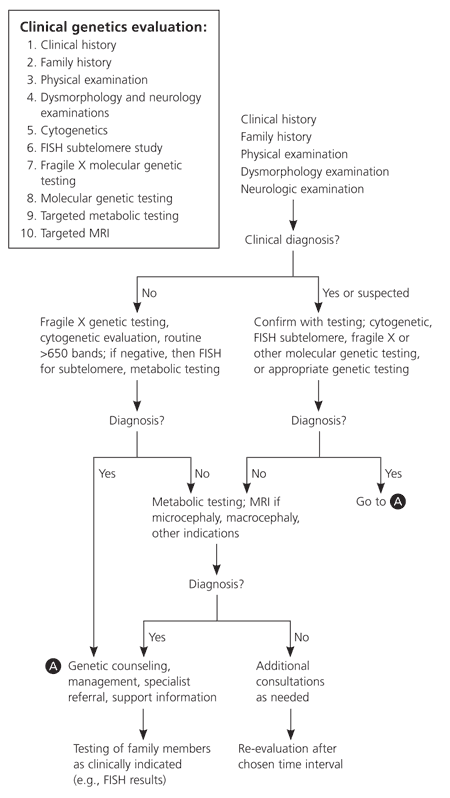
Approach to the clinical genetics evaluation for developmental delay and mental retardation. (FISH = fluorescence in situ hybridization; MRI = magnetic resonance imaging).
Reprinted with permission from Moeschler JB, Shevell M; for the American Academy of Pediatrics Committee on Genetics. Clinical genetic evaluation of the child with mental retardation or developmental delays. Pediatrics 2006;117:2307
Family History
The three-generation family history should focus on family members with mental retardation, developmental delays, psychiatric diagnoses, congenital malformations, histories of miscarriage or stillbirth, and early childhood deaths. Family history does not stand alone for the diagnosis of mental retardation.
Dysmorphologic Examination
Several studies have shown that mental retardation and physical anomalies often coexist, and examination by an experienced clinical geneticist often aids in diagnosis. One study showed that 42 percent of children with developmental delay or mental retardation had three or more minor anomalies.
Neurologic Examination
The neurologic examination (i.e., physical examination focused on detecting neurologic abnormalities) is critical in the evaluation of the child with developmental delays or mental retardation. Studies have shown that 42.9 percent of patients with developmental delay or mental retardation present with neurologic abnormalities. Detection of such abnormalities may signal a need for additional follow-up (e.g., electroencephalography, neuroimaging, genetic testing, referral).
Cytogenic Studies
When the etiology of developmental delay or mental retardation is unknown, cytogenic studies should be performed. Between 9 and 36 percent of patients with mental retardation have chromosomal abnormalities. In one study, four out of 10 patients with mental retardation attributed to chromosomal abnormalities had no dysmorphologic features, emphasizing the need for cytogenic testing.
Submicroscopic Subtelomeric Rearrangements
Fluorescence in situ hybridization (FISH) techniques can examine the subtelomeric regions of each chromosome to look for abnormalities that cause mental retardation. Recent testing has suggested that subtelomeric abnormalities are second only to Down syndrome as the most common cause of mental retardation. In children with mental retardation and normal results on routine chromosome analysis, the FISH technique detects subtelomeric abnormalities in 7.4 percent.
Molecular Genetic Diagnostic Testing
When the cause of mental retardation is suspected or established clinically, molecular genetic diagnostic testing is used to determine the genetic etiology.
FRAGILE X SYNDROME
Fragile X syndrome is the most common genetic cause of developmental delay and mental retardation, and it is advised that children with global developmental delay and all boys with unexplained mental retardation be tested for fragile X syndrome. Routine testing of girls is not needed unless there is increased risk of fragile X syndrome (e.g., family history).
OTHER MOLECULAR GENETIC TESTING
Genetic testing sometimes is used to confirm a clinical diagnosis, to describe the genetic mechanism for a diagnosis, or to improve the precision of genetic counseling. Molecular genetic testing also may be considered in patients who present with atypical features of a known syndrome.
Magnetic Resonance Imaging and Computed Tomography
Malformations of the brain are known to be an important finding in patients with mental retardation, but there is not universal agreement whether imaging should be used routinely as a diagnostic tool in patients with mental retardation. MRI in a young child with developmental delay or mental retardation almost always requires sedation, and MRI leads to a diagnosis in only up to 3.9 percent of patients.
Metabolic Studies
Approximately 1 percent of cases of mental retardation are caused by inborn errors of metabolism. The need for metabolic studies should be determined by the history and examination findings. Routine metabolic screening is not required.
Not all patients will receive an etiologic diagnosis after a complete diagnostic evaluation. These patients should be reevaluated by a clinical geneticist as new diagnostic testing becomes available.

