Breast cancer is one of the most significant health concerns in the United States. Recent reviews have questioned the value of traditional breast cancer screening methods. Breast self-examination has been shown not to improve cancer-specific or all-cause mortality in large studies, but it is commonly advocated as a noninvasive screen. Patients who choose to perform self-examination should be trained in appropriate technique and follow-up. The contribution of the clinical breast examination to early detection is difficult to determine, but studies show that sensitivity is highly dependent on time taken to do the examination. Up to 10 percent of cancers are mammographi-cally silent but evident on clinical breast examination. The U.S. Preventive Services Task Force recommends mammography for women older than 40 years who are in good health, but physicians should consider that sensitivity is lower for younger women. Digital mammography is somewhat more sensitive in younger women and women with dense breasts, but outcome studies are lacking. Although magnetic resonance imaging shows promise as a screening tool in some high-risk women, it is not currently recommended for general screening because of high false-positive rates and cost. The American Cancer Society recommends annual magnetic resonance imaging as an adjunct to screening mammography in high-risk women 30 years and older.
Breast cancer is one of the most significant health concerns in the United States. It is the most commonly diagnosed cancer in women and the second leading cause of cancer death in women.1 The risk of being diagnosed with breast cancer increases with age (Table 1).2 Most primary care physicians have seen the physical and emotional impact that a diagnosis of breast cancer has on patients, their families, and their friends. Breast cancer screening is an important component of health maintenance. Screening significantly contributed to the 23.5 percent decline in breast cancer mortality from 1990 to 2000.3
SORT: KEY RECOMMENDATIONS FOR PRACTICE
| Clinical recommendation | Evidence rating | References | Comments |
|---|---|---|---|
| Although there is good evidence that breast self-examination does not reduce mortality and may increase the rate of biopsy, it may be an option for some women who understand its limitations. | C | 5–8 | The U.S. Preventive Services Task Force reports insufficient evidence to recommend for or against breast self-examination. |
| Clinical breast examination can be part of a periodic health examination for women beginning in their 20s. | C | 8, 19, 39 | The USPSTF reports insufficient evidence to recommend for or against clinical breast examination. |
| Consider mammography every one to two years for women 40 years and older. | B | 8 | The Cochrane Collaboration found screening mammography to be unjustified. |
| Digital mammography is an option for younger women and those with denser breasts, but studies have not proven a mortality benefit. | C | 23 | |
| Magnetic resonance imaging is recommended as an adjunct to screening mammography in women 30 years and older who are at high risk for breast cancer. | C | 34 |
A = consistent, good-quality patient-oriented evidence; B = inconsistent or limited-quality patient-oriented evidence; C = consensus, disease-oriented evidence, usual practice, expert opinion, or case series. For information about the SORT evidence rating system, see page 1605 or https://www.aafp.org/afpsort.xml.
Table 1 Ten-Year Risk of Breast Cancer and Mortality from Breast Cancer
| Age (years) | Chance of being diagnosed with breast cancer within 10 years (%) | Chance of dying from breast cancer within 10 years (%) |
|---|---|---|
| 30 | 0.4 | 0.1 |
| 40 | 1.4 | 0.2 |
| 50 | 2.6 | 0.4 |
| 60 | 3.7 | 0.7 |
| 70 | 4.0 | 0.9 |
Information from reference 2.
However, controversy remains about some aspects of breast cancer screening. Although many physicians recommend the triad of breast self-examination, clinical breast examination, and mammography to screen women who are at average risk, recent studies and recommendations have questioned the appropriateness of each of these modalities. Additionally, there has been increasing attention in the medical literature and lay press regarding the use of digital mammography, magnetic resonance imaging (MRI), scintimammography, and other imaging methods to improve the sensitivity and specificity of screening.
Screening Methods
BREAST SELF-EXAMINATION
Although breast self-examination is often advocated as a noninvasive screening test, most women do not regularly perform it.4 Evidence from large, well-designed, randomized trials of adequate duration has shown that the practice of regular breast self-examination by trained women does not reduce breast cancer–specific or all-cause mortality.5,6 A Cochrane review concluded that breast self-examination has no beneficial effect and actually increases the number of biopsies performed.7 The Cochrane group viewed this as evidence of harm and recommended that women should not perform breast self-examination.7 The U.S. Preventive Services Task Force (USPSTF) found insufficient evidence to recommend breast self-examination.8 One study found that as many cancers were detected incidentally as were found by women trained to do routine breast self-examination.9
If women choose to perform breast self-examination after being informed of the lack of benefit and potential harms, the physician should train the patient in appropriate technique, timing, and follow-up. This allows motivated women to be in control of this aspect of their health care and allows for patient autonomy and education. Performance of the breast self-examination should follow recommendations for the clinical breast examination outlined in the next section. Women who incidentally note a change or lump in their breast should be advised to inform their physician immediately.
CLINICAL BREAST EXAMINATION
Available information suggests that approximately 5 percent of breast cancers are identified solely by clinical breast examination, with pooled data estimating 54 percent sensitivity and 94 percent specificity.10 In a community-based study, only 4 percent of women with an abnormal clinical breast examination suspicious for malignancy had cancer.11 The Canadian National Breast Screening Studies 1 and 2 used clinical breast examination alone compared with clinical breast examination plus mammography in a randomized controlled trial and found similar mortality between groups.12,13 Therefore, the USPSTF found insufficient evidence to recommend for or against clinical breast examination.8
In all other studies of clinical breast examination plus mammography, clinical breast examination contributed to breast cancer detection independent of mammography.14 The independent contribution of clinical breast examination is difficult to determine because of the lack of standardization of clinical breast examination technique. One study found that variation in clinical breast examination technique accounted for 29 percent variance in sensitivity and 33 percent variance in specificity.15
Family physicians should follow the latest guidelines and recommendations regarding technique when performing the breast screening examination to maximize the potential of finding concerning lesions. The clinical breast examination should include visual inspection of the breasts, lymph node evaluation, and palpation of the breast tissue in a systematic pattern, with the breast tissue spread evenly over the chest wall.10,14,16 Figures 1A–1C describe components of the clinical breast examination technique.
The most important component of an adequate clinical breast examination is the time taken to perform the examination. A careful examination of an average-sized breast may take several minutes. This should be explained to patients so they are not surprised or disturbed by the duration and thoroughness of the examination.
Figure 1.
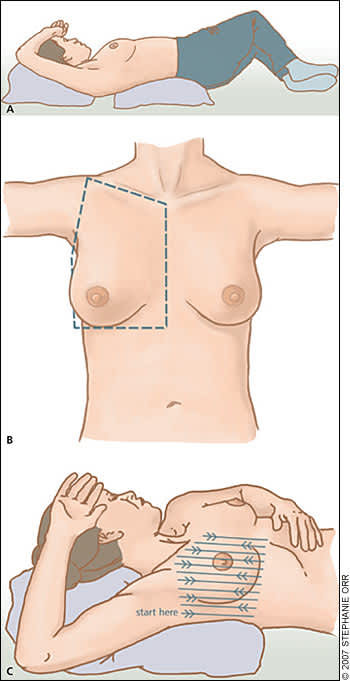
Techniques for breast examination. (A) To spread breast tissue evenly over the chest wall, experts recommend the patient roll on her contralateral hip with her shoulder rotated (so that she is in a supine position, with her ipsilateral hand on her forehead). A pillow or towel can be used underneath the patient's shoulder or lower back to aid in comfort and ensure most effective positioning. (B) The full perimeter of breast tissue should be evaluated during the clinical breast examination. This includes all tissue from the sternum to the inframammary ridge, to the midaxillary line, to the clavicle, and back to the sternum. (C) The pattern of examination should be systematic and performed in vertical strips to ensure all breast tissue is covered.
MAMMOGRAPHY
Several randomized controlled trials have evaluated mammography as a screening test. Most of these studies, begun between 1963 and 1980, reported a decreased risk of breast cancer death in women who were randomized to receive screening, particularly among women between 50 and 69 years of age. However, a meta-analysis questioned the value of mammography as a screening test.17 The authors excluded trials they felt were flawed and found no reduction in mortality with mammography; they concluded that screening for breast cancer with mammography is unjustified.17
The USPSTF performed a meta-analysis using data from the same trials. The researchers concluded that the flaws in some of the studies did not significantly influence outcomes; therefore, they included pooled effects from seven valid studies. The resulting recommendation was for screening mammography every one to two years for women 40 years and older.8
Both of these analyses should be interpreted with the understanding that technology has improved since the original studies were conducted and that increased experience with mammogram interpretation may positively impact the sensitivity and specificity of current mammography. Computer-aided detection systems have been designed to assist radiologists in reading mammograms and are available in many centers. Although these systems are intended to increase sensitivity, a recent study found that sensitivity actually decreases and false-positive results increase when such systems are used.18 The introduction of computer-aided detection systems has not significantly changed overall breast cancer detection rates.19
Studies estimate that the sensitivity of mammography is between 60 and 90 percent.20 The positive predictive value of mammography is higher in women with a family history of breast cancer.21 It is clear that mammography is less sensitive and results in less mortality reduction in younger women.20,21 This decreased benefit may be because of greater mammographic density of breasts and more rapid tumor growth in younger women.22
Digital mammography theoretically maximizes image acquisition and display as compared with film mammography. The overall diagnostic accuracy is similar to film mammography, but digital mammography is more sensitive in women younger than 50 years, pre- or perimenopausal women, and women with radiographically dense breasts.23
ULTRASONOGRAPHY
Because mammography is less sensitive and breast tissue is more dense in younger women, ultrasonography has been considered as a screening tool for younger women who are at high risk for breast cancer. A consensus statement published by the European Group for Breast Cancer Screening concluded that there is no evidence to support the use of ultrasonography for screening at any age.24
There has long been a role for ultrasonography in the work-up of a palpable breast lesion, and there may be a role for correlating ultrasound findings with mammography or MRI findings.25 For now, it is not recommended by any major group as a screening tool, but the American College of Radiology Imaging Network is conducting a multi-center trial of whole breast ultrasonography for screening women at higher risk.26
MAGNETIC RESONANCE IMAGING
The use of MRI as a screening test for breast cancer was first reported in the 1980s, and studies have demonstrated its benefits and limitations. Studies using MRI in high-risk women report that MRI is significantly more sensitive than mammography, and mammographic screening with or without ultrasonography is probably an insufficient screen for persons with a known genetic predisposition for breast cancer.27,28 MRI integrated in surveillance programs may make earlier diagnosis a possibility.29,30 In a recent study of high-risk women, MRI was found to be better at ruling out breast cancer but more likely to produce false-positive results.31,32 The combination of MRI and mammography was better than either alone.33
The American Cancer Society recently recommended that women at high risk of breast cancer undergo annual MRI screening as an adjunct to mammography beginning at age 30 (Table 2).34 However, the high expense and false-positive rates make MRI an inappropriate screening tool for the general population. A report published after the January 2005 international consensus conference described current recommendations, including the evolving role of MRI. The panel agreed that breast MRI may be helpful in several situations (Table 3).35
Table 3 MRI for the Evaluation of Breast Cancer
| MRI may be useful in the evaluation of breast cancer when: | |
| Defining the extent of an index lesion | |
| Determining whether additional foci of malignant disease are present in the ipsilateral breast | |
| Assessing whether contralateral malignant disease might be present | |
| Assessing the response and extent of residual disease after chemotherapy | |
| Evaluating the breasts in patients with newly diagnosed breast cancer who have had breast augmentation with silicone or saline-filled implants | |
| There is suspicion of residual disease in postoperative settings | |
| Mammography, ultrasonography, and clinical findings are inconclusive, and no physical abnormality is apparent | |
MRI = magnetic resonance imaging.
Adapted with permission from Silverstein MJ, Lagios MD, Recht A, Allred DC, Harms SE, Holland R, et al. Image-detected breast cancer: state of the art diagnosis and treatment. J Am Coll Surg 2005;201:587.
SCINTIMAMMOGRAPHY
Clinical studies have been conducted using technetium-99m sestamibi scintimammography to evaluate some breast abnormalities. In a meta-analysis summarizing studies from more than 5,000 patients, the sensitivity and specificity for detecting nonpalpable lesions were found to be 67 and 87 percent, respectively.36 Clinically, this has been used most often to evaluate patients with a palpable breast lesion and a negative mammogram.37 Whereas scintimammography may be of value as an adjunct to mammography and to reduce the rate of negative biopsies, no screening role has been identified.38
POSITRON-EMISSION TOMOGRAPHY
Positron-emission tomography (PET) scanning is based on increased glucose utilization by malignant cells. In the evaluation of suspicious lesions, PET scanning has been found to be reasonably sensitive and specific, but it is limited in detecting some breast tumors based on size, metabolic activity, and histologic subtype.39 There is no evidence demonstrating a clear advantage over other adjuvant imaging studies, and the high cost has limited its use as a routine diagnostic tool.37
DUCTAL LAVAGE
Ductal lavage is a minimally invasive procedure used to access ductal epithelial cells. As such, its value as a potential screening method has been considered. However, in women with known breast cancer, small studies have reported the sensitivity of ductal lavage to be around 20 percent.40,41 Therefore, it has significant limitations as a screening method and is not recommended.
Approach to the Patient
When applying guidelines to individual patients, risk assessment is important. The National Cancer Institute has an online tool that helps physicians estimate the five-year and lifetime breast cancer risk for patients (http://www.cancer.gov/bcrisktool). Another tool is available at http://www.breastcancer-prevention.org/raf_source.asp. More specific tools for women with BRCA1 or BRCA2 genetic defects are also available. A five-year risk of breast cancer of 1.66 percent or higher indicates high-risk status.42
For women 40 years and older who are at average risk, most major health organizations endorse mammographic screening every one to two years, and every year after age 50 as long as the woman is healthy.43 A clear upper limit has not been set, although cost-effectiveness analyses suggest that screening may be worthwhile until a woman has an estimated life expectancy of between five and 10 years.44 The clinical breast examination may also be important, because up to 10 percent of breast cancers may be clinically evident while being silent on mammography.42
For women at high risk, routine screening should be initiated sooner. For those with breast cancer genetic mutations, mammography should begin at age 25, or at an age 10 years younger than the youngest case diagnosed in the family.45 For patients in this age range, mammography is significantly less sensitive, ultrasonography does not improve sensitivity, and the clinical breast examination increases in importance. MRI may increase sensitivity and recently was recommended by the American Cancer Society as an adjunctive screening method in some high-risk women.

