Prompt recognition and appropriate treatment of ocular emergencies are essential in the primary care setting when the outcome may depend on timely management. All ocular emergencies, including a penetrating globe injury, retinal detachment, central retinal artery occlusion, acute angle-closure glaucoma, and chemical burns, should be referred immediately to the emergency department or an ophthalmologist. Careful eye examination and simple tests can help primary care physicians make decisions about appropriate treatment and referral. All patients with eye problems should be tested for visual acuity and ocular movements. Confrontation visual field examination, pupillary examination, and direct ophthalmoscopy of both eyes also should be performed. Ocular injury from high-velocity trauma or from chemicals may be easily misdiagnosed. After a chemical burn, thorough eye washing for at least 30 minutes or until the pH of the eye is within physiologic range is critical to prevent further damage. Use of an eye shield is required in patients with a ruptured globe to protect the injured eye and preserve the patient's vision.
Eye injury, retinal detachment, and central retinal artery occlusion (CRAO) are among the most common ocular emergencies seen in the emergency department.1 It is estimated that about 2 million eye injuries occur every year in the United States, a significant number of which lead to vision loss.2,3
SORT: KEY RECOMMENDATIONS FOR PRACTICE
| Clinical recommendation | Evidence rating | References |
|---|---|---|
| Eye injury from a high-velocity trauma should be immediately evaluated by an ophthalmologist. | C | 8 |
| Patients suspected of having globe rupture should be immediately referred to an ophthalmologist. | C | 8 |
| An eye exposed to chemicals should be irrigated until the pH is within normal range or with at least 1 to 2 liters of normal saline or other solution suitable for eye irrigation. | C | 9–12 |
| Lowering intraocular pressure in acute angle-closure glaucoma may save vision; laser iridotomy is the definitive treatment for acute angle-closure glaucoma. | C | 12,23,24 |
A = consistent, good-quality patient-oriented evidence; B = inconsistent or limited-quality patient-oriented evidence; C = consensus, disease-oriented evidence, usual practice, expert opinion, or case series. For information about the SORT evidence rating system, see page 749 orhttps://www.aafp.org/afpsort.xml.
Initial Evaluation
All patients with an ocular problem should have visual acuity testing. If visual acuity is difficult to assess because of eye pain, topical anesthetic drops can be used. If possible, physical examination of the eye should include the eyelids, globes, orbits, forehead, and cheeks; evaluation of extraocular motility; and pupillary reflex visual field testing (i.e., the confrontation visual field examination). Eye examination clinical findings and interpretations are listed in Table 1.
Table 1 Eye Examination Clinical Findings and Interpretations
| Clinical findings | Possible causes |
|---|---|
| Diplopia; dysconjugate gaze (i.e., both eyes cannot fix to an object at the same time); ptosis | Cranial nerves III, IV, or VI palsy; extraocular muscle paresis or paralysis |
| Eyelid laceration | Associated globe perforation; damage to the lacrimal drainage system or levator muscle |
| Intact contralateral direct reflex and contralateral consensual reflex; loss of ipsilateral consensual reflex and ipsilateral direct reflex | Oculomotor nerve (i.e., efferent nerve) dysfunction on one side |
| Intact contralateral direct reflex and ipsilateral consensual reflex; loss of contralateral consensual reflex and ipsilateral direct reflex | Optic nerve (i.e., afferent nerve) dysfunction on one side |
| Orbit and cheek deformity; tenderness over the supraorbital ridge or frontal bone | Step-off fracture of orbit |
| Pupillary reflex visual field testing (i.e., the confrontation visual field examination) abnormality | Defect in visual pathway |
| Sudden vision loss | Central retinal artery occlusion; retinal detachment; retinal hemorrhage |
| Unilateral vision loss with Marcus Gunn's pupillary phenomenon* | Defective pupillary response indicates optic nerve dysfunction or retinal detachment; unilateral loss of vision without Marcus Gunn's pupillary phenomenon indicates that the lesion is somewhere other than on the optic nerve or retina |
*—Marcus Gunn's pupillary phenomenon is determined by the swinging flashlight test, which is performed by illuminating the pupils alternately every two seconds to check for the dilation of the pupil on the contralateral side. Normally, both pupils constrict when one pupil is illuminated with a penlight, but with Marcus Gunn's pupillary phenomenon, there is an abnormally slight contraction or even dilation on the affected side.
Mechanical Injury to the Globe
Globe rupture involves compromised integrity of the cornea or sclera; it should always be considered in a patient with an eye injury. If not treated promptly and appropriately, the patient may develop endophthalmitis, which is a serious intraocular infection that can cause blindness. Indicators of globe rupture include moderate to severe pain, decreased vision, hyphema (blood in the anterior chamber), loss of anterior chamber depth, or deviation of the pupil toward the laceration (Table 2). Severe subconjunctival hemorrhage involving the area 360 degrees around the cornea is also highly suspicious for globe rupture.
Table 2 Diagnosis and Initial Outpatient Treatment of Ocular Emergencies
| Disorder | Signs and symptoms | Slit-lamp examination findings | Initial treatment |
|---|---|---|---|
| Mechanical injury to the globe | Normal or damaged cornea; moderate to severe pain; normal or decreased vision; hyphema; red eye/subconjunctival hemorrhage in the area 360 degrees around the cornea | Decreased anterior chamber depth relative to uninjured eye; irregular or deviated pupil toward the direction of injury; positive Seidel test | Eye shield |
| Tonometry contraindicated | |||
| Leave embedded foreign object in place | |||
| Scheduled analgesia and antiemetics; update tetanus immunization | |||
| Immediate referral to an ophthalmologist | |||
| Chemical injury | Cornea may have minor epithelial damage or be opaque; moderate to severe pain; blurred vision; reflex blepharospasm; photophobia; sensation of a foreign body; red eye/conjunctiva | Findings depend on the severity; corneal and scleral melting may occur if injury is severe | Eye irrigation with normal saline or lactated Ringer's solution until pH is normal |
| Continue eye wash on way to the emergency department or an ophthalmologist | |||
| Central retinal artery occlusion | Clear cornea; amaurosis fugax (transient, painless unilateral vision loss) or permanent vision loss; red eye/conjunctiva | Interrupted columns of blood within the retinal vessels (sometimes referred to as “boxcarring”) and attenuation of retinal arteries; cherry-red spot at the fovea; pale fundus; pupil may be dilated and react poorly to light | Lowering IOP with mannitol (Osmitrol) 0.25 to 2.0 g per kg IV once, acetazolamide (Diamox*) 500 mg IV or orally once, carbogen inhalation, administering oral nitrates, or by laying the patient on his or her back |
| Ocular-digital massage | |||
| Immediate referral to an ophthalmologist | |||
| Acute angle-closure glaucoma | Acute onset of severe pain; blurred vision; frontal headache; halos around lights; increased IOP; red eye/conjunctiva | Mid-dilated and sluggish pupil; normal or hazy cornea; shallow anterior chamber | Lowering IOP with acetazolamide 500 mg orally once; and one drop each of 0.5% timolol maleate (Timoptic), 1% apraclonidine (Iopidine), and 2% pilocarpine (Isopto Carpine) one minute apart and repeated three times at five-minute intervals |
| Immediate referral to an ophthalmologist | |||
| Retinal detachment | Normal to peripheral or central vision loss; absence of pain; increasing floaters; unilateral photopsia; metamorphopsia | Normal conjunctiva and cornea; normal pupil; pale, detached retina | Antitussives or antiemetics (if needed) |
| Referral to an ophthalmologist within 24 hours |
IOP = intraocular pressure; IV = intravenously.
*—Brand only available in the oral formulation.
If there is a possibility of globe penetration, the Seidel test should be performed. If a slit lamp is available, a moistened fluorescein dye strip can be gently applied directly to the site of the injury. Slit-lamp examination is performed with cobalt blue light. If a perforation or leak is present, the fluorescein dye will be diluted by the aqueous; it will appear as a dark (i.e., diluted) stream within a pool of bright green (i.e., concentrated) dye. This is known as the Seidel sign or a positive Seidel test.4,5 Any pressure on the eye must be avoided to prevent extrusion of intraocular tissue. The Seidel test should not be performed if there is obvious evidence of rupture or full-thickness laceration.
If globe rupture is suspected or confirmed, an eye shield (Figure 1) should be immediately placed over the affected eye and further direct examination should be deferred to avoid putting pressure on the eye. Computed tomography of the head and orbits (coronal and axial views) is recommended to evaluate for open globe injury, intraocular foreign body, or orbital wall fractures.6
Figure 1.
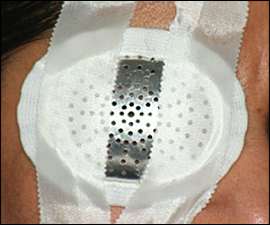
Eye shield to help protect the eye in case of globe injury.
Because the Valsalva maneuver or pressure on the globe may lead to extrusion of intraocular contents, appropriate analgesia and antiemetics should be provided on a scheduled basis. Tonometry should not be attempted if globe rupture is suspected. Tetanus prophylaxis should be given if immunization is not up to date. When possible, systemic antibiotics should be started within six hours of the injury.
The choice of antibiotic depends on patient age, cause of injury, and microorganism involved. Streptococcus species, Staphylococcus aureus, and Staphylococcus epidermidis are among the most common causes of endophthalmitis. Fluoroquinolones, aminoglycosides, and cephalosporins are typically used to treat these infections. An acceptable antibiotic regimen for an adult is intravenous cefazolin (Kefzol; brand not available in the United States) 1 g given every eight hours, plus intravenous ciprofloxacin (Cipro) 400 mg every 12 hours. Antibiotic treatment for children should include intravenous cefazolin 25 to 50 mg per kg daily (evenly divided and given every eight hours) plus intravenous gentamicin (Garamycin; brand not available in the United States) 2 mg per kg every eight hours.7
Globe injury from a cat or dog bite requires prophylactic antibiotic treatment for Pasteurella multocida and Eikenella corrodens infections, respectively. In case of injury from organic material (e.g., hay, leaves, grains), infection from gram-negative bacteria and fungi also should be treated with antibiotics.7 The patient should be given nothing by mouth, and an emergent referral to an ophthalmologist should be sought for further evaluation and surgical repair. If a protruding foreign body is present, it should not be removed if globe rupture is suspected.
Eye injury from high-velocity trauma (e.g., a flying object, motor vehicle collision) should be treated as a penetrating injury. Penetrating eye injury from a very small object at high speed may be detected as only a small corneal defect by fluorescein staining. If the injury is not treated appropriately and immediately, endophthalmitis and possible vision loss may occur within a few days. In this situation, given a history of high-velocity trauma, immediate referral to an ophthalmologist is recommended irrespective of the physical findings during examination.8
If conjunctival laceration is present, there should be a high index of suspicion for globe rupture or laceration, and the patient should be referred to an ophthalmologist. Examination with a slit lamp and fluorescein dye helps to differentiate between a conjunctival laceration and a globe rupture. Extremely gentle manipulation of the conjunctiva with a moist cotton swab under slit lamp may distinguish a superficial conjunctival laceration from involvement of the underlying sclera. A primary care physician can manage conjunctival lacerations smaller than 1 cm by using a topical antibiotic and close follow-up. Lacerations larger than 1 cm should be referred to an ophthalmologist for possible suture placement.
Chemical Injuries
A chemical injury to the eye is a true ocular emergency. Coming into contact with a gas or liquid is a common way by which an eye is exposed to a chemical. Eye injury from exposure to alkali is more detrimental and more common than injury from an acid. Severity of the eye injury depends on the pH concentration and the nature of the chemical. After chemical exposure, patients often complain of moderate to severe pain, photophobia, blurred vision, and sensation of a foreign body. Reflex blepharospasm and red eye are common signs of chemical injury (Table 2). If the affected eye looks white after exposure to an alkali, it could be an indication of a particularly severe eye injury with ischemia of the conjunctival and scleral vessels.
Chemical injury is the only eye emergency in which treatment should not be delayed to evaluate visual acuity. Topical anesthetics (e.g., tetracaine [Pontocaine]) help to relieve pain and thus facilitate eye irrigation. Eye irrigation with normal saline or lactated Ringer's solution should be initiated as soon as injury from a chemical is suspected. Non-sterile water can be used if no other options are available.9,10
In an office setting, the eye can be irrigated using an intravenous tube, nasal cannula, or Morgan Medi-flow lens. After about 30 minutes of irrigation, the eye should be closed for approximately five minutes. Then the pH of the injured eye should be tested with litmus paper that is touched to the conjunctival fornix (the area between the eyelid and globe) inside the lower eyelid. Irrigation should be continued until a neutral pH level (7.0) is achieved and maintained for at least 30 minutes. Once pH is stabilized, a cycloplegic agent (0.25% scopolamine [Isopto Hyoscine] eye drops) and a broad-spectrum antibiotic (ciprofloxacin, ofloxacin [Ocuflox], gentamicin, or tobramycin [Tobrex]) should be applied.
Duration of irrigation has been shown to have a direct positive impact on the outcome of treatment and hospital stay.11 The volume of irrigation necessary to reach neutral pH depends on the type of chemical and the duration of exposure. Up to 10 liters of an irrigant may be necessary to achieve neutral pH.12 If pH paper is not available, the eye should be irrigated with at least 1 to 2 liters of eyewashing solution or for at least 30 minutes.
In a retrospective study, 36 patients irrigated 49 injured eyes with tap water before seeking medical help, whereas 17 patients with 29 injured eyes did not irrigate after the injury. Of the patients who irrigated their eyes, 76 percent had a minimal grade 1 injury, whereas 86 percent of patients who did not irrigate their eyes had a more severe grade 2 injury. Mean healing time was eight days for the patients who irrigated and 29 days for those who did not.13
Acid should not be used to neutralize a base, and vice versa.14 Sweeping the conjunctival fornices with a moist cotton swab after instilling topical anesthetic may help to neutralize the pH by removing crystallized particles of the chemical. Once the pH is within normal range (7.0 to 7.3), patients should be referred immediately to an ophthalmologist without ceasing the irrigation.10 When possible, patients should be instructed to bring the container of the chemical that caused their eye injury.
Central Retinal Artery Occlusion
The central retinal artery is a branch of the ophthalmic artery, which supplies blood mainly to the retina via retinal branches. Occlusion of the central retinal artery quickly leads to ocular stroke, which can cause severe vision loss. Partial loss of the visual field may occur if only distal branches of the retinal arteries are occluded.
Patients with CRAO describe painless and sudden loss of vision in one eye. The patient may report a history of amaurosis fugax (transient, painless unilateral vision loss) before presentation. The pupil may be dilated with sluggish reaction to light (Table 2). If vision loss is acute, transient, and bilateral, other causes such as migraine auras, heart failure, and hypertensive emergencies should be suspected. Acute vision loss secondary to retinal ischemia can also be caused by severe unilateral or bilateral carotid artery stenosis. Funduscopic examination after CRAO reveals interrupted columns of blood within the retinal vessels (sometimes referred to as “boxcarring”) and retinal pallor secondary to retinal edema, with a characteristic cherry-red spot at the fovea and attenuation of the retinal arteries. An embolus in a retinal vessel may be seen (Figure 215).
Figure 2.
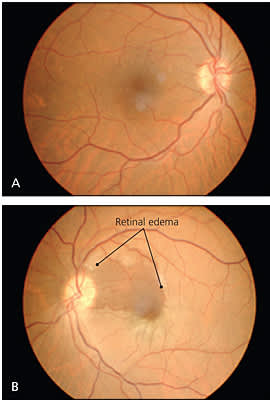
Risk factors for CRAO include age older than 70, atherosclerosis, diabetes, endarteritis, glaucoma, high cholesterol levels, hypertension, hypercoagulable states, and migraine.15–17 CRAO may occur in younger patients with systemic disease, such as collagen vascular diseases, hypercoagulopathies, or cardiac valvular disease. Although rare, CRAO also can occur in association with syphilis or sickle cell disease. CRAO is associated with giant cell arteritis in 5 to 10 percent of cases; an appropriate review of systems and measurement of erythrocyte sedimentation rate or C-reactive protein should be performed to rule it out.18
Patients with symptoms and signs of CRAO must be referred immediately to an ophthalmologist because irreversible damage occurs in as little as 100 minutes of occlusion.19 In the acute presentation of CRAO, some interventions to restore blood flow to the retina can be attempted. Several can be initiated in a primary care setting before the patient is seen by an ophthalmologist. These include ocular-digital massage or lowering of intraocular pressure with intravenous mannitol (Osmitrol) 0.25 to 2.0 g per kg once, oral or intravenous acetazolamide (Diamox; brand only available in oral formulation) 500 mg once, carbogen inhalation (95 percent oxygen and 5 percent carbon dioxide), administering oral nitrates, or laying the patient flat on his or her back. The theory behind ocular-digital massage is that the steep increase in pressure followed by a sudden return to normal pressure may plunge an embolus farther along the vasculature into a distal branch, minimizing the area of ischemia and vision loss. A systematic review of several randomized controlled trials concluded that there is currently not enough evidence to recommend one treatment over another for acute CRAO.20
After acute management of CRAO, carotid ultrasonography to evaluate for stenosis or plaque formation may identify the cause of CRAO. In patients with negative results on carotid ultrasonography, transesophageal echocardiography should be considered to exclude a cardiac origin of the embolus.15,21 Minimization of risk factors with diet and lifestyle modifications and treatment of hypercholesterolemia, hypertension, and diabetes may help prevent CRAO.
Acute Angle-Closure Glaucoma
If acute angle-closure glaucoma is not treated immediately, damage to the optic nerve and significant and permanent vision loss can occur within hours. Patients with acute angle-closure glaucoma often present with blurred vision, eye redness, frontal headache, colored halos around lights, severe eye pain, discomfort, nausea, and vomiting (Table 2). Physical findings include increased intraocular pressure greater than 30 mm Hg (normal range: 10 to 23 mm Hg), a mid-dilated pupil (4 to 6 mm), sluggish reaction of the pupil to direct illumination, a shallow anterior chamber, a hazy cornea, and hyperemic conjunctiva. Risk factors for acute angle-closure glaucoma include anterior placement of the lens, hyperopia, myopia, narrow angle, and shallow anterior chamber.
An attack of acute angle-closure glaucoma in predisposed persons can occur as a result of dim lighting or use of certain medications (e.g., dilating drops, anticholinergics, antidepressants). Medications such as sulfa derivatives and topiramate (Topamax) can cause swelling of the ciliary body and secondary angle closure.22 If a tonometer is available, intraocular pressure should be measured to see if it has increased.
During an acute attack, medical therapy is initiated to lower the increased intra-ocular pressure. If there is high suspicion of acute angle-closure glaucoma, one drop each of 0.5% timolol maleate (Timoptic), 1% apraclonidine (Iopidine), and 2% pilocarpine (Isopto Carpine), one minute apart, and a 500-mg tablet of acetazolamide may be given in the office; these should help decrease the intraocular pressure before the patient is seen by an ophthalmologist. The eyedrops should be repeated three times at five-minute intervals.12 If possible, intraocular pressure should be measured every hour and treatment should be continued until the patient can be seen by an ophthalmologist.
Therapy is initiated to lower the intraocular pressure, reduce pain, and clear corneal edema in preparation for iridotomy. Definitive treatment for primary acute angle-closure glaucoma is laser iridotomy. Surgical iridectomy can be performed if a laser iridotomy is not successfully performed.23,24
Retinal Detachment
Retinal detachment is the separation of the neurosensory layer of the retina from the choroid and retinal pigment epithelium underneath. Although retinal detachment is uncommon (affecting one in 10,000 persons per year), it leads quickly to degeneration of photoreceptors because of ischemia. Permanent vision loss can be prevented by early diagnosis and treatment.25 Myopia (nearsightedness) is a risk factor for retinal detachment; it accounts for about 55 percent of nontraumatic retinal detachments.26 Other risk factors for retinal detachment include cataract surgery, diabetic retinopathy, family history of retinal detachment, older age, and trauma.27
Patients with retinal detachment often complain of unilateral photopsia (i.e., sensation of flashing light), an increasing number of floaters in the affected eye signifying posterior vitreous detachment, decreased visual acuity, and metamorphopsia (i.e., wavy distortion of an object). Floaters may move in and out of central vision. Vision loss may be curtain-like, filmy, or cloudy. If the macula or the central vision is involved, the patient may lose the ability to read, have loss of light perception, or may not be able to see a hand waved in front of his or her face (Table 2).
Direct ophthalmoscopic examination aids in the diagnosis of retinal detachment (Figure 3). Asymptomatic retinal breaks and lattice degeneration (i.e., focal thinning in the periphery of the retina) are risk factors for retinal detachment. If funduscopic examination reveals either of these risk factors, laser demarcation of these areas may be considered to prevent future retinal detachment and vision loss. Of note, ophthalmologic consultation is necessary with this procedure, and there is insufficient evidence to prove its effectiveness.28 If the retina cannot be evaluated, and if retinal detachment is suspected from patient history alone, immediate referral to an ophthalmologist is warranted, especially for persons with known risk factors.
Figure 3.
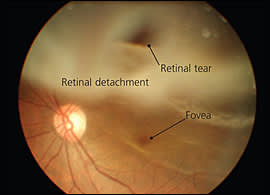
Extensive superior retinal detachment that extends to just below the fovea. Retinal tear is visible superiorly.

