ACOG Publishes Guidelines on Hemoglobinopathies in Pregnancy
Guideline source: American College of Obstetrics and Gynecology
Literature search described? Yes
Evidence rating system used? Yes
Published source: Obstetrics & Gynecology, January 2007
The American College of Obstetricians and Gynecologists (ACOG) estimates that more than 270 million persons worldwide are heterozygous carriers of hereditary disorders of hemoglobin (Hb), and at least 300,000 affected homozygotes or compound heterozygotes are born every year. To address this, ACOG has released recommendations for screening and clinical management of hemoglobinopathies during pregnancy.
Genetic screening can help physicians identify couples at risk of having children with hemoglobinopathies and counsel them about reproduction and prenatal diagnosis. DNA analysis of cultured amniocytes or chorionic villi is preferred for prenatal diagnosis of hemoglobinopathies. Persons of northern European, Japanese, Native American, Inuit, or Korean descent are considered to be at low risk of hemoglobinopathies. Those of southeast Asian, African, or Mediterranean descent are at higher risk of being carriers of hemoglobinopathies, and ACOG recommends that physicians offer these patients carrier screening. However, ethnicity is not always a good predictor of risk because patients from high-risk groups may marry outside of their ethnic group.
A complete blood count should be obtained to ensure that Hb is accurately identified for patients of non-African descent. For patients of African descent, ACOG recommends performing Hb electrophoresis in addition to obtaining a complete blood count (Figure 1). Solubility testing for Hb S (Sickledex), high-performance liquid chromatography, and isoelectric focusing have been used for primary screening; however, solubility tests alone often fail to identify transmissible Hb gene abnormalities that could affect fetal outcomes. Additionally, many persons with these genotypes (e.g., Hb C, beta thalassemia, Hb E, Hb B, Hb D) are asymptomatic. If their partners have the sickle cell trait or another hemoglobinopathy, they could have offspring with more serious hemoglobinopathies. Therefore, solubility testing could be valuable for rapid screening for sickling when the information is crucial to immediate patient care.
Figure 1. Evaluation for Hematologic Assessment
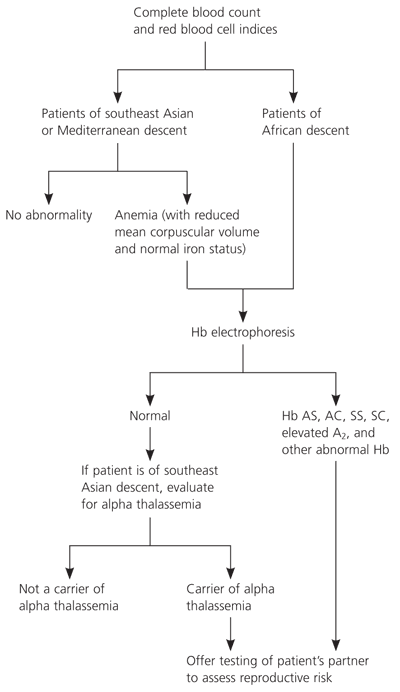
Specialized antepartum evaluation for hematologic assessment of patients of African, southeast Asian, or Mediterranean descent. Patients of southeast Asian or Mediterranean descent should undergo electrophoresis if their blood test results reveal anemia. (Hb = hemoglobin.)
Adapted with permission from the American College of Obstetricians and Gynecologists. Hemoglobinopathies in pregnancy. ACOG Practice Bulletin No. 78, January 2007. Obstet Gynecol 2007;109:232.
Women with sickle cell disease who are pregnant are at increased risk of morbidity and mortality because of the combination of underlying hemolytic anemia and multiorgan dysfunction associated with this disorder. These patients need increased prenatal folic acid supplementation. However, the standard of 1 mg of folate in prenatal vitamins is not adequate for patients with hemoglobinopathies; instead, 4 mg per day of folic acid is recommended.
Women with sickle cell disease who are pregnant also are at increased risk of spontaneous abortion, stillbirth, and preterm labor. Therefore, ACOG recommends that serial ultrasonography and antepartum fetal testing be performed. Most of these patients require narcotics for pain control, so physicians should interpret the results of abnormal antepartum testing with caution. Cesarean delivery is not recommended in women with sickle cell disease, and it should be performed only for obstetric indications. Epidural analgesia is usually well tolerated as long as hypotension and hypoxemia are avoided. Ideally, however, pregnant women with sickle cell disease should receive care at institutions that can manage complications of the disease and high-risk pregnancies.

