A 49-year-old white man who had type 1 diabetes that led to stage 5 chronic kidney disease presented with painful, erythematous, indurated plaques distributed bilaterally on the lower extremities (Figures 1 and 2). One year earlier, the patient underwent a kidney transplant and had postoperative recurrence of advanced renal failure caused by obstructive uropathy. He required multiple imaging studies and interventional procedures, including gadolinium–enhanced magnetic resonance imaging (MRI) of the abdomen and pelvis approximately one month before the plaques developed. He had a history of hypertension, coronary artery disease, peripheral arterial disease, and hypercoagulability caused by the factor II gene mutation.
Figure 1.
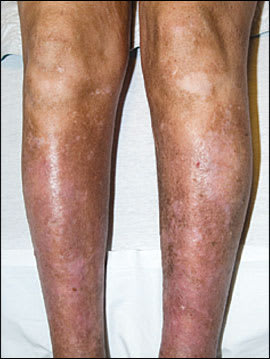
Figure 2.
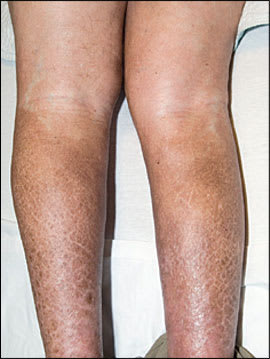
Question
Based on the patient's history and physical examination, which one of the following is the most likely diagnosis?
A. Eosinophilic fasciitis.
B. Lipodermatosclerosis.
C. Nephrogenic systemic fibrosis.
D. Scleromyxedema.
E. Systemic sclerosis (scleroderma).
Discussion
The answer is C: nephrogenic systemic fibrosis. Nephrogenic systemic fibrosis is a recently described, rare condition that involves systemic fibrosis and that usually affects the skin, although it may involve other systems. Initially, it was thought to occur only in patients receiving hemodialysis; however, it has since been reported in patients receiving peritoneal dialysis and in patients with acute renal failure, chronic kidney disease, and renal transplants.1 The condition affects all age groups.2 Underlying kidney dysfunction is a requisite for the disease; however, there is no known correlation between nephrotic systemic fibrosis and etiology of kidney disease, patient sex, or ethnicity.
The clinical manifestations of nephrogenic systemic fibrosis can be difficult to detect. The disease begins as subacute edema of the distal extremities, which may be misdiagnosed as simple peripheral edema or cellulitis. Progression leads to symmetric, indurated, brawny or erythematous, cutaneous papules and plaques involving the limbs and trunk with associated sharp or burning pain, pruritus, and muscle weakness.3 Fibrotic changes may progress rapidly, causing joint contractures and subsequent immobility.2 Involvement of other organs, including the lungs, skeletal muscle, heart, diaphragm, testes, and esophagus, has been reported.2,4 The chronic, progressive disease can be fatal; and although it sometimes stabilizes, it rarely resolves spontaneously.5
Nephrogenic systemic fibrosis may resemble systemic sclerosis (scleroderma), scleromyxedema, eosinophilic fasciitis, lipodermatosclerosis, or eosinophiliamyalgia syndrome.6,7 Confirming nephrogenic systemic fibrosis requires a histologic examination of a skin biopsy, which shows dermal mucin, increased immature collagen, prominent collagen bundles, and increased dermal spindle cells.
The exact pathogenesis for nephrogenic systemic fibrosis is uncertain, but gadolinium–based contrast media is most directly linked.1,2 Gadolinium is excreted by the kidneys, and reduced renal function significantly prolongs its half-life.8 The association between gadolinium and nephrogenic systemic fibrosis was first described in a small cluster of patients in Austria and was later substantiated in other cases.1,9 In most patients, exposure to gadolinium occurs days to months before the onset of the disease. The presence of gadolinium in biopsy specimens from some patients with nephrogenic systemic fibrosis supports a possible causative relationship.10,11 There is no definitive treatment for the disease; therefore, physicians must address sequelae, such as pain and fibrotic contractures.
The U.S. Food and Drug Administration (FDA) has issued a black box warning for all gadolinium–based contrast media.9 The FDA recommends screening patients for renal dysfunction before using gadolinium and using alternative imaging methods in patients with acute renal failure, advanced chronic kidney disease (i.e., glomerular filtration rate of less than 30 mL per minute per 1.73 m2), or acute renal insufficiency of any severity that is caused by hepatorenal syndrome or that occurs in the postoperative period after a liver transplant. However, contrast-enhanced MRI is still an option if it provides essential clinical information that is not provided by non–contrast-enhanced MRI or another imaging modality. When the risk-benefit assessment warrants the use of contrast-enhanced MRI, the lowest possible dose of gadolinium should be used.9
Although it is unknown whether dialysis prevents nephrogenic systemic fibrosis, prompt dialysis in patients receiving hemodialysis is recommended immediately following the administration of gadolinium.9 In patients receiving peritoneal dialysis or in those not receiving dialysis who have stage 4 or 5 chronic kidney disease, administration of short-term hemodialysis may be considered.12,13
Selected Differential Diagnosis of Progressive Skin Fibrosis
| Disease | Description | Physical findings | Histopathology |
|---|---|---|---|
| Eosinophilic fasciitis | Fasciitis, hypergammaglobulinemia, eosinophilia | Woody infiltration of arms, legs, feet, and hands (sparing the digits); contractures | Fibrosis, plasma cells, eosinophils, lymphocytes |
| Lipodermatosclerosis | Subcutaneous fibrosis caused by chronic edema, usually venous stasis | Indurated, inflamed plaques on lower legs | Stasis dermatitis, ischemic fat necrosis, septal fibrosis, obliteration of the fat lobules |
| Nephrogenic systemic fibrosis | Progressive systemic fibrosis in patients with renal impairment | Indurated plaques; hyperpigmentation; involves legs, feet, arms, hands, fingers; sclerodactyly | Dermal spindle cell proliferation (CD34) with variable mucin deposits; dystrophic calcifications |
| Scleromyxedema | Papular skin eruption, mucin deposits, associated monoclonal gammopathy | Papules, plaques, and nodules involving the face, neck, ears, and fingers | Dermal fibrosis, increased mucin deposition, lymphoplasma cell infiltration |
| Systemic sclerosis (scleroderma) | Autoimmune connective tissue disease with skin fibrosis and vasculopathy | Thick, tight skin with hyperpigmentation, telangiectasis involving the face, neck, arms, legs, feet, and hands; sclerodactyly with loss of digital pulp | Homogenized, thickened collagen; mucin deposition is not increased; variable inflammatory infiltration |
Information from Boin F, Hummers LK. Scleroderma-like fibrosing disorders. Rheum Dis Clin North Am. 2008;34(1):199–220.

