The American College of Cardiology and American Heart Association, in collaboration with the Canadian Cardiovascular Society, have issued an update of the 2004 guideline for the management of patients with ST-segment elevation myocardial infarction. The American Academy of Family Physicians endorses and accepts this guideline as its policy. Early recognition and prompt initiation of reperfusion therapy remains the cornerstone of management of ST-segment elevation myocardial infarction. Aspirin should be given to symptomatic patients. Beta blockers should be used cautiously in the acute setting because they may increase the risk of cardiogenic shock and death. The combination of clopidogrel and aspirin is indicated in patients who have had ST-segment elevation myocardial infarction. A stepped care approach to analgesia for musculoskeletal pain in patients with coronary heart disease is provided. Cyclooxygenase inhibitors and nonsteroidal anti-inflammatory drugs increase mortality risk and should be avoided. Primary prevention is important to reduce the burden of heart disease. Secondary prevention interventions are critically important to prevent recurrent events in patients who survive.
The American College of Cardiology (ACC) and American Heart Association (AHA) have updated their 2004 guideline1 for the management of patients with ST-segment elevation myocardial infarction (STEMI). The updated guideline was developed in collaboration with the Canadian Cardiovascular Society and published in 2008.2 The American Academy of Family Physicians endorses and accepts this guideline as its policy. This review summarizes the key recommendations as they apply to the prevention of coronary heart disease (CHD), acute management of STEMI, and secondary prevention of recurrent events. As always, the values and preferences of patients, and sound clinical judgment are of paramount importance in applying guideline recommendations.
SORT: KEY RECOMMENDATIONS FOR PRACTICE
| Clinical recommendation | ACC/AHA LOE* | SORT rating |
|---|---|---|
| Patients with STEMI who present within 12 hours of symptom onset and who do not have contraindications should receive immediate reperfusion therapy with fibrinolysis or PCI. | Class I, LOE A | A |
| Patients with STEMI should immediately chew 162 to 325 mg of aspirin on recognition of symptoms, unless there is an absolute contraindication. | Class I, LOE A | A |
| Intravenous beta blockers should not be given to patients with STEMI. They may be considered for treatment of hypertension if there are no contraindications (see Table 1). | Class III, LOE A | A |
| Oral clopidogrel (Plavix) at a dosage of 75 mg daily should be added to aspirin therapy in patients with STEMI, whether or not they undergo reperfusion therapy. | Class I, LOE A | A |
| Oral beta blocker therapy should be initiated within 24 hours of STEMI in patients with no contraindications. | Class I, LOE B | B |
| Patients undergoing reperfusion with PCI or stenting should begin clopidogrel therapy. Duration of therapy varies, depending on the type of stent used (no stent, 14 days; bare-metal stent, at least one month but ideally one year unless patient is at increased risk of bleeding; drug-eluting stent, one year). | Class I, LOE B | B |
| Patients who routinely took nonsteroidal anti-inflammatory drugs (except for aspirin) before STEMI should discontinue these agents because of increased risks of mortality, reinfarction, hypertension, heart failure, and myocardial rupture. | Class I, LOE C | C |
ACC = American College of Cardiology; AHA = American Heart Association; LOE = level of evidence; PCI = percutaneous coronary intervention; STEMI = ST-segment elevation myocardial infarction.
*— See Table 3 for explanations of ACC/AHA levels of evidence.
SORT ratings: A = consistent, good-quality patient-oriented evidence; B = inconsistent or limited-quality patient-oriented evidence; C = consensus, disease-oriented evidence, usual practice, expert opinion, or case series. For information about the SORT evidence rating system, go to https://www.aafp.org/afpsort.xml.
Primary Prevention
The ACC/AHA guideline stresses the importance of primary prevention. The consensus opinion is that a risk factor assessment for CHD should be performed every three to five years in all patients. Risk factors include smoking, diabetes, hypercholesterolemia, hypertension, family history, age, and sex. In patients with two or more major risk factors, calculation of a 10-year coronary artery disease risk score is recommended to assess the need for primary prevention.3 The risk score can also be an effective tool to start a dialogue with patients about lifestyle change.
Prehospital Care of Patients with Suspected STEMI
Time is of the essence in the acute management of STEMI. It is important that family physicians educate patients about the symptoms of myocardial infarction (MI) and instruct them to seek help immediately if these symptoms occur. Patients who have been prescribed nitroglycerin should take one dose sublingually at the onset of symptoms; if they do not improve or if they worsen within five minutes, they should seek medical attention. Once symptoms are recognized, patients should chew aspirin (162 to 325 mg) unless there is an absolute contraindication. At the community level, clear protocols must exist to minimize the time to transport the patient to the appropriate facility. Emergency medical service (EMS) providers should be trained and equipped to provide early defibrillation. In the prehospital setting, advanced cardiac life support-trained health care professionals should obtain 12-lead electrocardiography. Pre-hospital fibrinolysis protocols are reasonable if the EMS system is appropriately trained and staffed.
Acute Management
STEMI is defined as ST-segment elevation of greater than 0.1 mV in at least two contiguous precordial or adjacent limb leads, a new left bundle branch block, or a true posterior MI. The most important point in managing STEMI is minimizing the time from the onset of symptoms until the initiation of reperfusion therapy (fibrinolysis or percutaneous coronary intervention [PCI]). The goal is for fibrinolysis to begin less than 30 minutes from the time of the patient's first contact with the health care system, or for balloon inflation for PCI to begin in less than 90 minutes.
Figure 1 summarizes the recommended approach to a patient with STEMI, and it reviews conditions in which PCI or fibrinolysis is preferred or contraindicated.1 Patients with STEMI who present within 12 hours of symptom onset and do not have contraindications should receive reperfusion therapy. Patients with MI with ST-segment depression should not be treated with fibrinolysis unless they have a true posterior MI. Primary PCI is preferred to fibrinolysis only in high–PCI-volume hospitals (i.e., those with more than 36 procedures per year). If patients cannot reach such a facility within the required 90 minutes, they should receive fibrinolysis unless contraindicated.
Figure 1. Approach to the Patient with Suspected STEMI
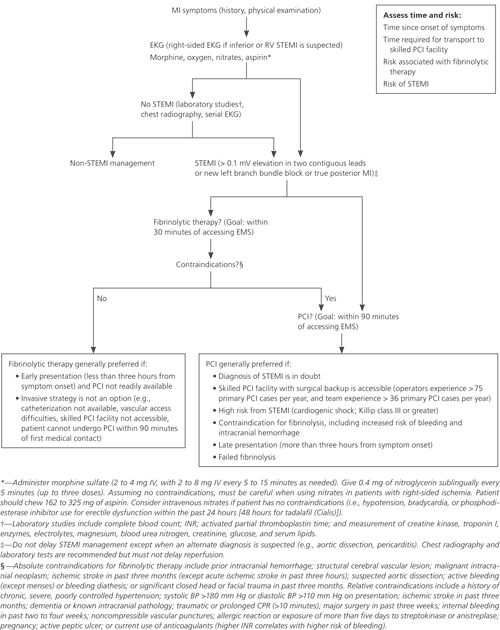
Treatment algorithm for patients with symptoms of MI. (BP = blood pressure; CPR = cardiopulmonary resuscitation; EKG = electrocardiography; EMS = emergency medical services; INR = International Normalized Ratio; IV = intravenously; MI = myocardial infarction; PCI = percutaneous coronary intervention; RV = right ventricular; STEMI = ST-segment elevation myocardial infarction.)
Adapted from Antman EM, Anbe DT, Armstrong PW, et al. ACC/AHA guidelines for the management of patients with ST-elevation myocardial infarction: a report of the American College of Cardiology/American Heart Association Task Force on Practice Guidelines (Committee to Revise the 1999 Guidelines for the Management of Patients with Acute Myocardial Infarction) [published corrections appear in Circulation. 2005;111(15):2013–2014, and Circulation. 2007;115(15):e411]. Circulation. 2004;110(9):e116–e118, e124, e127.
If patients with STEMI have cardiogenic shock, they should be transported to a facility capable of cardiac catheterization and rapid revascularization (PCI and coronary artery bypass graft surgery). Patients with contraindications to fibrinolysis should be immediately transported to such a facility, or transferred within 30 minutes. It is also reasonable to consider an invasive strategy in patients with severe congestive heart failure (CHF).
The strategy for facilitated PCI that was proposed in the 2004 guideline, in which higher-risk patients with low bleeding risk receive full-dose fibrinolysis and subsequent PCI, may be harmful and is no longer recommended. More study is required before clear recommendations can be made on other facilitated strategies (i.e., half-dose fibrinolysis, a glycoprotein IIb/IIIa inhibitor, or both). These strategies may be considered when patients are at high risk and PCI is not available within 90 minutes, provided bleeding risk is low (i.e., in younger patients and those with normal body weight, and in the absence of poorly controlled hypertension). Rescue PCI after failed thrombolysis is still appropriate and should be performed in patients who fail fibrinolysis, as evidenced by shock, severe CHF or pulmonary edema (Killip class III or greater), or hemodynamically compromising ventricular arrhythmias. Rescue PCI is reasonable in patients who have less than 50 percent resolution of ST-segment elevation 90 minutes after initiation of fibrinolytic therapy and a moderately large area of myocardium at risk.
The routine use of intravenous beta blocker therapy in the acute phase of STEMI is not recommended because of the increased risk of cardiogenic shock, based on findings from the COMMIT/CCS-2 study (Clopidogrel and Metoprolol in Myocardial Infarction Trial/Second Chinese Cardiac Study).4 However, it is reasonable to use intravenous beta blocker therapy in the acute setting to manage hypertension in patients with STEMI who have none of the contraindications listed in Table 1.5 Daily oral beta blocker therapy should be initiated within 24 hours to hemodynamically stable patients who have no contraindications; these agents are also important in secondary prevention.
Table 1 Contraindications to Beta Blocker Therapy in Patients with STEMI
| Evidence of low output state | |
| Increased risk of cardiogenic shock | |
| Age > 70 years | |
| Sinus tachycardia > 110 beats per minute or heart rate < 60 beats per minute | |
| Systolic blood pressure < 120 mm Hg | |
| Increased time since onset of STEMI | |
| Signs of heart failure | |
| Relative contraindication | |
| Active asthma or reactive airway disease | |
| PR interval > 0.24 seconds | |
| Second- or third-degree heart block | |
STEMI = ST-segment elevation myocardial infarction.
Information from reference 5.
Concerns have been raised about the safety of non-steroidal anti-inflammatory drugs (NSAIDs) in patients with acute or previous STEMI. The update contains new recommendations that do not pertain to aspirin, which has a clear benefit in these patients. Patients who routinely took NSAIDs (nonselective and cyclooxygenase-2 [COX-2] selective agents) before STEMI should discontinue those agents because of the increased risk of mortality, reinfarction, hypertension, heart failure, and myocardial rupture associated with their use. These drugs should not be administered during hospitalization for STEMI.
Changes in Post-STEMI Management
Patients who receive pharmacologic reperfusion therapy should receive subsequent anticoagulation therapy for a minimum of 48 hours. There is no evidence of benefit for unfractionated heparin beyond 48 hours unless there are ongoing indications for anticoagulation. Low-molecular-weight heparin may be used instead for the duration of the hospitalization, up to eight days, if the patient has no significant renal dysfunction. The update lists effective anticoagulation regimens.1
The 2004 guideline contained no specific recommendation for dual antiplatelet therapy with clopidogrel (Plavix) plus low-dose aspirin in patients at high risk of atherothrombotic events. The update recommends that 75 mg of oral clopidogrel be added to daily aspirin, whether or not the patient underwent reperfusion; this change is summarized in Table 2.1 Treatment with clopidogrel should continue for at least 14 days. Long-term maintenance therapy (e.g., one year) may be useful in these patients. It is reasonable to start clopidogrel therapy with a 300-mg oral loading dose in patients younger than 75 years; no data are available for older patients. In patients with baremetal stents, clopidogrel should be continued for at least one month; it should be continued for several months in patients with drug-eluting stents (at least three months for sirolimus [Rapamune], six months for paclitaxel [Taxol]) and 12 months in patients who are not at high risk of bleeding. The guideline update does not address whether longer-term clopidogrel therapy is needed in patients with drug-eluting stents.
Table 2 Secondary Prevention for Patients with STEMI
| Intervention | Recommendation and ACC/AHA level of evidence* | |
|---|---|---|
| Smoking cessation | Ask about tobacco use at every visit I(B) | |
| Advise every patient who uses tobacco to quit I(B) | ||
| Assess the patient's willingness to quit I(B) | ||
| Assist by counseling and developing a plan for quitting I(B) | ||
| Arrange follow-up, referral to special programs, or pharmacotherapy (including nicotine replacement therapy) I(B) | ||
| Urge avoidance of exposure to environmental tobacco smoke at work and at home I(B) | ||
| Blood pressure control (less than 140/90 mm Hg, or less than 130/80 mm Hg in patients with diabetes or chronic kidney disease) | Initiate or maintain lifestyle modification in all patients (weight control; increased physical activity; alcohol moderation; sodium reduction; increased consumption of fresh fruits, vegetables, and low-fat dairy products) I(B) | |
| If blood pressure is 140/90 mm Hg or greater (or 130/80 mm Hg or greater in patients with diabetes or chronic kidney disease), start beta blockers and/or ACE inhibitors, then add thiazides or other agents as needed I(A) | ||
| Lipid management (LDL-C level substantially less than 100 mg per dL [2.60 mmol per L]; non–HDL-C level† less than 130 mg per dL [3.35 mmol per L] in patients with triglyceride levels 200 mg per dL [2.26 mmol per L] or greater) | Start dietary therapy in all patients. Reduce intake of trans-fatty acids, saturated fat (to less than 7 percent of total calories), and cholesterol (to less than 200 mg per day) I(B) | |
| Reasonable to add plant stanols or sterols (2 g per day) and viscous fiber (more than 10 g per day) to lower LDL-C level IIa(A) | ||
| Promote daily physical activity and weight management I(B) | ||
| Reasonable to encourage increased consumption of omega-3 fatty acids in the form of fish‡ or in capsule form (1 g per day) for risk reduction. For treatment of elevated triglyceride levels, higher dosages are usually necessary for risk reduction IIb(B) | ||
| Assess fasting lipid levels in all patients, and within 24 hours of hospitalization for STEMI. For hospitalized patients, initiate lipid-lowering therapy before discharge according to the following schedule: I(A) | ||
| • LDL-C level should be substantially less than 100 mg per dL I(A) | ||
| • Further reduction of LDL-C level to less than 70 mg per dL (1.80 mmol per L) is reasonable IIa(A) | ||
| • If baseline LDL-C level is 100 mg per dL or greater, initiate LDL-lowering drug therapy§I(A) | ||
| • If therapy lowers LDL-C level to 100 mg per dL or greater, intensify therapy (may require LDL-C–lowering drug combination)‖I(A) | ||
| • If baseline LDL-C level is 70 to 100 mg per dL, it is reasonable to treat to less than 70 mg per dL IIa(B) | ||
| • If triglyceride level is 150 mg per dL (1.70 mmol per L) or greater, or if HDL-C level is less than 40 mg per dL (1.05 mmol per L), weight management, physical activity, and smoking cessation should be emphasized I(B) | ||
| • If triglyceride level is 200 to 499 mg per dL (2.26 to 5.64 mmol per L), non–HDL-C level should be less than 130 mg per dL IIa(B) | ||
| • Further reduction of non–HDL-C level to less than 100 mg per dL is reasonable IIa(B) | ||
| Therapeutic options to reduce non–HDL-C level include more intense LDL-C–lowering therapy I(B), niacin therapy¶ (after LDL-C–lowering therapy) IIa(B), or fibrate therapy¶ (after LDL-C–lowering therapy) IIa(B) | ||
| If triglyceride level is 500 mg per dL (5.66 mmol per L),** therapeutic options to prevent pancreatitis include fibrate¶ or niacin¶ before LDL-C–lowering therapy; treat LDL-C to goal after triglyceride-lowering therapy. Achieve non–HDL-C level of less than 130 mg per dL if possible I(C) | ||
| Physical activity (30 minutes at least five days per week) | Assess risk with a physical activity history or an exercise test to guide prescription I(B) | |
| Encourage 30 to 60 minutes of moderate-intensity aerobic activity on most (and preferably all) days of the week, supplemented by an increase in daily lifestyle activities (e.g., walking breaks at work, gardening, household work) I(B) | ||
| Reasonable to encourage resistance training two days per week IIb(C) | ||
| Advise medically supervised programs for high-risk patients (e.g., those with recent acute coronary syndromes or revascularization) I(B) | ||
| Weight management (BMI 18.5 to 24.9 kg per m2; waist circumference less than 40 inches in men, less than 35 inches in women) | Assess BMI and waist circumference at each visit and encourage weight maintenance or reduction through an appropriate balance of physical activity, caloric intake, and behavioral programs when indicated I(B) | |
| If waist circumference (measured horizontally at the iliac crest) is 35 inches or more in women and 40 inches or more in men, initiate lifestyle changes and consider treatment strategies for metabolic syndrome as indicated I(B) | ||
| The initial goal of weight loss therapy should be to reduce body weight by approximately 10 percent from baseline. Further weight loss can be attempted if indicated I(B) | ||
| Diabetes management (A1C less than 7 percent) | Initiate lifestyle modification and pharmacotherapy to achieve near-normal A1C level I(B) | |
| Begin vigorous modification of other risk factors (e.g., physical activity, weight management, blood pressure control, lipid management) I(B) | ||
| Antiplatelet and anticoagulant therapy | Aspirin | |
| Start aspirin at a dosage of 75 to 162 mg daily and continue indefinitely in all patients unless contraindicated I(A) | ||
| Increase dosage to 162 to 325 mg daily in patients with bare metal stent (one-month course) and in those with drug-eluting stents (three-month course for sirolimus [Rapamune]; six-month course for paclitaxel [Taxol]). After high-dose course, continue indefinitely at a dosage of 75 to 162 mg per day I(B) | ||
| If bleeding is a concern, a lower dosage after stenting is reasonable IIa(C) | ||
| Clopidogrel (Plavix) | ||
| Start and continue clopidogrel at a dosage of 75 mg daily after PCI with stent placement (minimum of one month and up to 12 months for bare-metal stent [two weeks if patient is at increased risk of bleeding], at least 12 months for drug-eluting stent if patient is not at increased risk of bleeding) I(B) | ||
| Long-term daily maintenance therapy (one year) with 75 mg of clopidogrel is reasonable in patients with STEMI, regardless of whether they underwent reperfusion with fibrinolytic therapy IIa(C) | ||
| Continue clopidogrel for at least 14 days after PCI without stent placement I(B) | ||
| Warfarin (Coumadin) | ||
| Manage warfarin therapy to achieve an INR of 2.0 to 3.0 in patients after STEMI when clinically indicated (e.g., atrial fibrillation, left ventricular thrombus) I(A) | ||
| Monitor patients closely, because the use of warfarin in conjunction with aspirin or clopidogrel is associated with an increased risk of bleeding I(B) | ||
| In patients who require warfarin, clopidogrel, and aspirin therapy, an INR of 2.0 to 2.5 is recommended, with low-dose aspirin (75 to 81 mg) and clopidogrel (75 mg) I(C) | ||
| Renin-angiotensin-aldosterone system blocker therapy | ACE inhibitors | |
| Start ACE inhibitors in patients with an LVEF of 40 percent or less, and in those with hypertension, diabetes, or chronic renal disease, unless contraindicated I(A) | ||
| Consider ACE inhibitor therapy in all other patients I(B) | ||
| Angiotensin receptor blockers | ||
| Start angiotensin receptor blockers in patients who are intolerant of ACE inhibitors and in those with clinical or radiologic signs of HF or an LVEF of 40 percent or less I(A) | ||
| Aldosterone blockers | ||
| Start aldosterone blockers in patients without significant renal dysfunction†† or hyperkalemia‡‡ who are already receiving therapeutic doses of an ACE inhibitor and beta blocker, and who have an LVEF of 40 percent or less and have diabetes or HF I(A) | ||
| Beta blocker therapy | Start and continue indefinitely in all patients unless contraindicated I(A) | |
| Influenza vaccination | Patients with cardiovascular disease should have annual influenza vaccination I(B) | |
ACC = American College of Cardiology; ACE = angiotensin-converting enzyme; AHA = American Heart Association; BMI = body mass index; HDL-C = high-density lipoprotein cholesterol; HF = heart failure; INR = International Normalized Ratio; LDL-C = low-density lipoprotein cholesterol; LVEF = left ventricular ejection fraction; MI = myocardial infarction; PCI = percutaneous coronary intervention; STEMI = ST-segment elevation myocardial infarction.
*— See Table 3 for explanations of ACC/AHA levels of evidence.
†— Non–HDL-C = total cholesterol – HDL-C.
‡— Pregnant and lactating women should limit their intake of fish to minimize exposure to methylmercury.
§— When LDL-lowering medications are used, a 30 to 40 percent reduction in LDL-C levels should be obtained. If an LDL-C level of less than 70 mg per dL is desired, consider drug titration to minimize side effects and cost. When an LDL-C level of less than 70 mg per dL is not achievable because of high baseline LDL-C levels, it may be possible to achieve LDL-C reductions of greater than 50 percent by using statins or LDL-C–lowering drug combinations.
‖— Standard dose of statin with ezetimibe (Zetia), bile acid sequestrant, or niacin. (note:This guideline update was published before the controversy over ezetimibe.)
¶— The combination of high-dose statin plus fibrate can increase the risk of severe myopathy. Statin doses should be kept relatively low with this combination. Supplemental dietary niacin must not be used as a substitute for prescription niacin, and over-the-counter niacin should be used only if approved and monitored by a physician.
**— Patients with very high triglyceride levels should not consume alcohol. The use of bile acid sequestrants is relatively contraindicated when triglyceride levels are greater than 200 mg per dL.
††— Creatinine levels should be less than 2.5 mg per dL (220 μmol per L) in men and less than 2.0 mg per dL (180 μmol per L) in women.
‡‡— Potassium levels should be less than 5.0 mEq per L (5.0 mmol per L).
Adapted from Antman EM, Anbe DT, Armstrong PW, et al. ACC/AHA guidelines for the management of patients with ST-elevation myocardial infarction: a report of the American College of Cardiology/American Heart Association Task Force on Practice Guidelines (Committee to Revise the 1999 Guidelines for the Management of Patients with Acute Myocardial Infarction) [published corrections appear in Circulation. 2005;111(15):2013–2014, and Circulation. 2007;115(15):e411]. Circulation. 2004;110(9):e213–e214.
The update highlights changes in the management of musculoskeletal pain, recommending a stepped-care approach. Acetaminophen or aspirin (used with caution), tramadol (Ultram), short-term narcotic analgesics, or nonacetylated salicylates should be used first; if these agents are ineffective, non–COX-2 selective NSAIDs (e.g., naproxen [Naprosyn]) are reasonable. In patients with intolerable persistent discomfort, NSAIDs with increasing degrees of relative COX-2 selectivity may be considered. These agents should be used at the lowest effective dosage for the shortest possible time.
Secondary Prevention
Secondary prevention has a key role in the management of STEMI. Patients with CHD or a CHD equivalent (e.g., diabetes, peripheral vascular disease, chronic kidney disease, risk factor calculation of more than 20 percent) should receive intensive secondary prevention interventions. These interventions confer large absolute risk reductions for subsequent events and mortality. Tables 2 and 3 summarize recommendations for secondary prevention in patients with coronary and other vascular diseases, and rate the evidence for each recommendation.1
Table 3 ACC/AHA Classification of Recommendations
| Rating | Explanation | |
|---|---|---|
| Class I | Conditions for which there is evidence or general agreement that a procedure or treatment is beneficial, useful, and effective | |
| Class II | Conditions for which there is conflicting evidence or a divergence of opinion about the effectiveness of a procedure or treatment | |
| Class IIa | Weight of evidence or opinion is in favor of effectiveness | |
| Class IIb | Effectiveness is less well-established by evidence or opinion | |
| Class III | Conditions for which there is evidence or general agreement that a procedure or treatment is not effective and in some cases may be harmful | |
| LOE A | Data derived from multiple randomized clinical trials or meta-analyses | |
| LOE B | Data derived from a single randomized trial or nonrandomized studies | |
| LOE C | Consensus opinion of experts, case studies, or standard of care | |
ACC = American College of Cardiology; AHA = American Heart Association; LOE = level of evidence.
Adapted from Antman EM, Anbe DT, Armstrong PW, et al. ACC/AHA guidelines for the management of patients with ST-elevation myocardial infarction: a report of the American College of Cardiology/American Heart Association Task Force on Practice Guidelines (Committee to Revise the 1999 Guidelines for the Management of Patients with Acute Myocardial Infarction) [published corrections appear in Circulation. 2005;111(15):2013–2014, and Circulation. 2007;115(15):e411]. Circulation. 2004;110(9):e86.

