A 52-year-old, otherwise healthy woman presented with a cough and purulent sputum that persisted for more than 10 days. She denied dyspnea on exertion, palpitations, or light-headedness. Physical examination findings were unremarkable. Chest radiography showed multiple small nodules in both lung fields. Contrast-enhanced computed tomography (CT) of the chest showed multiple lesions with a serpiginous appearance (Figure 1) and dilation of adjacent arteries and veins. Magnetic resonance angiography (MRA) showed several nodular and tubular lesions (Figure 2) in the peripheral portions of the lungs, with dilation of feeding arteries and drainage veins, which is consistent with vascular malformations.
Figure 1.
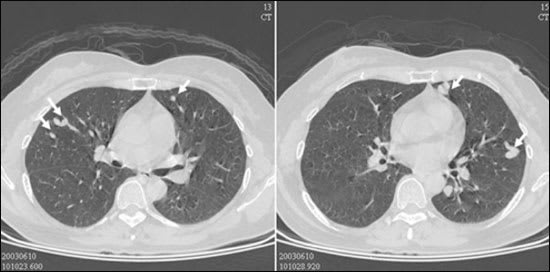
Contrast-enhanced computed tomography of the chest showing multiple lesions (arrows) with a serpiginous appearance.
Figure 2.
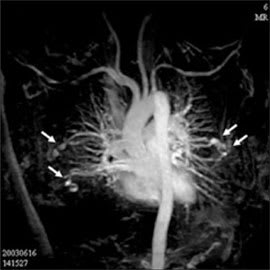
Magnetic resonance angiography showing nodular and tubular lesions (arrows).
Question
Based on the patient's history, physical examination, and imaging studies, which one of the following is the most likely diagnosis?
A. Malignancy with lung metastasis.
B. Pulmonary cryptococcosis.
C. Pulmonary tuberculosis.
D. Rendu-Osler-Weber syndrome.
E. Wegener granulomatosis.
Discussion
The answer is D: Rendu-Osler-Weber syndrome. Rendu-Osler-Weber syndrome, also called hereditary hemorrhagic telangiectasia, is a disorder of vascular development that is inherited as an autosomal dominant trait. It is underdiagnosed because mild phenotypes may be difficult to detect clinically. According to epidemiologic studies, the incidence of Rendu-Osler-Weber syndrome is one in 5,000 to 8,000 persons worldwide.1
Pulmonary arteriovenous malformations associated with Rendu-Osler-Weber syndrome are usually asymptomatic; however, cyanosis, dyspnea, clubbing, and thoracic murmurs may occur. Recurrent epistaxis and mucocutaneous telangiectasis with a family history of the syndrome are diagnostic. Other extrapulmonary presentations of the syndrome include seizures, transient cerebrovascular ischemia, hepatic involvement, and brain abscess.2
Diagnosis of pulmonary arteriovenous malformations is dependent on imaging studies, including chest radiography, contrast echocardiography, angiography, and MRA. Chest radiography may show increased vascular markings and a pulmonary mass, which may be mistaken for a neoplasm or tuberculoma.3 Dyspnea or orthodeoxia on pulse oximetry should prompt evaluation for pulmonary arteriovenous malformations using more sensitive methods, such as contrast echocardiography. This method traces the circulatory transit of microbubbles generated by intravenous echocontrast media. Angiography may help to define the morphology of vascular lesions, including complexity and size of vessels. MRA is becoming a more common diagnostic tool for pulmonary arteriovenous malformations.4
Based on patient history and chest radiograph findings, it is difficult to distinguish Rendu-Osler-Weber syndrome from pulmonary cryptococcosis, malignancy with lung metastasis, pulmonary tuberculosis, and Wegener granulomatosis. Pulmonary arteriovenous malformations may have a sharper nodule margin and a more homogenous density on chest radiography. However, the chest CT and MRA findings make the diagnosis more definitive. The patient's family history further confirms the diagnosis.
Selected Differential Diagnosis of Multiple Pulmonary Nodules
| Condition | Characteristics |
|---|---|
| Cryptococcosis | Immunocompromise; fever; malaise; cough with scant sputum; central nervous system coinfection is common; infiltrates may be bilateral, unilateral, multilobar, or lobar |
| Malignancy with lung metastasis | History of malignancy; nonspecific respiratory symptoms; multiple, well-defined, bilateral nodules that vary in size; lower lung field predominance |
| Pulmonary tuberculosis | Occurs in endemic areas, cough, fever, loss of body weight, tuberculoma predominant in upper lung field, solitary or multiple nodules, nodule with calcification, cavitation, and satellite lesion |
| Rendu-Osler-Weber syndrome | Family history, cyanosis, dyspnea, clubbing, thoracic murmurs, recurrent epistaxis, mucocutaneous telangiectatic lesions, seizures, transient cerebrovascular ischemia, hepatic involvement, brain abscess, solitary or multiple nodules, dilation of feeding arteries and drainage veins present on magnetic resonance angiography |
| Wegener granulomatosis | Autoimmune disease with antineutrophil cytoplasmic antibodies, sinusitis, otitis media, hearing loss, hematuria, multiple bilateral nodes (with or without cavitation), or parenchymal infiltrates |

