Pelvic organ prolapse, or genital prolapse, is the descent of one or more of the pelvic structures (bladder, uterus, vagina) from the normal anatomic location toward or through the vaginal opening. Women of all ages may be affected, although pelvic organ prolapse is more common in older women. The cause is a loss of pelvic support from multiple factors, including direct injury to the levator ani, as well as neurologic injury from stretching of the pudendal nerves that may occur with vaginal childbirth. Previous hysterectomy for pelvic organ prolapse; ethnicity; and an increase in intra-abdominal pressure from chronic coughing, straining with constipation, or repeated heavy lifting may contribute. Most patients with pelvic organ prolapse are asymptomatic. A sense of bulging or protrusion in the vagina is the most specific symptom. Evaluation includes a systematic pelvic examination. Management options for women with symptomatic prolapse include observation, pelvic floor muscle training, mechanical support (pessaries), and surgery. Pessary use should be considered before surgery in women who have symptomatic prolapse. Most women can be fitted with a pessary regardless of the stage or site of predominant prolapse. Surgical procedures are obliterative or reconstructive.
Pelvic organ prolapse is the descent of one or more of the pelvic structures (bladder, uterus, vagina) from the normal anatomic location toward or through the vaginal opening.1 Pelvic organ prolapse may be associated with urinary incontinence or defecatory dysfunction.
Epidemiology
The prevalence of pelvic organ prolapse varies widely across studies, depending on the population studied and entry criteria. Women of all ages may be affected, although it is more common in older women. In the Women's Health Initiative study, investigators found a 41.1 percent prevalence of pelvic organ prolapse at a standard physical assessment in postmenopausal women older than 60 years who had not had a hysterectomy.2
SORT: KEY RECOMMENDATIONS FOR PRACTICE
| Clinical recommendation | Evidence rating | References |
|---|---|---|
| Women should be asked about symptoms of pelvic organ prolapse because they may not volunteer the information. | C | 6, 16 |
| Lifestyle interventions such as weight loss may help improve or prevent symptoms of pelvic organ prolapse, although the evidence is conflicting. | B | 21, 40 |
| Pessaries can be used for the nonsurgical treatment of pelvic organ prolapse in appropriate patients. | B | 33, 34, 37 |
A = consistent, good-quality patient-oriented evidence; B = inconsistent or limited-quality patient-oriented evidence; C = consensus, disease-oriented evidence, usual practice, expert opinion, or case series. For information about the SORT evidence rating system, go to https://www.aafp.org/afpsort.xml.
Etiology
The cause of pelvic organ prolapse is multi-factorial, resulting from loss of the support maintained by a complex interaction among the levator ani, the vagina, and the connective tissue, as well as neurologic injury from stretching of the pudendal nerves that may occur during childbirth. In a healthy woman in whom the levator ani has normal tone and the vagina has adequate depth, the upper vagina lies nearly horizontal when she is upright. The result is a “flap valve” in which the upper vagina presses against the levator plate when there is an increase in intra-abdominal pressure. When the levator ani loses tone, it moves from a horizontal to a semi-vertical position, creating a widened genital hiatus (i.e., the distance between the external urethral meatus and the posterior midline hymen) that forces the pelvic structures to rely on connective tissue for support. When the connective tissue support also fails, as a result of possible collagen decrease and tearing, prolapse may occur.3,4 Table 1 lists risk factors associated with pelvic organ prolapse.2,5–12
Table 1. Risk Factors for Pelvic Organ Prolapse
| Category | Risk factors |
|---|---|
| Ethnicity | Hispanic adults2 |
| General | Advancing age, increasing body mass index, menopause,5,6 low socioeconomic status7 |
| Increased intra-abdominal pressure | Chronic cough caused by smoking, chronic lung disease,8 straining with chronic constipation or repeated heavy lifting7,9 |
| Obstetric | Current pregnancy, previous prolonged labor, instrumental delivery, episiotomy,10 increasing parity, weight of babies5 * |
| Previous surgery | Hysterectomy,11 previous prolapse surgery |
*— Cesarean delivery may not prevent prolapse.12
Clinical Presentation
TERMINOLOGY
Older terms describing pelvic organ prolapse (e.g., cystocele, urethrocele, rectocele) have been replaced because they imply an unrealistic certainty about the structures on the other side of the vaginal bulge, particularly in women who have had previous pelvic organ prolapse surgery. The current practice is to divide the pelvis into anterior, posterior, and middle or apical compartments.13 Following hysterectomy, prolapse of the vaginal apex with or without prolapse of the anterior and/or posterior vaginal wall is referred to as vault prolapse.13
HISTORY
Most patients with pelvic organ prolapse are asymptomatic.1 Seeing or feeling a bulge of tissue that protrudes to or past the vaginal opening is the most specific symptom.1,14
During a well-woman examination, screening questions (e.g., “Do you see or feel a bulge in your vagina?”) with a thorough pelvic examination are important.1 This is true of patients who are older, obese, or otherwise at risk.14,15 The report of a bulge has an 81 percent positive predictive value and a 76 percent negative predictive value for pelvic organ prolapse.16
The uterus and surrounding pelvic support tend to be dynamic in prolapse, resulting in a variation of symptoms depending on the position of the uterus and pressure of the surrounding structures.1 Consequently, as the day progresses, bulging and discomfort may increase.1 Extensive standing, lifting, coughing, and physical exertion may increase patient awareness of discomfort in the pelvis, vagina, abdomen, and low back. Vaginal discharge may be present in patients with complete uterine prolapse (i.e., procidentia) who have a decubitus ulcer of the cervix or vagina.
Pelvic organ prolapse may progress with increasing body mass index.17 Weight loss does not reverse the prolapse.18 Patients may have difficulty urinating—stress incontinence affects 40 percent of patients with pelvic organ prolapse—or defecating19; therefore, they should be asked about these symptoms because they may not volunteer such information.6,16 Urinary outlet obstruction may occur because of pressure on the urethra in anterior vaginal prolapse and sometimes in large posterior vaginal prolapse. Symptoms may not correlate with the location or severity of the prolapsed compartment.1,20,21 Patients with posterior vaginal prolapse sometimes use manual pressure on the perineum or posterior vagina to help with defecation. These maneuvers are called “splinting.” Sexual activity, body image, and quality of life may be affected.22–24
EXAMINATION
If prolapse is visible at the vaginal introitus or a bulge is noted during the Valsalva maneuver, a systematic examination should be performed. With the patient in a supine position and the head of the examination table elevated to 45 degrees, an appropriately sized vaginal speculum is placed in the vagina to view the cervix or vaginal cuff. While the patient is performing the Valsalva maneuver, the speculum is slowly removed. The extent to which the cervix or the vaginal vault follows the speculum through and out of the vagina is noted. The speculum is disassembled and the posterior or fixed blade is used for examination.
To examine the anterior vaginal wall, the posterior vaginal wall is retracted with the fixed blade and the extent of any anterior vaginal prolapse during the Valsalva maneuver is noted. To examine the posterior vaginal wall, the fixed blade is inverted, the anterior vaginal wall is retracted, and the patient is instructed to repeat the Valsalva maneuver. Any resulting prolapse is noted. Decubitus ulcers are inspected and palpated. Bimanual and rectovaginal examinations help identify any coexisting pelvic abnormalities, including those of the perineal body. If pelvic organ prolapse is not evident, especially in a woman feeling a bulge, the patient should be examined in the standing position while she performs the Valsalva maneuver.1
STAGING
The Baden-Walker (grades 0 through 4) and pelvic organ prolapse–quantification (pelvic organ prolapse-Q; stages 0 through IV) are the two main systems for staging the degree of pelvic organ prolapse. Both systems measure the most distal portion of the prolapse during straining/Valsalva maneuver (Table 2).1,13,25 The Baden-Walker system is a reasonable clinical method to evaluate the three pelvic compartments.1,26 The pelvic organ prolapse-Q, an international system that involves taking several measurements, is more complex but highly reliable and is used in clinical assessment and research.1,13,27
Table 2. Evaluation/Staging of Pelvic Organ Prolapse
| Baden-Walker system | Pelvic organ prolapse–quantification system | ||
|---|---|---|---|
| Grade | Description | Stage | Description |
| 0 | Normal position for each respective site, no prolapse | 0 | No prolapse |
| 1 | Descent halfway to the hymen | I | > 1 cm above the hymen |
| 2 | Descent to the hymen | II | ≤ 1 cm proximal or distal to the plane of the hymen |
| 3 | Descent halfway past the hymen | III | > 1 cm below the plane of the hymen, but protrudes no farther than 2 cm less than the total vaginal length |
| 4 | Maximal possible descent for each site | IV | Eversion of the lower genital tract is complete |
Adapted with permission from Onwude JL. Genital prolapse in women. Clin Evid (Online) . 2007. http://clinicalevidence.bmj.com/ceweb/conditions/who/0817/0817_T1.jsp. Accessed March 1, 2010, with additional information from references 1 and 13.
FURTHER EVALUATION
Further studies depend on the symptoms, stage of pro-lapse, and treatment plan. If needed for definitive treatment planning, multichannel urodynamic studies can help identify those patients with urinary symptoms who are most likely to benefit from surgery.28–30 Patients with defecatory symptoms and/or fecal incontinence may need anal manometry, dynamic defecography, and endoanal ultrasonography.31
Treatment
Management options for women with symptomatic pelvic organ prolapse include observation, pelvic floor muscle training, mechanical support (pessaries), and surgery. The goal of conservative management is to improve symptoms, reduce progression, and avoid or delay surgical treatment.32–37 There are no studies comparing surgical and nonsurgical approaches. A patient's perception of discomfort from pelvic organ prolapse and subsequent treatment will vary depending on the stage of the pelvic organ prolapse and her ethnicity.6,20,38
OBSERVATION
Asymptomatic or mildly symptomatic women with pelvic organ prolapse can be observed without intervention at regular intervals.1 Pelvic organ prolapse can regress. Stage I observation is an option.39 Lifestyle modifications (e.g., weight management, smoking cessation, avoidance of heavy lifting and constipation) may reduce symptoms. Although weight loss does not treat or prevent pelvic organ prolapse,1 it can reduce symptoms and may be recommended as a preoperative measure.40
PELVIC FLOOR MUSCLE TRAINING
Pelvic floor muscle training, the systematic contraction of the muscles of the pelvic floor, may improve pelvic function.1 These exercises, commonly known as Kegel exercises, can be accomplished by conscious contractions, electrical stimulation, or via biofeedback training. The use of Kegel cones (weighted cones used to help women isolate pelvic floor muscles) can also help (Figure 1). The effectiveness of pelvic floor muscle training in reversing or treating pelvic organ prolapse has not been studied. However, pelvic floor muscle training has been shown to improve symptoms associated with stress, urge, and mixed urinary incontinence.1,32
Figure 1.
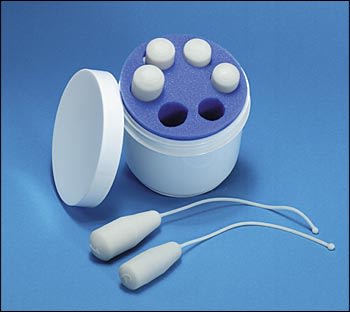
Kegel cones. A set of weighted cones used as an exercise aid with Kegel exercises, especially when patients have trouble isolating pelvic floor muscles.
MECHANICAL SUPPORT
A pessary is a device placed in the vagina to restore pro-lapsed organs to their normal anatomic position. Pessaries are supportive or space occupying and are used for all stages of pelvic organ prolapse in women with or without urinary incontinence41 (Figure 2, Tables 342 and 442). Medical-grade silicone or latex rubber pessaries are used because of their durability, ease of cleaning, inertness, and decreased absorption of secretions and odor.41 The option of a pessary should be discussed with all women who have pelvic organ prolapse that warrants treatment based on symptoms.1
Figure 2.
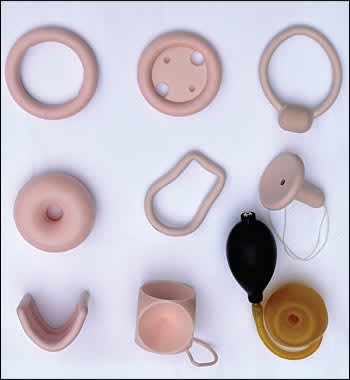
Some common pessaries. First row (left to right): ring, ring with support, and incontinence ring; second row: donut, Smith-Hodge, and Gellhorn; third row: Gehrung, cube, and Inflatoball.
Table 3. Considerations for Choosing an Appropriate Pessary
| Type of pessary | Type of pelvic organ prolapse for which pessary is best suited (Table 2) | Daily removal | Difficulty of use | Erosion risk | Intercourse possible | Urinary incontinence aid | Comments |
|---|---|---|---|---|---|---|---|
| Ring* | All | No | Low | Low | Yes | Minimal | Most commonly used pessary; most practical and acceptable to patients |
| Ring with support*† | All | No | Low | Low | Yes | Minimal | Less helpful in severe forms of prolapse |
| Incontinence ring* | All, plus stress urinary incontinence | No | Low | Low | Yes | Yes | — |
| Donut†‡ | All, especially stages III and IV | No | Moderate | Low | No | No | — |
| Smith-Hodge* | Pregnancy uterine retroversion | No | Low | Low | Yes | No | Used in mid-trimester pregnancy if symptomatic |
| Gellhorn‡ | All, especially advanced stages III and IV; apical (uterine/vault prolapse) | No | High | Moderate | No | No | — |
| Gehrung* | Anterior and posterior vaginal wall prolapse | No | High | Low | Yes | No | Rarely used |
| Cube‡ | Advanced apical (uterine/vault prolapse) | Yes | High | High | No | No | Usually used as a last resort after trying other pessaries because of need for daily removal, vaginal discharge, difficulty of use, and risk of erosion |
| Inflatoball‡ | Advanced apical (uterine/vault prolapse) | Yes | Low to moderate | Low | No | No | Avoid in patients allergic to latex; preferable over cube as a last resort; easy to insert but difficult to retain; pessary requires inflation |
note: Pessaries are listed in relative order from most common/easy to use to least common/easy to use.
*— Support pessaries; all are available with incontinence knobs.
†— Most commonly used pessaries.
‡— Space-occupying pessaries; these need to be taken out more often for drainage, odor, or sexual activity; preferred in widened genital hiatus (i.e., greater than 4 cm).
Information from reference 42.
Traditionally, pessaries have been used for short-term symptom relief in women awaiting surgery or for long-term treatment in women with higher stages of prolapse, who are poor surgical candidates, or who have declined surgery.1,33–35 However, pessaries can be used in almost all circumstances when a nonsurgical option is desired.1,33–37
Arthritis, dementia, and comorbidities including active pelvic inflammatory disease, vaginitis, and endometriosis may limit use of a pessary. A pessary should not be placed in patients unlikely to follow instructions for care or follow-up. Follow-up care instruction should be arranged at the time of the pessary fitting to reduce the likelihood of complications.41
PESSARY SELECTION
There are no randomized trials to guide pessary selection for any particular type of device, indications, pattern of replacement, follow-up care, or degree of pelvic organ prolapse.34 In one study, a ring pessary was more likely to be successfully fitted in patients with stage II or III prolapse, and a Gellhorn pessary was more likely to be successfully fitted in patients with stage IV prolapse.36 In another study (PESSRI), the ring with support and Gellhorn were equally effective in relieving prolapse symptoms and voiding dysfunction.37
The following approach to pessary selection is based on clinical experience (Table 4).42 In pelvic organ pro-lapse without incontinence, a ring pessary with support may be tried first.41 If the ring fails, a Gellhorn or a donut pessary may be tried, followed by a combination of pessaries such as a ring plus a Gellhorn or a ring plus a donut. If the patient is not allergic to latex and has an atrophic or narrow vagina, an Inflatoball (made of latex) or a cube pessary is the last option. Women with pelvic organ prolapse and incontinence should try a ring with support and a knob first, followed by a Gellhorn, a combination such as a ring with support and knob pessary plus a donut, and finally a cube.41 Most pessaries designed to treat incontinence have a knob that is positioned in the midline of the vagina under the urethra.43 A Smith-Hodge pessary may be used during pregnancy.41 A Gehrung pessary can be manually shaped but is not often used because insertion and removal are difficult.41
Table 4. A Simplified Approach to Pessary Selection
| Trial | Pessary type |
|---|---|
| 1 | Ring with support (ring with support and knob if urinary incontinence is present) |
| 2 | Gellhorn |
| 3 | Donut |
| 4 | Combination of pessaries: ring plus Gellhorn, ring plus donut, or two donuts (ring with support and knob if urinary incontinence is present) |
| 5 | Cube or Inflatoball |
note: Fitting is successful if the pessary is not expelled with cough or Valsalva maneuver and if the patient is not aware of having the pessary in place during ambulation, voiding, sitting, and defecation.
Information from reference 42.
PESSARY FITTING
A pessary is fitted by trial and error and requires a fitting set much like that used for diaphragm fitting.44 The largest size that can be comfortably accommodated should be tried. Physicians should estimate the length and diameter of the vagina with their fingers to choose the correct size.44 A ring pessary should fit between the pubic symphysis and the posterior fornix. The patient should be instructed to bear down to ensure that the rim of the pessary is positioned at more than one finger breadth above the vaginal introitus.44 A trial of standing, sitting, walking, and toilet use is done to ensure comfort and correct placement.44 The patient should also void to be sure that the urethra is not blocked.44 She should be able to personally place and remove the pessary. The most common complications include spontaneous expulsion, irritation of the vaginal wall, ulceration, bleeding, pain, and odor.45
FOLLOW-UP
Follow-up examinations may be scheduled at two weeks, three months, six months, and one year,46 although less frequent follow-up may also be safe.47 Patients should be asked if they have experienced any vaginal discharge, bleeding, pain, or discomfort.46 The pessary is palpated in situ then removed to check the vagina for ulcerations or erosions. The pessary can be washed with soap and water, dried, and reinserted. If vaginal lesions are noted, the pessary should be removed until the lesions have healed. Although there is no evidence on the effectiveness of vaginal estrogen in the treatment of pelvic organ prolapse, it may be appropriate for postmenopausal women who have substantial atrophy if no contraindications exist.46 Information, videos, and DVDs for physicians on pessaries can be obtained from the manufacturers (Milex: http://www.coopersurgical.com/ourproducts/incontinence; Superior Medical Limited: http://www.superiormedical.com/ob_equip_main.html).
SURGERY
Surgery for pelvic organ prolapse may be obliterative or reconstructive. Patients with notable comorbidities, those who do not desire to maintain the vagina for sexual function, or those who prefer to avoid hysterectomy may be candidates for obliterative surgery (colpocleisis).1 Reconstructive surgery is performed via an open or laparoscopic, abdominal or vaginal route. The open abdominal approach (abdominal sacral colpopexy) is associated with increased cost and longer operating times, hospital stays, and time to return to daily activities compared with the vaginal approach (vaginal sacrospinous ligament fixation).48 The addition of the Burch procedure to sacral colpopexy reduces the risk of postoperative stress incontinence in women with no preoperative stress incontinence.1 The laparoscopic approach allows a quicker return to daily activities, although the operative time is longer than open sacral colpopexy.1 The mortality from urogynecologic surgery increases with each decade of life, with the most common complications occurring in women 80 years and older.1,49

