Life-threatening dermatologic conditions include Rocky Mountain spotted fever; necrotizing fasciitis; toxic epidermal necrolysis; and Stevens-Johnson syndrome. Rocky Mountain spotted fever is the most common rickettsial disease in the United States, with an overall mortality rate of 5 to 10 percent. Classic symptoms include fever, headache, and rash in a patient with a history of tick bite or exposure. Doxycycline is the first-line treatment. Necrotizing fasciitis is a rapidly progressive infection of the deep fascia, with necrosis of the subcutaneous tissues. It usually occurs after surgery or trauma. Patients have erythema and pain out of proportion to the physical findings. Immediate surgical debridement and antibiotic therapy should be initiated. Stevens-Johnson syndrome and toxic epidermal necrolysis are acute hypersensitivity cutaneous reactions. Stevens-Johnson syndrome is characterized by target lesions with central dusky purpura or a central bulla. Toxic epidermal necrolysis is a more severe reaction with full-thickness epidermal necrosis and exfoliation. Most cases of Stevens-Johnson syndrome and toxic epidermal necrolysis are drug induced. The causative drug should be discontinued immediately, and the patient should be hospitalized for supportive care.
Because family physicians may be the first to see patients with life- threatening dermatologic conditions, they must be able to promptly recognize these conditions. The goal of this review is to provide information to facilitate early recognition of emergent dermatoses, aid in identification of distinguishing features, and provide guidelines for early interventions. The primary conditions discussed in this article include Rocky Mountain spotted fever, necrotizing fasciitis, toxic epidermal necrolysis, and Stevens-Johnson syndrome. Other potential emergencies that can present with dermatologic findings are also summarized briefly (Table 1). 1–12
SORT: KEY RECOMMENDATIONS FOR PRACTICE

| Clinical recommendation | Evidence rating | References |
|---|---|---|
| Doxycycline or tetracycline should be used to treat Rocky Mountain spotted fever in nonpregnant adults and in children older than eight years. | C | 7–9 |
| Patients with suspected necrotizing fasciitis should be hospitalized, and a surgical consultation should be obtained for evaluation and debridement. | C | 5, 6 |
| Patients with suspected toxic epidermal necrolysis or Stevens-Johnson syndrome should be admitted for supportive care, and the causative drug should be discontinued. | C | 10–12 |
A = consistent, good-quality patient-oriented evidence; B = inconsistent or limited-quality patient-oriented evidence; C = consensus, disease-oriented evidence, usual practice, expert opinion, or case series. about the SORT evidence rating system, go to https://www.aafp.org/afpsort.xml.
Table 1. Selected Conditions Presenting with Dermatologic Findings
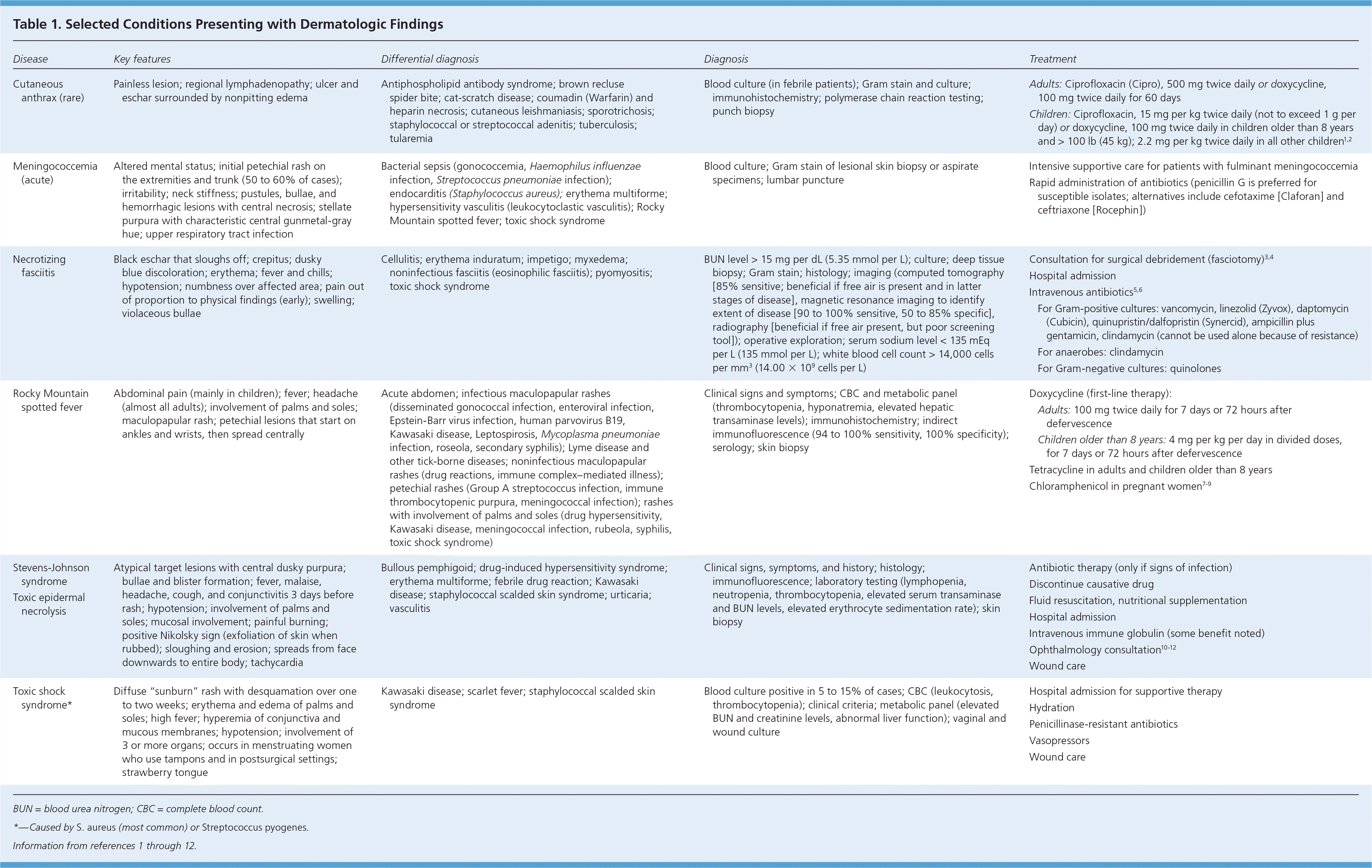
| Disease | Key features | Differential diagnosis | Diagnosis | Treatment | |
|---|---|---|---|---|---|
| Cutaneous anthrax (rare) | Painless lesion; regional lymphadenopathy; ulcer and eschar surrounded by nonpitting edema | Antiphospholipid antibody syndrome; brown recluse spider bite; cat-scratch disease; coumadin (Warfarin) and heparin necrosis; cutaneous leishmaniasis; sporotrichosis; staphylococcal or streptococcal adenitis; tuberculosis; tularemia | Blood culture (in febrile patients); Gram stain and culture; immunohistochemistry; polymerase chain reaction testing; punch biopsy | Adults: Ciprofloxacin (Cipro), 500 mg twice daily or d oxycycline, 100 mg twice daily for 60 days Children: Ciprofloxacin, 15 mg per kg twice daily (not to exceed 1 g per day) or doxycycline, 100 mg twice daily in children older than 8 years and > 100 lb (45 kg); 2.2 mg per kg twice daily in all other children1,2 | |
| Meningococcemia (acute) | Altered mental status; initial petechial rash on the extremities and trunk (50 to 60% of cases); irritability; neck stiffness; pustules, bullae, and hemorrhagic lesions with central necrosis; stellate purpura with characteristic central gunmetalgray hue; upper respiratory tract infection | Bacterial sepsis (gonococcemia, Haemophilus influenzae infection, Streptococcus pneumoniae infection); endocarditis (Staphylococcus aureus); erythema multiforme; hypersensitivity vasculitis (leukocytoclastic vasculitis); Rocky Mountain spotted fever; toxic shock syndrome | Blood culture; Gram stain of lesional skin biopsy or aspirate specimens; lumbar puncture | Intensive supportive care for patients with fulminant meningococcemia Rapid administration of antibiotics (penicillin G is preferred for susceptible isolates; alternatives include cefotaxime [Claforan] and ceftriaxone [Rocephin]) | |
| Necrotizing fasciitis | Black eschar that sloughs off; crepitus; dusky blue discoloration; erythema; fever and chills; hypotension; numbness over affected area; pain out of proportion to physical findings (early); swelling; violaceous bullae | Cellulitis; erythema induratum; impetigo; myxedema; noninfectious fasciitis (eosinophilic fasciitis); pyomyositis; toxic shock syndrome | BUN level > 15 mg per dL (5.35 mmol per L); culture; deep tissue biopsy; Gram stain; histology; imaging (computed tomography [85% sensitive; beneficial if free air is present and in latter stages of disease], magnetic resonance imaging to identify extent of disease [90 to 100% sensitive, 50 to 85% specific], radiography [beneficial if free air present, but poor screening tool]); operative exploration; serum sodium level < 135 mEq per L (135 mmol per L); white blood cell count > 14,000 cells per mm3 (14.00 × 109 cells per L) | Consultation for surgical debridement (fasciotomy)3,4 Hospital admission Intravenous antibiotics5,6
| |
| Rocky Mountain spotted fever | Abdominal pain (mainly in children); fever; headache (almost all adults); involvement of palms and soles; maculopapular rash; petechial lesions that start on ankles and wrists, then spread centrally | Acute abdomen; infectious maculopapular rashes (disseminated gonococcal infection, enteroviral infection, Epstein-Barr virus infection, human parvovirus B19, Kawasaki disease, Leptospirosis, Mycoplasma pneumoniae infection, roseola, secondary syphilis); Lyme disease and other tick-borne diseases; noninfectious maculopapular rashes (drug reactions, immune complex–mediated illness); petechial rashes (Group A streptococcus infection, immune thrombocytopenic purpura, meningococcal infection); rashes with involvement of palms and soles (drug hypersensitivity, Kawasaki disease, meningococcal infection, rubeola, syphilis, toxic shock syndrome) | Clinical signs and symptoms; CBC and metabolic panel (thrombocytopenia, hyponatremia, elevated hepatic transaminase levels); immunohistochemistry; indirect immunofluorescence (94 to 100% sensitivity, 100% specificity); serology; skin biopsy | Doxycycline (first-line therapy):
Chloramphenicol in pregnant women7–9 | |
| Stevens-Johnson syndrome Toxic epidermal necrolysis | Atypical target lesions with central dusky purpura; bullae and blister formation; fever, malaise, headache, cough, and conjunctivitis 3 days before rash; hypotension; involvement of palms and soles; mucosal involvement; painful burning; positive Nikolsky sign (exfoliation of skin when rubbed); sloughing and erosion; spreads from face downwards to entire body; tachycardia | Bullous pemphigoid; drug-induced hypersensitivity syndrome; erythema multiforme; febrile drug reaction; Kawasaki disease; staphylococcal scalded skin syndrome; urticaria; vasculitis | Clinical signs, symptoms, and history; histology; immunofluorescence; laboratory testing (lymphopenia, neutropenia, thrombocytopenia, elevated serum transaminase and BUN levels, elevated erythrocyte sedimentation rate); skin biopsy | Antibiotic therapy (only if signs of infection) Discontinue causative drug Fluid resuscitation, nutritional supplementation Hospital admission Intravenous immune globulin (some benefit noted) Ophthalmology consultation10–12 Wound care | |
| Toxic shock syndrome* | Diffuse “sunburn” rash with desquamation over one to two weeks; erythema and edema of palms and soles; high fever; hyperemia of conjunctiva and mucous membranes; hypotension; involvement of 3 or more organs; occurs in menstruating women who use tampons and in postsurgical settings; strawberry tongue | Kawasaki disease; scarlet fever; staphylococcal scalded skin syndrome | Blood culture positive in 5 to 15% of cases; CBC (leukocytosis, thrombocytopenia); clinical criteria; metabolic panel (elevated BUN and creatinine levels, abnormal liver function); vaginal and wound culture | Hospital admission for supportive therapy Hydration Penicillinase-resistant antibiotics Vasopressors Wound care | |
BUN = blood urea nitrogen; CBC = complete blood count.
*—Caused by S. aureus (most common) or Streptococcus pyogenes.
Rocky Mountain Spotted Fever
Key findings in patients with Rocky Mountain spotted fever include history of a tick bite, fever, and a characteristic rash. Between 60 and 75 percent of patients with this condition are given an alternate diagnosis at the initial physician visit.13
EPIDEMIOLOGY
Rocky Mountain spotted fever is the most common rickettsial disease in the United States. It has an overall mortality rate of 5 to 10 percent.8,14 In the United States, cases occur most often in the southern Atlantic and south central states (Arkansas, Louisiana, Oklahoma, Texas, Kentucky, Tennessee, Alabama, and Mississippi).7 Rocky Mountain spotted fever is associated with seasonal outbreaks; 90 percent of cases occur between April and September.7 Persons who are most likely to be exposed to ticks (e.g., persons living near wooded, high-grass areas; dog owners) are at greatest risk. Cases of Rocky Mountain spotted fever are most often reported in American Indians, whites, males, and children.7,15
ETIOLOGY
Rocky Mountain spotted fever is transmitted by Rickettsia rickettsii, a small gram-negative bacterium that grows in eukaryotic cells. The natural hosts for this organism consisting of the wood tick (Dermacentor andersoni) in the western United States and the dog tick (Dermacentor variabilis) in the eastern United States. R. rickettsii is transmitted via tick bites and spreads through the lymphatic system, causing a vasculitis that can affect all organs, most commonly the skin and adrenals. Ultimately, R. rickettsii causes increased vascular permeability with consequential decreased osmotic pressure, edema, hypoalbuminemia, hyponatremia, and hypotension.
DIAGNOSIS
Clinical Findings. The diagnosis of Rocky Mountain spotted fever is primarily clinical. The classic triad of symptoms associated with the disease includes fever, headache, and rash in a person with a history of tick bite or exposure to ticks.8 Patients also may present with nausea, vomiting, myalgia, anorexia, and severe abdominal pain (especially in children). The rash appears two to five days after the onset of fever in 80 to 90 percent of patients.9 Many patients present before the rash appears; therefore, a high index of suspicion is warranted in endemic areas when a patient presents with severe symptoms that mimic a viral illness.
The rash begins as small, blanching, nonpruritic macules that develop into a maculopapular rash and progress after several days to petechial lesions. The classic spotted appearance occurs when the petechial lesions coalesce and form ecchymosis (Figure 1). 3 In later stages of the disease, the skin can become tender and desquamate. The early rash of Rocky Mountain spotted fever mimics many infectious and noninfectious diseases that have an associated maculopapular rash.16 The petechial rash that occurs in Rocky Mountain spotted fever also occurs in other conditions17 (Table 1).1–12 It begins around the wrists and ankles and spreads to the palms and soles, eventually covering the extremities and trunk. The face is usually spared. A rash on the palms and soles is a distinct distribution that occurs in approximately 40 percent of patients with Rocky Mountain spotted fever.9,17 However, other exanthems can present with fever and rash on the palms and soles.16 In patients with severe disease in whom treatment is delayed, complications can include cardiac and renal failure, meningitis, and pneumonitis.
Figure 1.
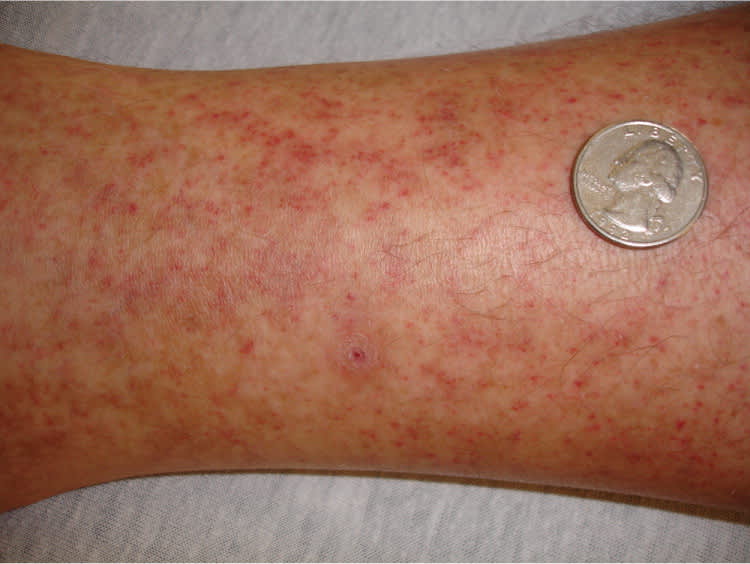
Petechial lesions on the leg of a patient who developed Rocky Mountain spotted fever after a tick bite.
Reprinted with permission from Mayeaux EJ Jr, Usatine R. Cutaneous vasculitis. In: Usatine R, Smith MA, Mayeaux EJ Jr, Chumley H, Tysinger J. The Color Atlas of Family Medicine. New York, NY: McGraw-Hill; 2009:765.
Laboratory Testing. Basic laboratory tests that should be ordered in patients with suspected Rocky Mountain spotted fever include a complete blood count and blood chemistry profile, including measurement of electrolytes and creatinine, and liver function tests. Laboratory findings may include thrombocytopenia, hyponatremia, and elevated hepatic transaminase levels with a normal white blood cell count. Cerebrospinal fluid analysis can be performed if neurologic symptoms are present. If a rash is present, direct immunofluorescence of a punch biopsy fixed in special media can detect rickettsial organisms within the vascular endothelium. A standard punch biopsy fixed in formalin may show necrotizing vasculitis. Indirect immunofluorescence of blood samples drawn for immunoglobulin G (IgG) and IgM anti- R. rickettsii antibodies will show a fourfold increase in titers from the acute to convalescent stages.8,9,18 It may take seven to 10 days after the onset of illness before titers are detectable.9
TREATMENT
Treatment for Rocky Mountain spotted fever should begin immediately after diagnosis, and cases must be reported to the state health department. Patients treated more than five days after the onset of symptoms have a mortality rate three times greater than that in patients treated earlier.8,18 The mainstay of treatment is antibiotics; doxycycline is the first-line agent. Tetracycline is an alternative in adults and children older than eight years, and chloramphenicol is preferred in pregnant women.7–9 Hospitalization should be considered for severely ill patients and those with a history of glucose-6-phosphate dehydrogenase deficiency, who are at higher risk of complications.
Necrotizing Fasciitis
Necrotizing fasciitis is a rapidly progressive infection of the deep fascia, with necrosis of the subcutaneous tissues.
EPIDEMIOLOGY
Necrotizing fasciitis has a mortality rate of 25 percent, and up to 70 percent in patients who develop sepsis.3,5 The most common risk factors for necrotizing fasciitis are diabetes mellitus, severe peripheral vascular disease, trauma, alcohol or injection drug use, and the use of nonsteroidal anti-inflammatory drugs.
ETIOLOGY
There are two types of necrotizing fasciitis, which are grouped according to etiology and infectious pathogen. Type I, polymicrobial necrotizing fasciitis, is usually preceded by trauma or surgery. The most common pathogens are Streptococci(non-group A), Bacteroides, Enterobacteriaceae, and Pepto-streptococcus.3 Type II necrotizing fasciitis is caused by a single organism. The most common pathogens associated with this type include group A beta-hemolytic streptococci and Staphylococcus aureus.3 The most common sites of necrotizing fasciitis are the lower extremities, abdomen, and perineum. Necrotizing fasciitis of the genital and perineal areas is called Fournier gangrene. In all types, bacterial infection leads to edema, vascular occlusion, ischemia, tissue necrosis, and, if not treated promptly, sepsis.
DIAGNOSIS
Clinical Findings. Patients with necrotizing fasciitis often have a history of a wound or trauma. Common symptoms include fever and chills, with heat, erythema, edema, and pain over a focal area of skin (Figure 2). 19 The pain is typically out of proportion to the physical findings, which can help distinguish this condition from cellulitis.
Figure 2.
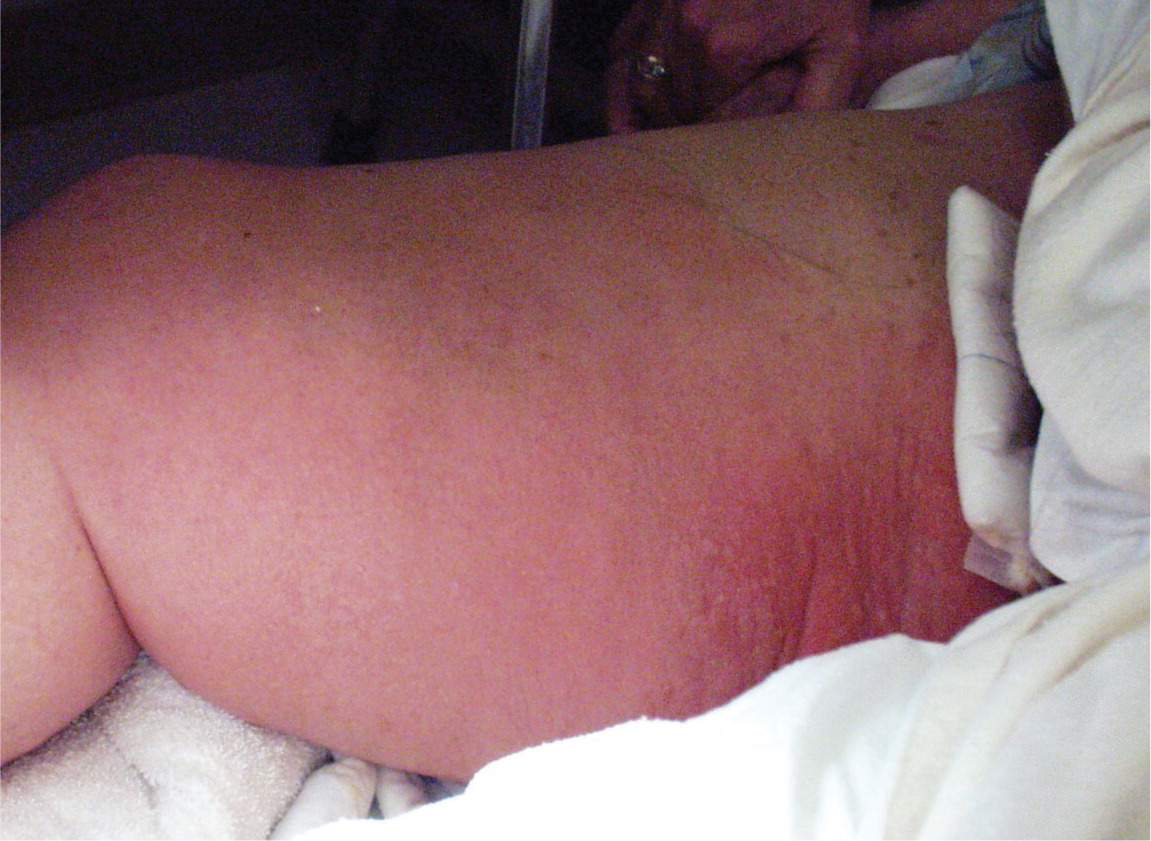
Erythema, swelling, and bullae on the leg and groin of a patient with necrotizing fasciitis.
Reprinted with permission from Dufel S, Martino M. Simple cellulitis or a more serious infection? J Fam Pract. 2006;55(5):396.
The erythematous skin develops a dusky blue discoloration. Yellow vesicular and bullous lesions form over the erythematous skin, with some serosanguineous drainage. Impetigo, cellulitis, and toxic shock syndrome can mimic these lesions. After four to five days, the bullae become violaceous, and the skin becomes gangrenous.20 The presence of violaceous bullae strongly signals necrotizing fasciitis. Anesthesia, another key feature of necrotizing fasciitis, develops as a result of infarction of cutaneous nerves. The black eschar that is formed sloughs off by day 10. Crepitus also occurs in 35 percent of patients.6,21
Laboratory Testing and Imaging. There is no specific diagnostic test for necrotizing fasciitis. Common nonspecific findings include a white blood cell count greater than 14,000 cells per mm3 (14.00 × 109 cells per L), a serum sodium level less than 135 mEq per L (135 mmol per L), and a blood urea nitrogen level greater than 15 mg per dL (5.35 mmol per L). These parameters can help distinguish necrotizing fasciitis from other soft tissue infections. Histology and culture of deep tissue biopsy are essential; surface cultures cannot be relied on alone. Gram staining of the exudate may provide clues about the pathogens while the physician awaits culture results. Radiography, computed tomography, ultrasonography, and magnetic resonance imaging can be used to detect pyomyositis or gas within soft tissues or muscles.9,22 Any such findings should prompt an immediate surgical referral.
TREATMENT
First-line treatment for necrotizing fasciitis is immediate hospitalization, surgical debridement, and antibiotic therapy.5,6 A broad-spectrum antibiotic with coverage of gram-negative and gram-positive anaerobes and aerobes should be administered empirically3,5,20 (Table 1).1–12 Fluid resuscitation is also necessary. Although they are controversial, hyperbaric oxygen therapy and intravenous immune globulin may have a role as adjunct therapies.22
Toxic Epidermal Necrolysis and Stevens-Johnson Syndrome
Toxic epidermal necrolysis and Stevens-Johnson syndrome are acute hypersensitivity cutaneous reactions that present as dermatologic emergencies. They are thought to be severe presentations of erythema multiforme. By definition, Stevens-Johnson syndrome affects less than 10 percent of the body surface area, toxic epidermal necrolysis affects more than 30 percent of the body surface area, and Stevens-Johnson syndrome/toxic epidermal necrolysis involves 15 to 30 percent of the body surface area.3,10
EPIDEMIOLOGY
In the United States, Stevens-Johnson syndrome and toxic epidermal necrolysis most often occur during early spring and winter. Most affected patients are 20 to 40 years of age, and the male-to-female ratio is 2:1.3,10 Mortality is approximately 5 percent for Stevens-Johnson syndrome, and 30 percent for toxic epidermal necrolysis.3,10
ETIOLOGY
Approximately 50 percent of cases of Stevens-Johnson syndrome and 80 to 90 percent of cases of toxic epidermal necrolysis are drug induced.23 Common causative agents include sulfa drugs, antiepileptic drugs, antibiotics, and nonsteroidal anti-inflammatory drugs.20 Noniatrogenic associations include infections, vaccinations, radiation, sunlight exposure, pregnancy, connective tissue disease, and neoplasms. The exact pathogenesis of these conditions is unknown.
DIAGNOSIS
Clinical Findings. After one to three weeks of exposure to the causative agent, a prodrome of fever, malaise, headache, cough, and conjunctivitis develops (Figure 3). 3 The skin lesions appear one to three days after the prodrome23 (Figure 4). 3 The lesions initially appear as erythematous macules with dark purpuric centers, then form atypical target lesions with central dusky purpura or a central bulla, with surrounding macular erythema. The bullae coalesce, forming flaccid blisters with full-thickness epidermal necrosis. Patients with toxic epidermal necrolysis have large areas of epidermal detachment (sloughing) and erosions, which increase the risk of blood and fluid loss, and of superinfection (Figure 5). 3 Lesions begin on the face and trunk, then spread rapidly. The palms and soles become painful, erythematous, and edematous. Mucosal involvement occurs in almost all affected patients.3,20,23 Complications include hypotension, renal failure, corneal ulcerations, anterior uveitis, erosive vulvovaginitis or balanitis, respiratory failure, seizures, and coma. The most common sequelae are ocular scarring and blindness.
Figure 3.
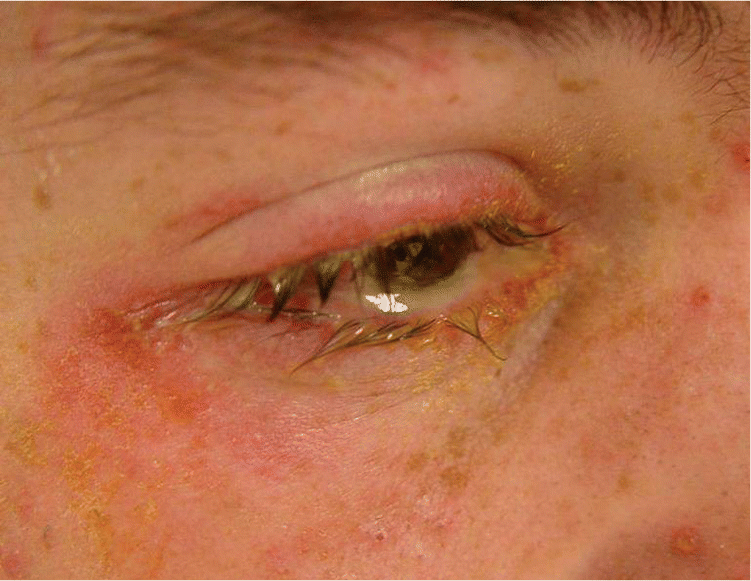
Mucosal involvement of the eye in a patient with Stevens-Johnson syndrome.
Reprinted with permission from Mayeaux EJ Jr, Usatine R. Cutaneous vasculitis. In: Usatine R, Smith MA, Mayeaux EJ Jr, Chumley H, Tysinger J. The Color Atlas of Family Medicine. New York, NY: McGraw-Hill; 2009:752.
Figure 4.
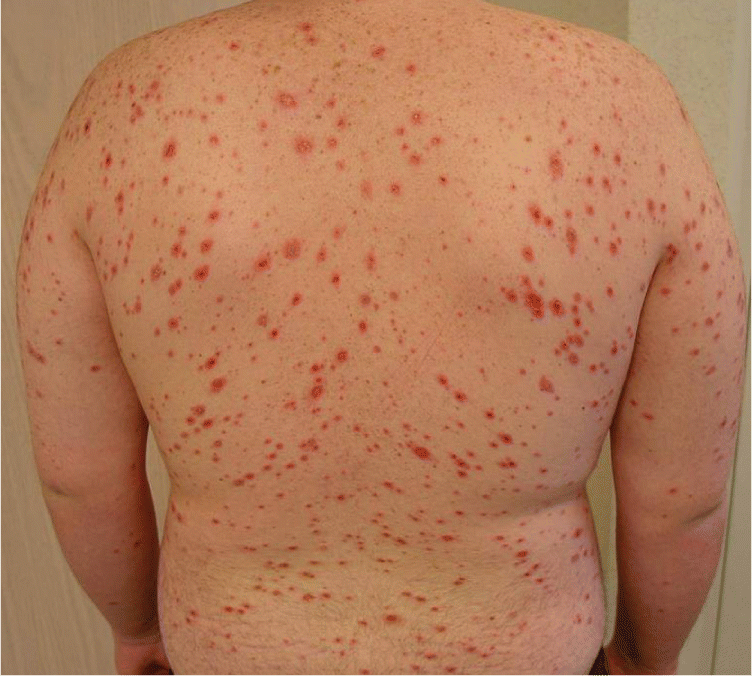
Skin lesions in a 14-year-old boy who developed Stevens-Johnson syndrome after taking penicillin for pneumonia.
Reprinted with permission from Mayeaux EJ Jr, Usatine R. Cutaneous vasculitis. In: Usatine R, Smith MA, Mayeaux EJ Jr, Chumley H, Tysinger J. The Color Atlas of Family Medicine. New York, NY: McGraw-Hill; 2009:752.
Figure 5.
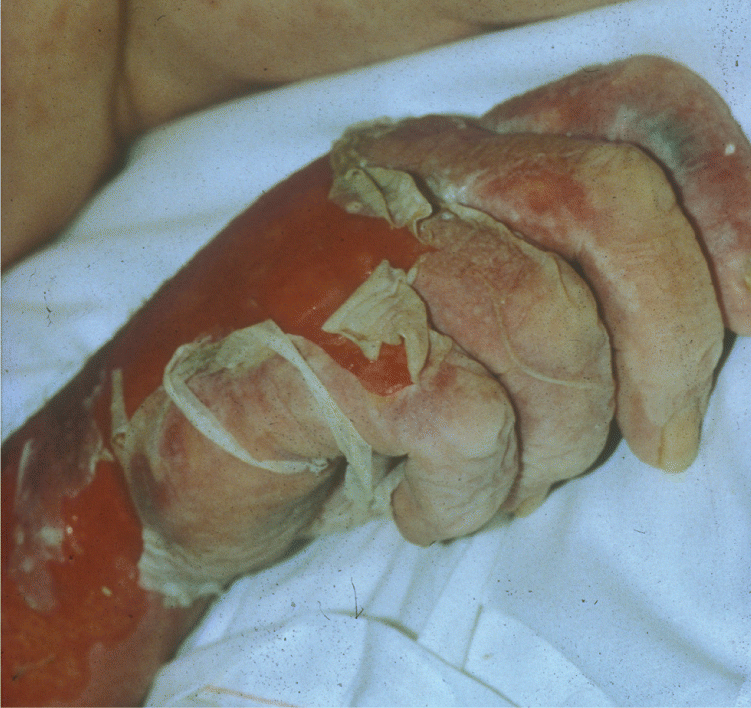
Desquamation of the hand in a patient with toxic epidermal necrolysis.
Reprinted with permission from Mayeaux EJ Jr, Usatine R. Cutaneous vasculitis. In: Usatine R, Smith MA, Mayeaux EJ Jr, Chumley H, Tysinger J. The Color Atlas of Family Medicine. New York, NY: McGraw-Hill; 2009:754.
Laboratory Testing. There is no specific diagnostic test for Stevens-Johnson syndrome or toxic epidermal necrolysis. Lymphopenia may be present, as well as neutropenia (30 percent of patients), thrombocytopenia (15 percent of patients), elevated erythrocyte sedimentation rate, and elevated serum transaminase and blood urea nitrogen levels. Clinical findings and a skin biopsy are useful in diagnosis. Frozen section analysis can facilitate rapid diagnosis and, in combination with immunofluorescence, can exclude other diagnoses.10,20
TREATMENT
Patients with Stevens-Johnson syndrome or toxic epidermal necrolysis should be admitted to an intensive care or burn unit immediately, because rapid deterioration is possible.10–12 The causative drug and any unnecessary medications should be discontinued.10–12 Aggressive management should include fluid resuscitation, nutritional supplementation, and wound care. Antibiotics should be used only if there is evidence of infection. Intravenous immune globulin has been incorporated into some treatment protocols.12 An ophthalmology consultation is necessary to assess for associated ocular sequelae.

