Proper care of patients with asthma involves the triad of systematic chronic care plans, self-management support, and appropriate medical therapy. Controller medications (inhaled corticosteroids, long-acting beta2 agonists, and leukotriene receptor antagonists) are the foundation of care for persistent asthma and should be taken daily on a long-term basis to achieve and maintain control of symptoms. Inhaled corticosteroids are the preferred controller medication; studies have demonstrated that when inhaled corticosteroids are used consistently, they improve asthma control more effectively than any other single long-term control medication. Combining long-acting beta2 agonists and inhaled corticosteroids is effective and safe when inhaled corticosteroids alone are insufficient, and such combinations are an alternative to increasing the dosage of inhaled corticosteroids. For patients with mild persistent asthma, leukotriene receptor antagonists are an alternative, second-line treatment option. They are easy to use, have high rates of compliance, and can provide good symptom control in many patients. Leukotriene receptor antagonists can also be used as an adjunctive therapy with inhaled corticosteroids, but for persons 12 years and older the addition of long-acting beta2 agonists is preferred. Inhaled short-acting beta2agonists are the most effective therapy for rapid reversal of airflow obstruction and prompt relief of asthmatic symptoms. Increasing the use of short-acting beta2agonists or using them more than two days per week or more than two nights per month generally indicates inadequate control of asthma and the need to initiate or intensify anti-inflammatory therapy. Oral systemic corticosteroids should be used to treat moderate to severe asthma exacerbations.
Advances in medical therapy have greatly increased the options for the care of patients with asthma. However, inadequate medical therapy for asthma is still one of the most common reasons for patients to seek unplanned care.1 Each year, despite the availability of a wide and effective array of asthma treatments, patients with asthma make approximately 1.8 million visits to emergency departments, 2 with a fivefold greater rate among blacks than among whites.3 The rate of asthma-related deaths is two per 100,000 population per year, and has not changed since 1995.4 In one study of emergency department visits for asthma, chronic underuse of appropriate medication was apparent among many patients.5 Proper asthma care involves the triad of systematic chronic care plans, support for self-management of asthma, and appropriate medical therapy.6 In this article, we examine the recommendations for medical therapy of asthma from the Expert Panel Report 3 (EPR-3) of the National Asthma Education and Prevention Program (NAEPP).7
SORT: KEY RECOMMENDATIONS FOR PRACTICE
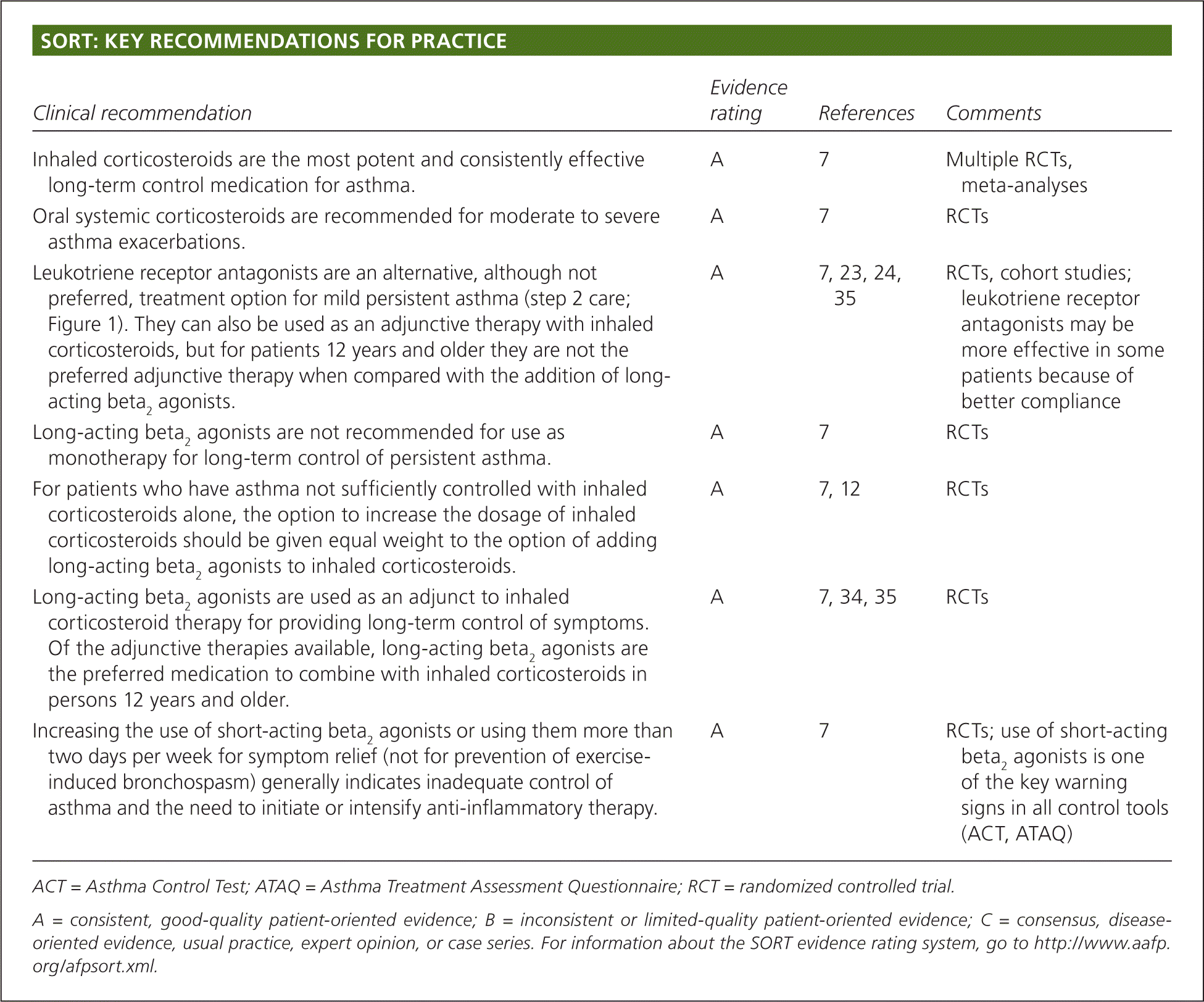
| Clinical recommendation | Evidence rating | References | Comments |
|---|---|---|---|
| Inhaled corticosteroids are the most potent and consistently effective long-term control medication for asthma. | A | 7 | Multiple RCTs, meta-analyses |
| Oral systemic corticosteroids are recommended for moderate to severe asthma exacerbations. | A | 7 | RCTs |
| Leukotriene receptor antagonists are an alternative, although not preferred, treatment option for mild persistent asthma (step 2 care; Figure 1). They can also be used as an adjunctive therapy with inhaled corticosteroids, but for patients 12 years and older they are not the preferred adjunctive therapy when compared with the addition of long-acting beta2 agonists. | A | 7, 23, 24, 35 | RCTs, cohort studies; leukotriene receptor antagonists may be more effective in some patients because of better compliance |
| Long-acting beta2 agonists are not recommended for use as monotherapy for long-term control of persistent asthma. | A | 7 | RCTs |
| For patients who have asthma not sufficiently controlled with inhaled corticosteroids alone, the option to increase the dosage of inhaled corticosteroids should be given equal weight to the option of adding long-acting beta2 agonists to inhaled corticosteroids. | A | 7, 12 | RCTs |
| Long-acting beta2 agonists are used as an adjunct to inhaled corticosteroid therapy for providing long-term control of symptoms. Of the adjunctive therapies available, long-acting beta2 agonists are the preferred medication to combine with inhaled corticosteroids in persons 12 years and older. | A | 7, 34, 35 | RCTs |
| Increasing the use of short-acting beta2agonists or using them more than two days per week for symptom relief (not for prevention of exercise-induced bronchospasm) generally indicates inadequate control of asthma and the need to initiate or intensify anti-inflammatory therapy. | A | 7 | RCTs; use of short-acting beta2 agonists is one of the key warning signs in all control tools (ACT, ATAQ) |
ACT = Asthma Control Test; ATAQ = Asthma Treatment Assessment Questionnaire; RCT = randomized controlled trial.
A = consistent, good-quality patient-oriented evidence; B = inconsistent or limited-quality patient-oriented evidence; C = consensus, diseaseoriented evidence, usual practice, expert opinion, or case series. For information about the SORT evidence rating system, go to https://www.aafp.org/afpsort.xml.
Long-Term Control Medications
Control medications are the foundation of care for persistent asthma and should be taken daily on a long-term basis to achieve and maintain control of symptoms. The major medications are inhaled corticosteroids, leukotriene receptor antagonists, and long-acting beta2 agonists. There is a difference of opinion about calling long-acting beta2agonists “controllers” because when used by themselves, they can be associated with increases in asthma exacerbations and death.8 For the purposes of this article, we will retain the current terminology as used in the EPR-3 guidelines.
INHALED CORTICOSTEROIDS
Studies have shown that when inhaled corticosteroids are used consistently, they improve asthma symptoms more effectively than any other single long-term control medication in both children and adults.7 Patients with mild to moderate persistent asthma treated with inhaled corticosteroids demonstrate improved symptom scores, lower exacerbation rates, and reduced symptom frequency. They also use fewer supplemental short-acting beta2 agonists, take fewer courses of oral systemic corticosteroids, and have fewer hospitalizations, compared with patients taking other single long-term control medications.9
There are no clinically meaningful differences among the various types of inhaled corticosteroids. Bronchoprotective effects of inhaled corticosteroids delivered via dry powder inhaler and hydrofluoroalkane- propelled metered dose inhalers are equivalent.10,11 When used correctly, metered dose inhalers deposit 20 to 30 percent of the dose in the lungs. The use of spacers (more properly termed “valved holding chambers”) markedly increases this percentage.12
There are clinically significant differences in sensitivity and responsiveness to inhaled corticosteroid therapy. This may be related to high levels of inflammation or reduced corticosteroid sensitivity.13 Smokers have a decreased responsiveness to steroids, possibly because of persistent irritation and scarring.14,15 Black children may have an increased risk of corticosteroid insensitivity because of deficiencies in T cell pathways.16,17
Systemic effects of inhaled corticosteroids may occur but typically are not clinically important, except with long-term, high-dose use. Adding a spacer device or changing either the inhaled corticosteroid medication or the delivery system can usually overcome asthma-related cough. Dysphonia, generally intermittent, is thought to be caused by laryngeal edema and mucosal thickening or possibly myopathy.18 It typically resolves with temporary cessation of the medication, but may also resolve with a change from a dry-powder inhaler to a metered dose inhaler with spacer.
Because of their delayed onset of action, inhaled steroids are insufficient for moderate to severe exacerbations.7 Instead, oral steroid treatment is recommended: 1 to 2 mg per kg per day for three to 10 days in children, or 40 to 60 mg per day in one or two divided doses for five to 10 days in adults. Tapering is not necessary.19
LEUKOTRIENE RECEPTOR ANTAGONISTS
There are two widely available leukotriene receptor antagonists: montelukast (Singulair; for patients older than one year) and zafirlukast (Accolate; for patients seven years and older). Montelukast and zafirlukast are appropriate alternative therapies for mild persistent asthma in patients who are unable or unwilling to use inhaled corticosteroids. 7 Leukotriene receptor antagonists have the advantages of ease of use and high rates of compliance,20 and they can provide good control of asthma symptoms in many patients. A recent randomized controlled trial compared fluticasone (Flovent) twice daily, fluticasone/salmeterol (Advair) once daily, and montelukast once daily in children with mild persistent asthma that was well controlled on twice-daily fluticasone. Lung function outcomes improved with the combination therapy, but there were similar results in patient-oriented outcomes among the three arms, and fewer respiratory infections in the montelukast group.21
Montelukast is taken once daily, whereas zafirlukast is used twice daily. For patients 12 years and older, combining leukotriene receptor antagonists and inhaled corticosteroids is an alternative for moderate persistent asthma, but studies of this combination have been limited. Leukotriene receptor antagonists are indicated in exercise-induced bronchospasm and can improve the condition in up to 50 percent of patients. They are the treatment of choice for aspirin-sensitive asthma.7,22
Leukotriene receptor antagonists are an alternative, although not preferred, treatment option for mild persistent asthma (step 2 care; Figure 17 ). They can also be used as an adjunctive therapy with inhaled corticosteroids, but for patients 12 years and older they are not the preferred adjunctive therapy compared with the addition of long-acting beta2 agonists.7,23,24
Figure 1. Stepwise Approach for Asthma Management
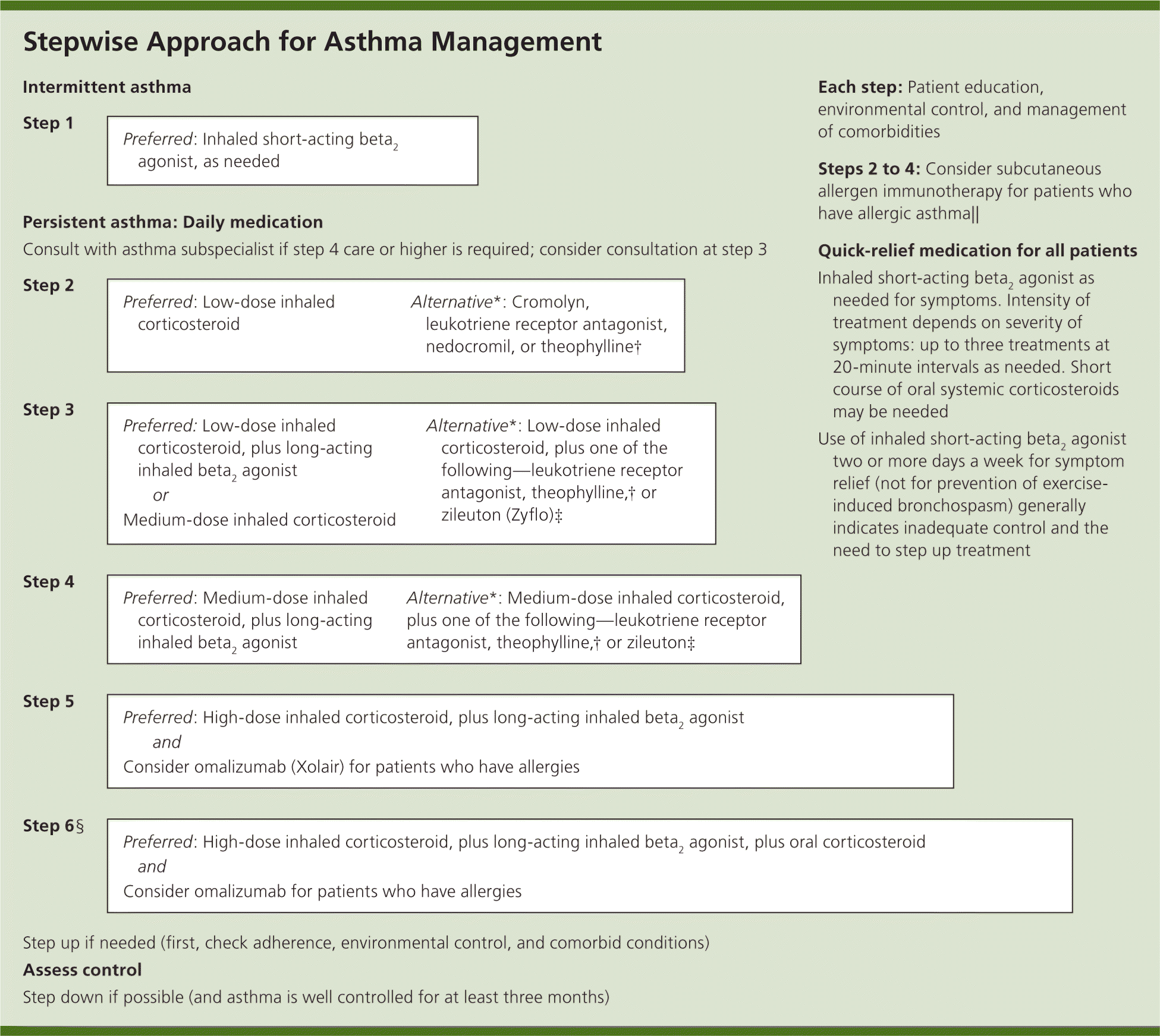
Stepwise approach for managing asthma in patients 12 years and older. Alphabetical order is used when more than one treatment option is listed within preferred or alternative therapy.
note: The stepwise approach is meant to assist, not replace, the clinical decision making required to meet individual patient needs.
*—If alternative treatment is used and response is inadequate, discontinue it and use the preferred treatment before stepping up.
†—Theophylline requires monitoring of serum concentration levels.
‡—Zileuton is a less desirable alternative because of limited studies as adjunctive therapy and the need to monitor liver function.
§—In step 6, before oral systemic corticosteroids are introduced, a trial of high-dose inhaled corticosteroid, plus long-acting inhaled beta2 agonist, plus a leukotriene receptor antagonist, theophylline, or zileuton may be considered, although this approach has not been studied in clinical trials.
∥—Immunotherapy for house-dust mites, animal danders, and pollens is recommended; evidence is weak or lacking for molds and cockroaches. Evidence is strongest for immunotherapy for single allergens. The role of allergy in asthma is greater in children than in adults.
Adapted from National Heart, Lung, and Blood Institute, National Asthma Education and Prevention Program. Expert panel report 3: guidelines for the diagnosis and management of asthma. Bethesda, Md.: National Heart, Lung, and Blood Institute; Revised August 2007. NIH publication no. 07-4051. http://www.nhlbi.nih.gov/guidelines/asthma/asthgdln.pdf. Accessed May 10, 2009.
LONG-ACTING BETA2 AGONISTS
Salmeterol (Serevent) and formoterol (Foradil) are bronchodilators that have a duration of action of more than 12 hours. They are very specific for β2-adrenergic receptors and, thus, have low rates of tremor and palpitations or tachycardia.23 Regular use of long-acting beta2 agonists results in only mild tachyphylaxis to the maximal bronchodilator effect and the duration of action of these drugs. However, the bronchoprotective effect of long-acting beta2 agonists (e.g., inhibition of exercise-induced bronchoconstriction) rapidly wanes with regular use.24,25 In general, the effectiveness of short-acting beta2 agonists is not impaired in regular users of long-acting beta2 agonists.26 In some ethnic populations, most notably blacks, there may be genetic variations in the β2-adrenergic receptor that may reduce the effectiveness of long-acting beta2 agonists, although more recent research has called this into question.27–29
There have been concerns about the safety of long-acting beta2 agonists in children and adults, with an increase in severe exacerbations and deaths when added to usual asthma therapy.8 This has prompted the U.S. Food and Drug Administration to review these medications and add a new warning labeling. Available data strongly suggest that long-acting beta2 agonists should never be used as monotherapy for long-term control of persistent asthma7; they should be used only in combination therapy, and increasing the dose of inhaled corticosteroids should be given equal weight to the option of adding a long-acting beta2 agonist if the initial dosage of inhaled corticosteroids is not effective.7,12
CROMOLYN SODIUM AND NEDOCROMIL
Cromolyn sodium and nedocromil stabilize mast cells and interfere with chloride channel function. They are an alternative, but not preferred, medication for the treatment of mild persistent asthma. With newer agents available, their use has markedly decreased.
IMMUNOMODULATORS
Omalizumab (Xolair) is a monoclonal antibody that prevents binding of immunoglobulin E (IgE) to the high-affinity receptors on basophils and mast cells. It has been shown to reduce the need for both oral and inhaled steroids (number needed to treat [NNT] = 6 to 12).30 A reduction in protocol-defined exacerbations was observed in approximately 15 percent of patients (NNT = 6).31 Omalizumab is used as additive therapy for patients 12 years and older with severe persistent asthma who have demonstrated immediate hypersensitivity to inhaled allergens. Anaphylaxis may occur in patients receiving this medication. Usual dosages range from 150 to 375 mg administered subcutaneously every two to four weeks, depending on IgE level and patient weight. The approximate retail price is $738 for a 150-mg vial.32,33 Because of the required monitoring and side effects involved in its use, omalizumab therapy should probably be instituted only in collaboration or consultation with an asthma subspecialist.
METHYLXANTHINES
Sustained-release theophylline is a mild to moderate bronchodilator used as an alternative, although not preferred, adjunctive therapy with inhaled corticosteroids. Theophylline may have mild anti-inflammatory effects. Monitoring of serum theophylline concentration is essential. Theophylline still has a role as add-on therapy in some clinical situations.7
COMBINATION THERAPY
Numerous studies have demonstrated excellent control of moderate persistent asthma with combination therapy in patients 12 years and older.7 Combination therapy has not been studied in children younger than four years. Strong evidence in patients 12 years and older indicates that the combination of inhaled corticosteroids and longacting beta2 agonists leads to clinically meaningful improvements in lung function and symptoms and a reduced need for quickrelief short-acting beta2 agonists. Adding a leukotriene receptor antagonist or theophylline to inhaled corticosteroids or doubling the dose of inhaled corticosteroids also improves outcomes, but the evidence is not as substantial as with the addition of long-acting beta2 agonists.34,35
Step-down strategies in combination therapy may mean changing to a new device with a lower dose of inhaled steroid, which can result in having two combination devices and two co-pays. However, a recent study showed that reducing the frequency of fluticasone/salmeterol therapy to once-daily dosing maintained good control of asthma symptoms.21
Finally, a study published after the EPR-3 guidelines compared continuous therapy with inhaled corticosteroids plus as-needed short-acting beta2 agonists with as-needed use only of inhaled corticosteroids plus short-acting beta2 agonists during exacerbations for patients with mild persistent asthma. The researchers found similar clinical outcomes over a six-month period, with a lower total steroid dose in the as-needed dosing group.36
Fast-Acting Agents
INHALED SHORT-ACTING BETA2 AGONISTS
Short-acting beta2 agonists are the most effective therapy for rapid reversal of airflow obstruction and prompt relief of asthmatic symptoms. Albuterol, levalbuterol (Xopenex), and pirbuterol (Maxair) are the most commonly used short-acting beta2 agonists in the United States. Short-acting beta2 agonists have an onset of action of five minutes or less, peaking within 30 to 60 minutes, and a duration of action of four to six hours.37 Regular use (i.e., four or more times daily) does not affect potency but is associated with a reduction in the duration of action.38,39
EPR-3 recommends using short-acting beta2 agonists only as needed for relief of symptoms or before anticipated exposure to known asthmatic triggers (e.g., animals, exercise). Puffs can be taken in 10- to 15-second intervals; longer intervals offer no benefits.40 Increasing the use of shortacting beta2 agonists or using them more than two days per week for symptom relief (not for prevention of exercise-induced bronchospasm) generally indicates inadequate control of asthma and the need to initiate or intensify anti-inflammatory therapy.7
Tremor, anxiety, heart pounding, and tachycardia (but not hypertension) are common dose-dependent side effects. Some patients are highly sensitive to short-acting beta2 agonists, but most tolerate them well. Beta blockers may diminish the effectiveness of short-acting beta2 agonists but are not contraindicated.41,42
Levalbuterol, the R-enantiomer of albuterol, has an effectiveness and side effect profile indistinguishable from that of the racemic mixture of molecules in albuterol, but is more expensive (one inhaler: $54 for levalbuterol versus $40 to $55 for albuterol). Standard doses for each are two puffs every two to six hours as needed.33,43,44
Metered dose inhalers are the delivery mechanism for all short-acting beta2 agonists. There has been a recent change to all hydrofluoroalkane propellants with the exception of pirbuterol.45 The new inhalers have a different “feel” of the spray, and many patients have reported that they did not think they were getting the full dose of medication. However, the potency of hydrofluoroalkane inhalers is equal to that of chlorofluorocarbon-propelled inhalers.46 Use of spacers is still encouraged.47 It is recommended that physicians observe and regularly review patients' inhaler use, because many if not most patients have difficulty with proper inhalation technique.
In many settings, metered dose inhalers with spacers may be more acceptable and less costly than nebulizer treatment. Bronchodilation by short-acting beta2 agonists delivered with a spacer is comparable to nebulized albuterol when a sufficient number of puffs are administered and inhalation technique is good.48 Most studies showing comparable effects of nebulizers and spacers in emergency department settings used six to 10 puffs from a metered dose inhaler (each puff given sequentially).7 As most health care professionals have recognized, two puffs are not equivalent to a nebulizer treatment.
ORAL SHORT-ACTING BETA2 AGONISTS
Oral short-acting beta2 agonists are less potent, take longer to act, and have more side effects compared with inhaled shortacting beta2 agonists.49 Their use is strongly discouraged. Anticholinergic bronchodilators, such as ipratropium (Atrovent), are not recommended as monotherapy for quick relief of asthmatic symptoms. They have a longer onset of action (20 to 30 minutes) and cause less bronchodilation than inhaled beta2 agonists.50 Anticholinergic agents combined with short-acting beta2 agonists, however, may be beneficial in treating severe asthmatic attacks or those induced by beta blockers.51
Using Asthma Medications Wisely
Clinical trials have had mixed findings on the effect of action plans on clinical asthma outcomes, but the consensus is that education in asthma self-management involving self-monitoring by peak expiratory flow or symptoms, coupled with regular medical review and a written action plan, improves health outcomes for patients with asthma52–54 (Figures 2 and 355 ).
Figure 2. Asthma Action Plan for Children
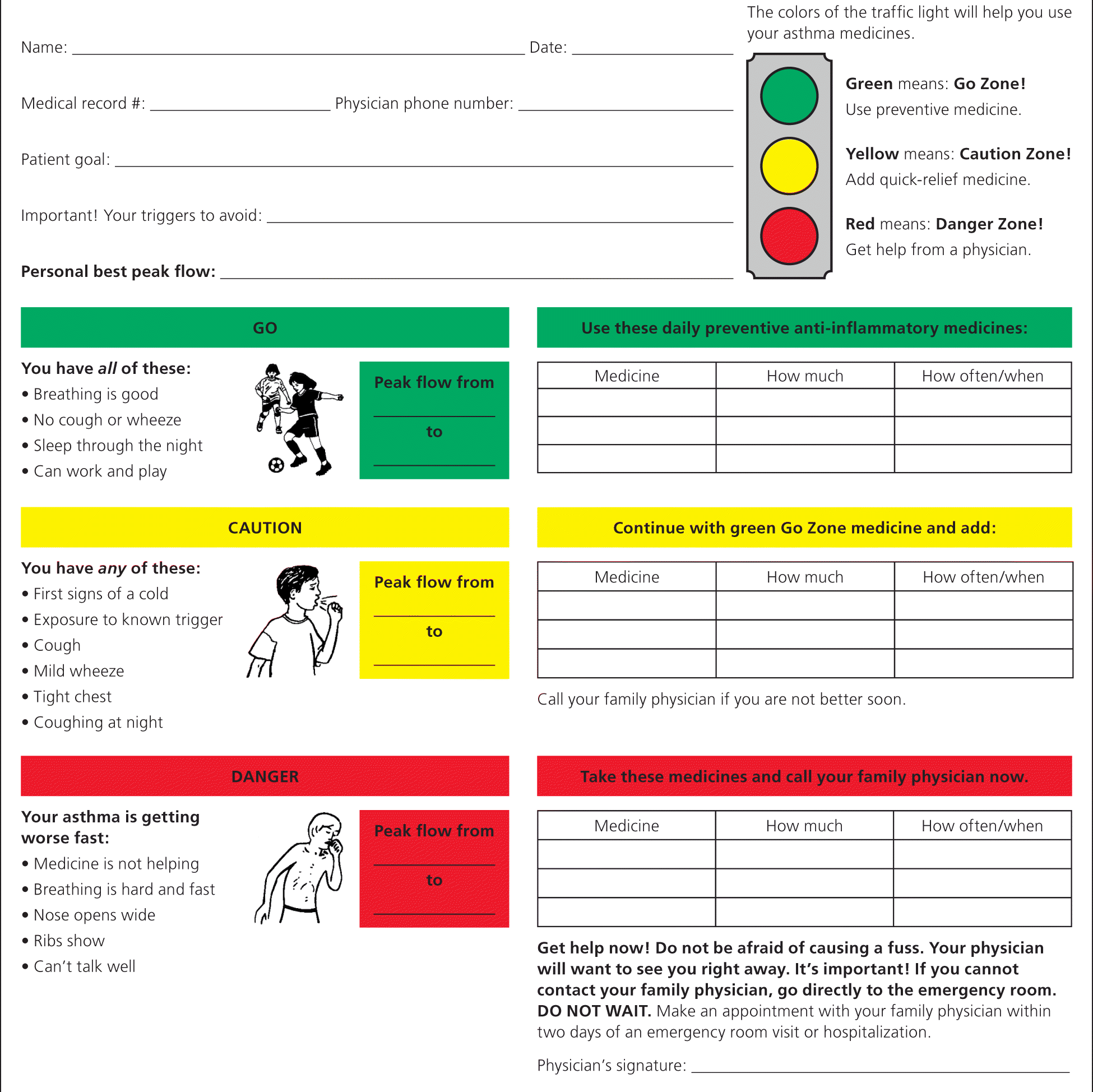
Asthma action plan for children.
Figure 3.
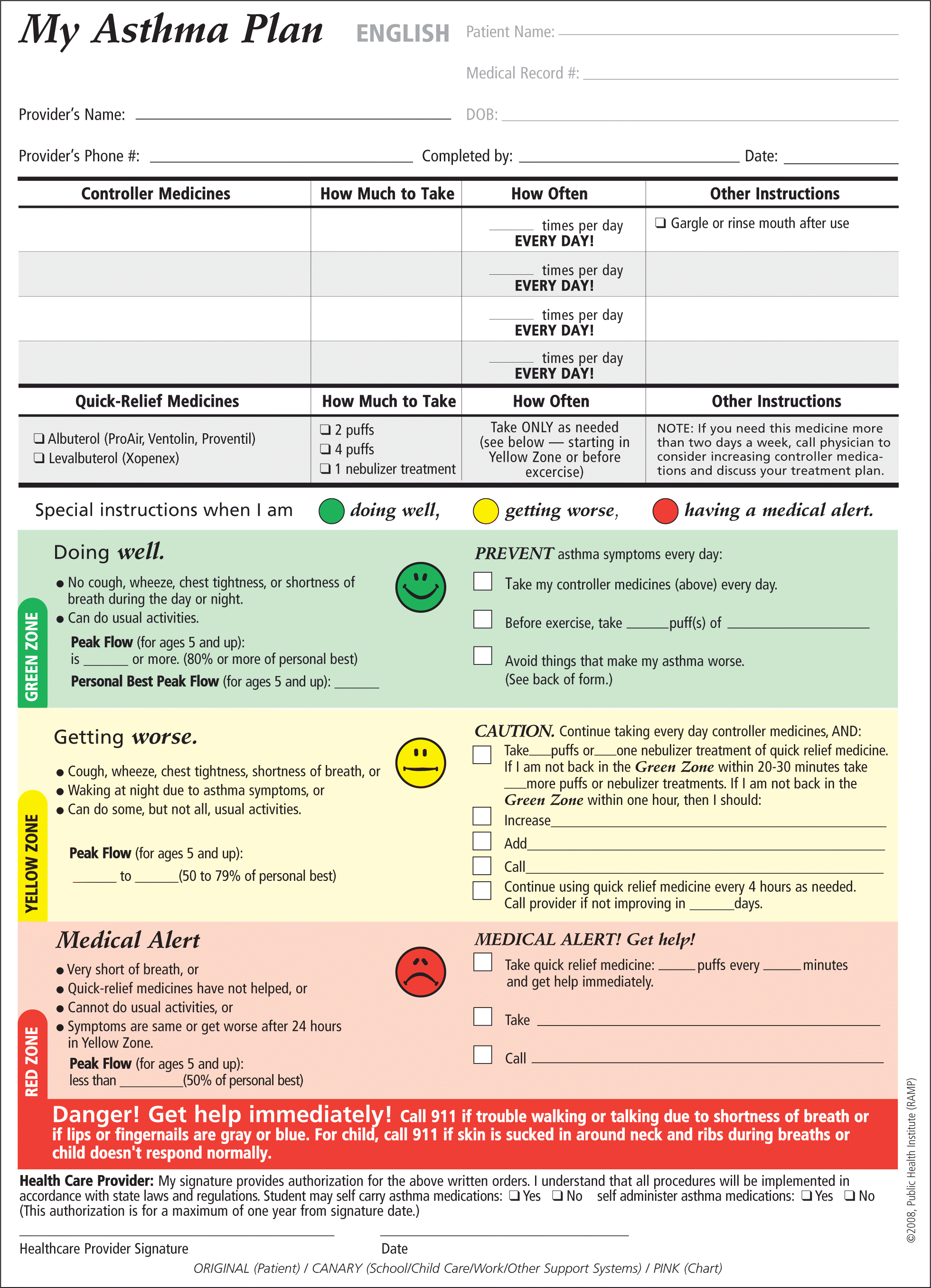
Regional Asthma Management and Prevention (RAMP) Asthma Action Plan.
Reprinted with permission from Regional Asthma Management and Prevention (RAMP), a program of the Public Health Institute. The RAMP Asthma Action Plan was supported by Cooperative Agreement Number 1U58DP001016-01 from the Centers for Disease Control and Prevention (CDC). The contents of the RAMP Asthma Action Plan are solely the responsibility of the authors and do not necessarily represent the official views of the CDC.
For initiation of maintenance care, two approaches are low-dose and high-dose strategies. In either strategy, a critical element is assessing both severity and control as outlined in the EPR-3 (Figure 17 ). Low-dose strategies involve using a low-dose inhaled steroid or leukotriene receptor antagonist with asneeded short-acting beta2 agonists to gradually attain control within a two-week period. This approach is appropriate for patients with mild symptoms and no recent acute events.
A high-dose strategy (treating with higher-dose inhaled steroids or combination agents, usually a step higher than their severity assessment to achieve rapid control) can be used initially and then reduced (stepped down) as control is achieved. Follow-up should be scheduled within two weeks to assess initial response; if low-dose therapy does not provide adequate control, a step up is indicated. High-dose therapy, if effective, should be continued for three months before considering a step down in therapy.
Planned follow-up visits for patients with persistent asthma should occur at least twice yearly, and more often for those with moderate to severe persistent asthma. Some of these visits can be combined with health maintenance care, with others scheduled before patients' most symptomatic seasons. Planned care visits should be used as an opportunity to review use of delivery devices, action plans, and triggers. Care can be stepped down if patients are consistently doing well, based on spirometry and control assessment (e.g., with the Asthma Control Test or Asthma Treatment Assessment Questionnaire).7

