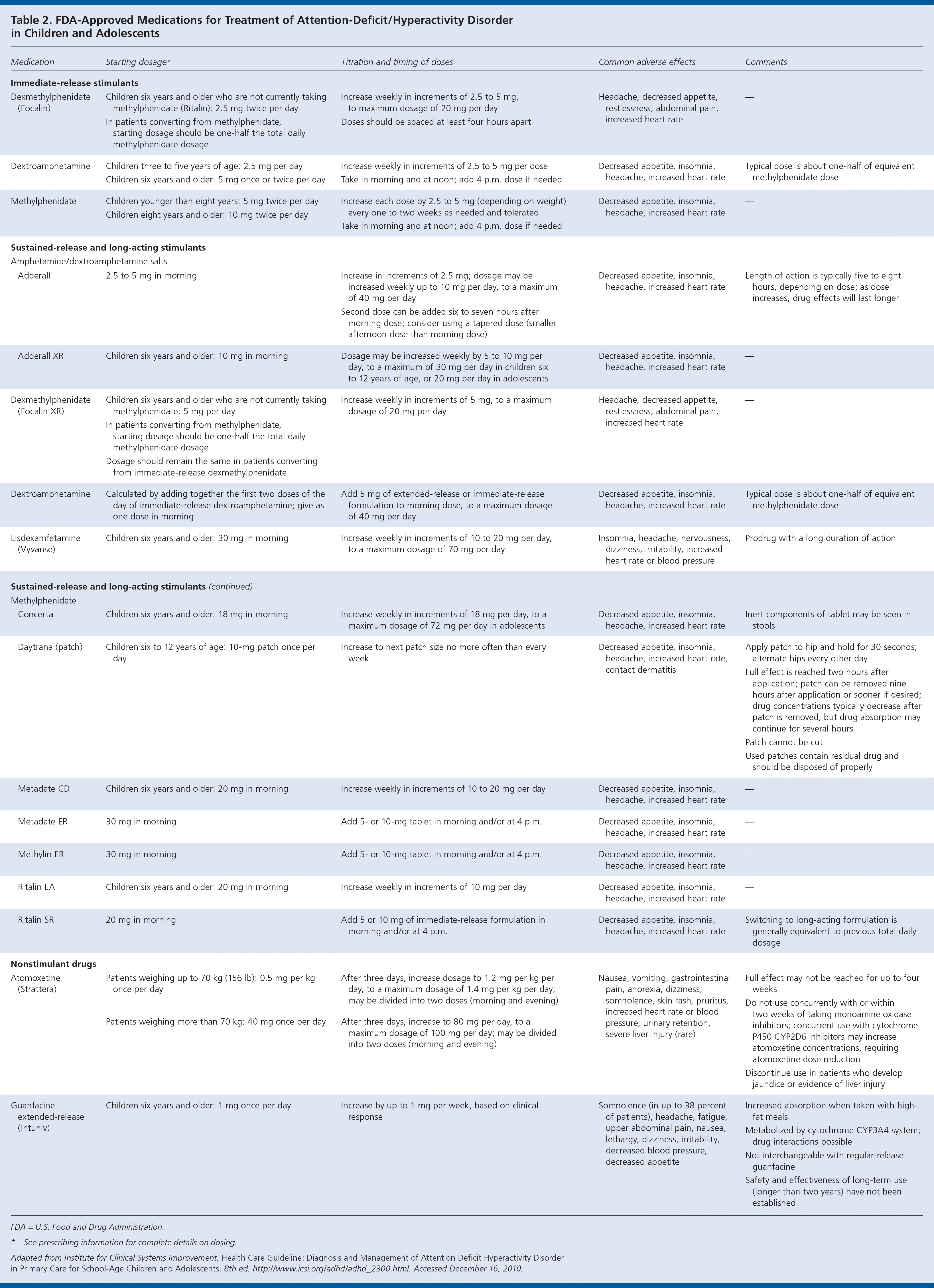
| Immediate-release stimulants |
| Dexmethylphenidate(Focalin) | Children six years and older who are not currently taking methylphenidate (Ritalin): 2.5 mg twice per day | Increase weekly in increments of 2.5 to 5 mg, to maximum dosage of 20 mg per day Doses should be spaced at least four hours apart
| Headache, decreased appetite, restlessness, abdominal pain, increased heart rate | |
| In patients converting from methylphenidate, starting dosage should be one-half the total daily methylphenidate dosage |
| Dextroamphetamine | Children three to five years of age: 2.5 mg per day | | Decreased appetite, insomnia, headache, increased heart rate | |
| Children six years and older: 5 mg once or twice per day |
| Methylphenidate | Children younger than eight years: 5 mg twice per day | | Decreased appetite, insomnia, headache, increased heart rate | |
| Children eight years and older: 10 mg twice per day |
| Sustained-release and long-acting stimulants |
| Amphetamine/dextroamphetamine salts |
| Adderall | 2.5 to 5 mg in morning | Increase in increments of 2.5 mg; dosage may be increased weekly up to 10 mg per day, to a maximum of 40 mg per day Second dose can be added six to seven hours after morning dose; consider using a tapered dose (smaller afternoon dose than morning dose)
| Decreased appetite, insomnia, headache, increased heart rate | |
|
| Adderall XR | Children six years and older: 10 mg in morning | | Decreased appetite, insomnia, headache, increased heart rate | |
| Dexmethylphenidate (Focalin XR) | Children six years and older who are not currently taking methylphenidate: 5 mg per day | | Headache, decreased appetite, restlessness, abdominal pain, increased heart rate | |
| In patients converting from methylphenidate, starting dosage should be one-half the total daily methylphenidate dosage |
| Dosage should remain the same in patients converting from immediate-release dexmethylphenidate |
| Dextroamphetamine | Calculated by adding together the first two doses of the day of immediate-release dextroamphetamine; give as one dose in morning | | Decreased appetite, insomnia, headache, increased heart rate | |
| Lisdexamfetamine (Vyvanse) | Children six years and older: 30 mg in morning | | Insomnia, headache, nervousness, dizziness, irritability, increased heart rate or blood pressure | |
| Methylphenidate |
| Concerta | Children six years and older: 18 mg in morning | | Decreased appetite, insomnia, headache, increased heart rate | |
| Daytrana (patch) | Children six to 12 years of age: 10-mg patch once per day | | Decreased appetite, insomnia, headache, increased heart rate, contact dermatitis | Apply patch to hip and hold for 30 seconds; alternate hips every other day Full effect is reached two hours after application; patch can be removed nine hours after application or sooner if desired; drug concentrations typically decrease after patch is removed, but drug absorption may continue for several hours Patch cannot be cut Used patches contain residual drug and should be disposed of properly
|
|
|
|
| Metadate CD | Children six years and older: 20 mg in morning | | Decreased appetite, insomnia, headache, increased heart rate | |
| Metadate ER | 30 mg in morning | | Decreased appetite, insomnia, headache, increased heart rate | |
| Methylin ER | 30 mg in morning | | Decreased appetite, insomnia, headache, increased heart rate | |
| Ritalin LA | Children six years and older: 20 mg in morning | | Decreased appetite, insomnia, headache, increased heart rate | |
| Ritalin SR | 20 mg in morning | | Decreased appetite, insomnia, headache, increased heart rate | |
| Nonstimulant drugs |
| Atomoxetine (Strattera) | Patients weighing up to 70 kg (156 lb): 0.5 mg per kg once per day | | Nausea, vomiting, gastrointestinal pain, anorexia, dizziness, somnolence, skin rash, pruritus, increased heart rate or blood pressure, urinary retention, severe liver injury (rare) | Full effect may not be reached for up to four weeks Do not use concurrently with or within two weeks of taking monoamine oxidase inhibitors; concurrent use with cytochrome P450 CYP2D6 inhibitors may increase atomoxetine concentrations, requiring atomoxetine dose reduction Discontinue use in patients who develop jaundice or evidence of liver injury
|
| Patients weighing more than 70 kg: 40 mg once per day | |
| Guanfacine extended-release (Intuniv) | Children six years and older: 1 mg once per day | | Somnolence (in up to 38 percent of patients), headache, fatigue, upper abdominal pain, nausea, lethargy, dizziness, irritability, decreased blood pressure, decreased appetite | Increased absorption when taken with high-fat meals Metabolized by cytochrome CYP3A4 system; drug interactions possible Not interchangeable with regular-release guanfacine Safety and effectiveness of long-term use(longer than two years) have not been established
|