Guideline source: American Academy of Child and Adolescent Psychiatry
Evidence rating system used? Yes
Literature search described? Yes
Guideline developed by participants without relevant financial ties to industry? No
Published source: Journal of the American Academy of Child and Adolescent Psychiatry, January 2012
Obsessive-compulsive disorder (OCD) is common in children, with a prevalence of approximately 1 to 2 percent in the United States, and causes significant disability. However, it has been called the “hidden epidemic” because of the secretive and variable nature of its symptoms. A British survey of five- to 15-year-olds found that approximately 90 percent of children and adolescents with OCD had not been diagnosed with the condition. The American Academy of Child and Adolescent Psychiatry (AACAP) recently published updated recommendations on the assessment and treatment of OCD in children, incorporating new research and empiric evidence to guide physicians who treat children with OCD.
Screening and Assessment
Physicians performing psychiatric assessment in children and adolescents should routinely screen for obsessions and compulsions or repetitive behaviors. Symptoms of OCD may be of mild to moderate severity, wax and wane over time, be prominent in one setting but not another, and be hidden from others. The commonly used parent-report Child Behavior Checklist includes eight items that have been shown to have good sensitivity and specificity as a screening tool for OCD in children.
If screening suggests that the patient has obsessive-compulsive symptoms, he or she should be fully evaluated using criteria from the Diagnostic and Statistical Manual of Mental Disorders, 4th ed., text revision. The diagnostic criteria of time occupied by symptoms, the level of subjective distress, and functional impairment are best captured by a reliable instrument such as the Children's Yale-Brown Obsessive-Compulsive Scale, a 10-item clinician-administered instrument that is most informative when given to children and their parents.
A complete psychiatric evaluation should also be performed, including a standard patient history and mental state examination, to detect any comorbid psychiatric disorders, which are common in children with OCD. The identification of major depressive disorder and bipolar disorder is especially important before the initiation of a selective serotonin reuptake inhibitor (SSRI). Comorbid eating disorders are uncommon in preadolescent children with OCD, but become more prevalent during adolescence. In children with these conditions, medical considerations outweigh other concerns (except suicidality) and must be stabilized to permit mental health interventions. Some self-injury disorders (e.g., trichotillomania, compulsive nail biting, skin picking) share some features of OCD, but also have important differences. Although stress may exacerbate these behaviors, they usually are not preceded by obsessions, but rather a sense of general or localized tension. The behaviors can be a source of temporary gratification, but may be followed by remorse and shame. Behavioral therapy is the primary treatment for these disorders; SSRIs are often less effective.
HISTORY
A full family, school, and medical history should be obtained in children with positive screens for OCD. Parental efforts to relieve a child's anxiety may inadvertently lead to reinforcement of obsessive-compulsive behaviors; therefore, the role of individual family members in the maintenance and management of these symptoms should be assessed.
School histories provide an important measurement of function and illness severity. Obsessive-compulsive symptoms that are apparent in the school setting imply more anxiety, stronger compulsions, less insight, and less resistance and control. Therefore, problems such as falling grades, the need for extra help, or special class placement indicate more urgency for treatment and may justify more aggressive interventions, including medications.
The medical history should focus on the central nervous system, with particular attention to trauma and neurologic symptoms. Group A streptococcal infection has recently been suggested as a potential precipitant for OCD associated with PANDAS (pediatric autoimmune neuropsychiatric disorders associated with streptococcal infections). When treating preadolescent patients or children in remission who suddenly relapse, physicians should determine whether the patient has a history of such infection. Neurologic signs such as chorea can be evidence of rheumatic fever, but may not occur for several months after infection. “Soft” neurologic signs (e.g., tremor, coordination difficulties) are one criterion of PANDAS.
Treatment
Cognitive behavioral therapy (CBT) is the first-line treatment for mild to moderate OCD in children. However, a survey of clinicians who treat children with OCD found that only one-third regularly use exposure techniques. The goals of cognitive therapy typically include an increased sense of self-efficacy, predictability, and control. Most CBT programs use positive reinforcement for exposure and response prevention; punishment is not helpful in the treatment of OCD. Multimodal treatment is recommended if CBT alone does not result in a clinical response after several months.
In children with moderate to severe OCD, medication is indicated in addition to CBT. Any situation that could impede delivery of CBT is also an indication for medical treatment. Concurrent psychopathology, including multiple anxiety disorders, major mood disturbance, and disruptive behavioral disorders, may decrease the acceptance of and adherence to CBT, and may require medication. Individual and family factors also are important considerations.
SSRIs are the first-line medications for treatment of OCD in children. However, no comparative treatment studies have been performed, and there is little evidence to guide physicians in their choice of SSRIs. Titration should be conservative, with modest dose increases approximately every three weeks (Table 1). Treatment is generally continued for six to 12 months after stabilization, then gradually withdrawn over several months. CBT “booster” sessions may be helpful to address symptom recurrences during or after medication discontinuation. Long-term treatment should be considered in patients with at least two relapses of at least moderate severity.
Table 1. Dosing Guidelines for Antidepressants in Children and Adolescents with Obsessive-Compulsive Disorder
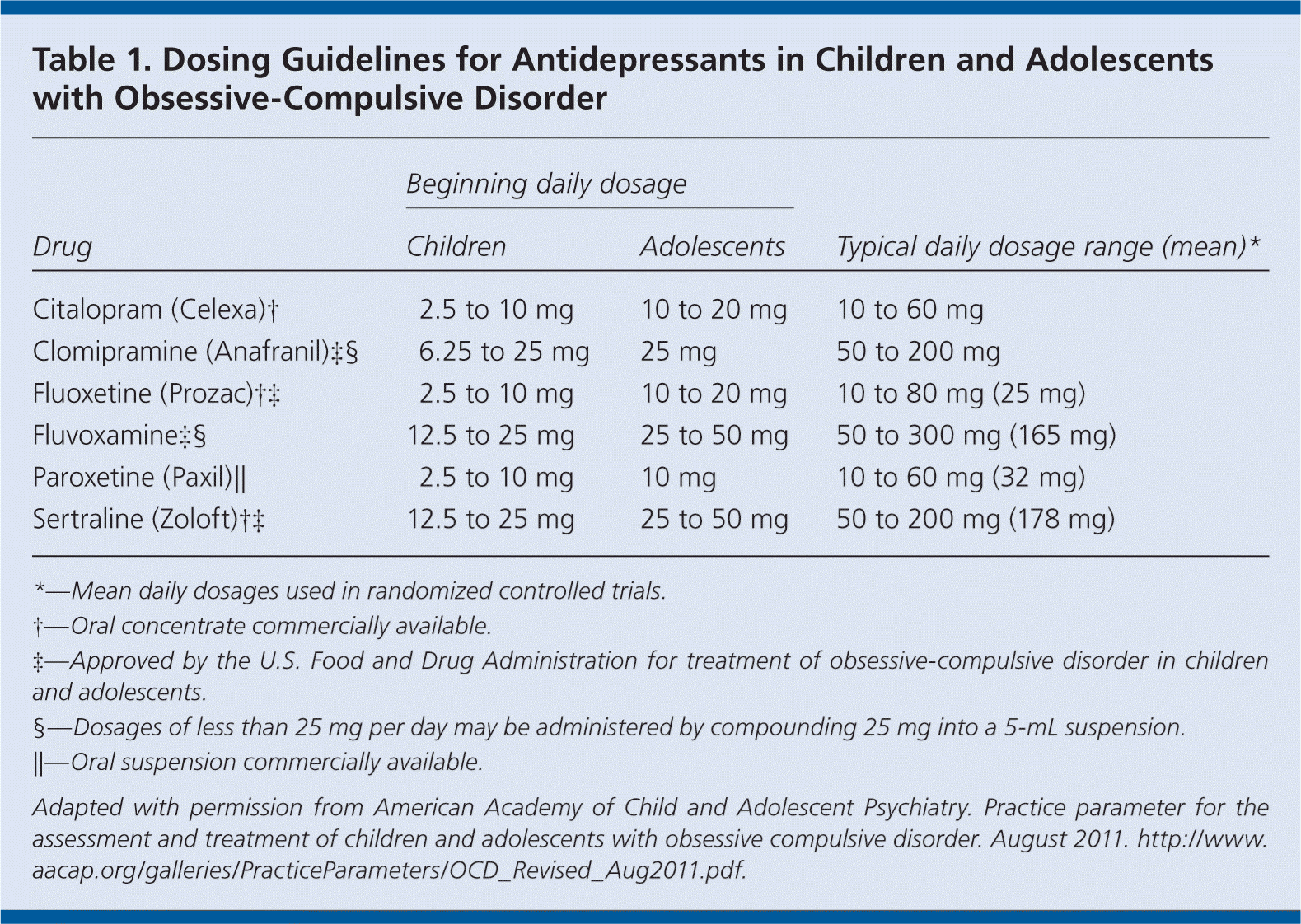
| Drug | Beginning daily dosage | Typical daily dosage range (mean)* | |
|---|---|---|---|
| Children | Adolescents | ||
| Citalopram (Celexa)† | 2.5 to 10 mg | 10 to 20 mg | 10 to 60 mg |
| Clomipramine (Anafranil)ठ| 6.25 to 25 mg | 25 mg | 50 to 200 mg |
| Fluoxetine (Prozac)†‡ | 2.5 to 10 mg | 10 to 20 mg | 10 to 80 mg (25 mg) |
| Fluvoxamineठ| 12.5 to 25 mg | 25 to 50 mg | 50 to 300 mg (165 mg) |
| Paroxetine (Paxil)∥ | 2.5 to 10 mg | 10 mg | 10 to 60 mg (32 mg) |
| Sertraline (Zoloft)†‡ | 12.5 to 25 mg | 25 to 50 mg | 50 to 200 mg (178 mg) |
*—Mean daily dosages used in randomized controlled trials.
†—Oral concentrate commercially available.
‡—Approved by the U.S. Food and Drug Administration for treatment of obsessive-compulsive disorder in children and adolescents.
§—Dosages of less than 25 mg per day may be administered by compounding 25 mg into a 5-mL suspension.
∥—Oral suspension commercially available.
Adapted with permission from American Academy of Child and Adolescent Psychiatry. Practice parameter for the assessment and treatment of children and adolescents with obsessive compulsive disorder. August 2011. http://www.aacap.org/galleries/PracticeParameters/OCD_Revised_Aug2011.pdf.
Behavioral adverse effects of SSRI treatment are more likely in younger children. Peripubertal children exposed to antidepressants are at higher risk of conversion to mania compared with adolescents and young adults. The goal is to find a therapeutic window that provides an adequate clinical response but acceptable degrees of behavioral activation. If this goal is not achievable, switching to another SSRI is indicated. All antidepressants have boxed warnings about the risk of suicide; however, no suicides have occurred in any randomized controlled trials of SSRI use in children.
A cardiac evaluation should be performed in children who receive the tricyclic antidepressant clomipramine (Anafranil), which was the first agent approved for use in children with OCD. The baseline evaluation should include a systems review and determination of personal or family history of heart disease. A history of nonfebrile seizures should be noted, but is not an absolute contraindication. Baseline electrocardiography should also be obtained. Contraindications for clomipramine use include a PR interval longer than 200 milliseconds, a QRS interval more than 30 percent above baseline or longer than 120 milliseconds, a prolonged corrected QT interval (greater than 450 milliseconds), blood pressure greater than 140 mm Hg (systolic) or 90 mm Hg (diastolic), or resting heart rate greater than 130 beats per minute.
The choice of treatment should be guided by empirical evidence on the moderators and predictors of treatment response. Psychiatric comorbidity may have a significant influence on treatment response. In one trial of children and adolescents with OCD who were treated with an SSRI, those with comorbid attention-deficit/hyperactivity disorder, tic disorder, or oppositional defiant disorder had significantly lower response rates than patients with OCD only. Comorbidity is also associated with a higher rate of relapse after treatment. Children with comorbid tics should be assigned to CBT or a comblination of CBT and medication as a first option.
Medication augmentation strategies should be reserved for patients with treatment-resistant OCD and moderate impairment in at least one important domain of function despite adequate monotherapy. The addition of clomipramine to an SSRI may be helpful to combine the serotonergic effects of each medication while minimizing adverse events. Fluvoxamine has the most synergistic effect when added to clomipramine. Low-dose augmentation (25 to 75 mg per day) may be useful, but care must be taken when combining clomipramine with fluvoxamine and cytochrome P450 2D6 inhibitors such as fluoxetine (Prozac) or paroxetine (Paxil).
The most common drug augmentation strategies have used atypical neuroleptics. Some children with treatment-resistant OCD may benefit from judicious augmentation with neuroleptics, particularly those with tic disorders, poor insight, pervasive developmental disorder symptoms, and mood instability. At least 12 weeks of SSRI treatment may be required before neuroleptic augmentation is effective.

