Guideline source: Advisory Committee on Immunization Practices
Evidence rating system used? No
Literature search described? Yes
Guideline developed by participants without relevant financial ties to industry? Not reported
Published source:Morbidity and Mortality Weekly Report, August 17, 2012
Endorsed by the AAFP, August 2012. https://www.aafp.org/patient-care/clinical-recommendations/all/endorsed.html
The Advisory Committee on Immunization Practices (ACIP) of the Centers for Disease Control and Prevention (CDC) has issued its annual guideline on influenza vaccination. This year’s guideline provides information on dosing considerations for children six months to eight years of age, febrile seizures associated with trivalent inactivated vaccine (TIV) and the 13-valent pneumococcal conjugate vaccine (PCV13), and vaccination of persons allergic to eggs.
The 2012-2013 seasonal influenza vaccine contains A/California/7/2009 (H1N1)-like, A/Victoria/361/2011 (H3N2)-like, and B/Wisconsin/1/2010-like (Yamagata lineage) antigens, which differ from the 2011-2012 vaccine. The influenza A (H1N1) vaccine virus strain is derived from an influenza A (H1N1)pdm09 (2009[H1N1]) virus and was included in the 2009(H1N1) monovalent pandemic vaccine, as well as the 2010-2011 and 2011-2012 seasonal vaccines.
To ensure full production of protective antibody levels before influenza activity begins, health care professionals should provide vaccination as soon as the vaccine is available. Annual vaccination is recommended for all persons six months and older. Table 1 lists the vaccines available for the 2012-2013 influenza season.
Table 1. Vaccines for 2012-2013 Influenza Season
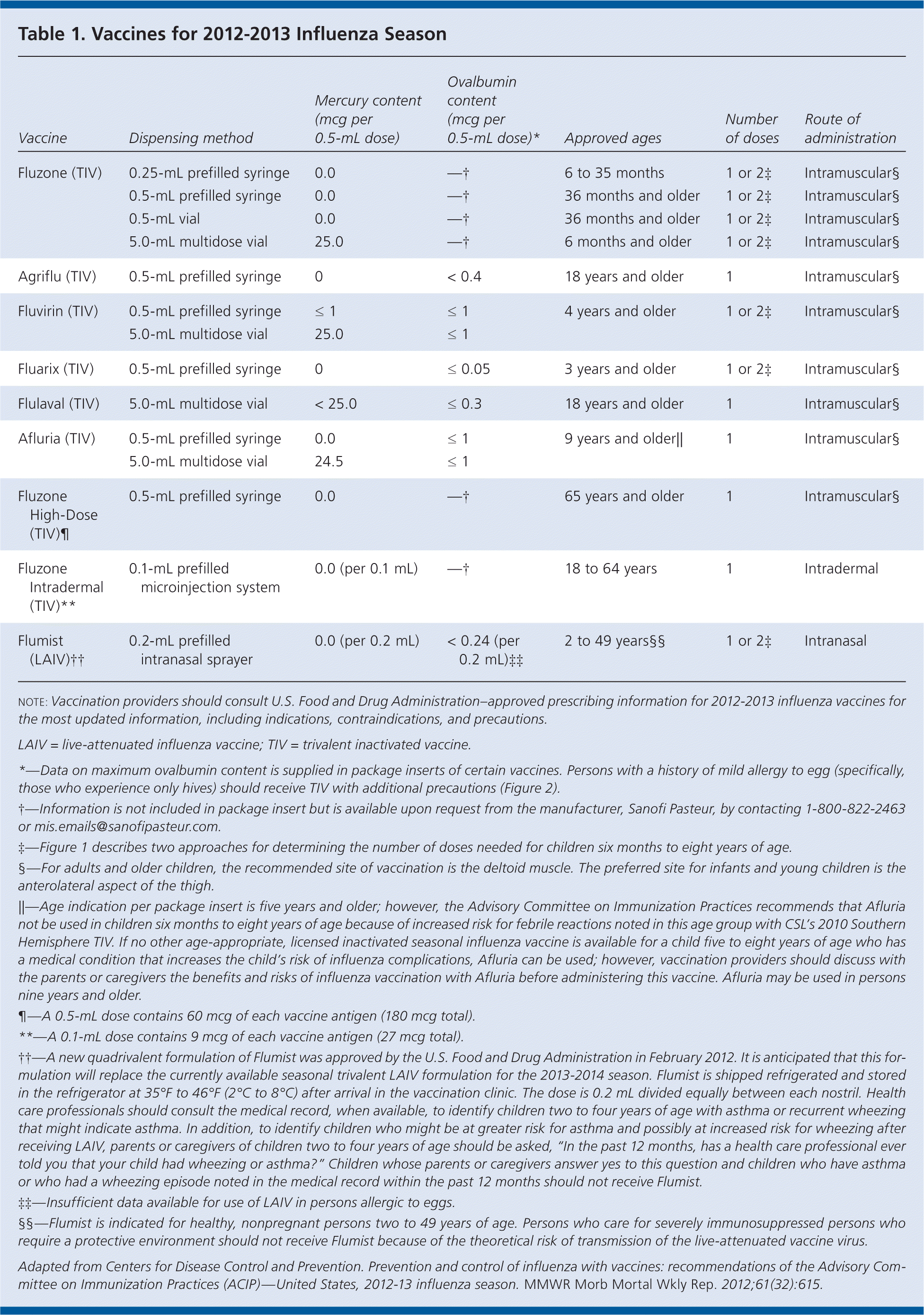
| Vaccine | Dispensing method | Mercury content (mcg per 0.5-mL dose) | Ovalbumin content (mcg per 0.5-mL dose)* | Approved ages | Number of doses | Route of administration |
|---|---|---|---|---|---|---|
| Fluzone (TIV) | 0.25-mL prefilled syringe | 0.0 | —† | 6 to 35 months | 1 or 2‡ | Intramuscular§ |
| 0.5-mL prefilled syringe | 0.0 | —† | 36 months and older | 1 or 2‡ | Intramuscular§ | |
| 0.5-mL vial | 0.0 | —† | 36 months and older | 1 or 2‡ | Intramuscular§ | |
| 5.0-mL multidose vial | 25.0 | —† | 6 months and older | 1 or 2‡ | Intramuscular§ | |
| Agriflu (TIV) | 0.5-mL prefilled syringe | 0 | < 0.4 | 18 years and older | 1 | Intramuscular§ |
| Fluvirin (TIV) | 0.5-mL prefilled syringe | ≤ 1 | ≤ 1 | 4 years and older | 1 or 2‡ | Intramuscular§ |
| 5.0-mL multidose vial | 25.0 | ≤ 1 | ||||
| Fluarix (TIV) | 0.5-mL prefilled syringe | 0 | ≤ 0.05 | 3 years and older | 1 or 2‡ | Intramuscular§ |
| Flulaval (TIV) | 5.0-mL multidose vial | < 25.0 | ≤ 0.3 | 18 years and older | 1 | Intramuscular§ |
| Afluria (TIV) | 0.5-mL prefilled syringe | 0.0 | ≤ 1 | 9 years and older|| | 1 | Intramuscular§ |
| 5.0-mL multidose vial | 24.5 | ≤ 1 | ||||
| Fluzone High-Dose (TIV)¶ | 0.5-mL prefilled syringe | 0.0 | —† | 65 years and older | 1 | Intramuscular§ |
| Fluzone Intradermal (TIV)** | 0.1-mL prefilled microinjection system | 0.0 (per 0.1 mL) | —† | 18 to 64 years | 1 | Intradermal |
| Flumist (LAIV)†† | 0.2-mL prefilled intranasal sprayer | 0.0 (per 0.2 mL) | < 0.24 (per 0.2 mL)‡‡ | 2 to 49 years§§ | 1 or 2‡ | Intranasal |
note: Vaccination providers should consult U.S. Food and Drug Administration–approved prescribing information for 2012-2013 influenza vaccines for the most updated information, including indications, contraindications, and precautions.
LAIV = live-attenuated influenza vaccine; TIV = trivalent inactivated vaccine.
*—Data on maximum ovalbumin content is supplied in package inserts of certain vaccines. Persons with a history of mild allergy to egg (specifically, those who experience only hives) should receive TIV with additional precautions (Figure 2).
†—Information is not included in package insert but is available upon request from the manufacturer, Sanofi Pasteur, by contacting 1-800-822-2463 or mis.emails@sanofipasteur.com.
‡—Figure 1 describes two approaches for determining the number of doses needed for children six months to eight years of age.
§—For adults and older children, the recommended site of vaccination is the deltoid muscle. The preferred site for infants and young children is the anterolateral aspect of the thigh.
||—Age indication per package insert is five years and older; however, the Advisory Committee on Immunization Practices recommends that Afluria not be used in children six months to eight years of age because of increased risk for febrile reactions noted in this age group with CSL’s 2010 Southern Hemisphere TIV. If no other age-appropriate, licensed inactivated seasonal influenza vaccine is available for a child five to eight years of age who has a medical condition that increases the child’s risk of influenza complications, Afluria can be used; however, vaccination providers should discuss with the parents or caregivers the benefits and risks of influenza vaccination with Afluria before administering this vaccine. Afluria may be used in persons nine years and older.
¶—A 0.5-mL dose contains 60 mcg of each vaccine antigen (180 mcg total).
**—A 0.1-mL dose contains 9 mcg of each vaccine antigen (27 mcg total).
††—A new quadrivalent formulation of Flumist was approved by the U.S. Food and Drug Administration in February 2012. It is anticipated that this formulation will replace the currently available seasonal trivalent LAIV formulation for the 2013-2014 season. Flumist is shipped refrigerated and stored in the refrigerator at 35°F to 46°F (2°C to 8°C) after arrival in the vaccination clinic. The dose is 0.2 mL divided equally between each nostril. Health care professionals should consult the medical record, when available, to identify children two to four years of age with asthma or recurrent wheezing that might indicate asthma. In addition, to identify children who might be at greater risk for asthma and possibly at increased risk for wheezing after receiving LAIV, parents or caregivers of children two to four years of age should be asked, “In the past 12 months, has a health care professional ever told you that your child had wheezing or asthma?” Children whose parents or caregivers answer yes to this question and children who have asthma or who had a wheezing episode noted in the medical record within the past 12 months should not receive Flumist.
‡‡—Insufficient data available for use of LAIV in persons allergic to eggs.
§§—Flumist is indicated for healthy, nonpregnant persons two to 49 years of age. Persons who care for severely immunosuppressed persons who require a protective environment should not receive Flumist because of the theoretical risk of transmission of the live-attenuated vaccine virus.
Adapted from Centers for Disease Control and Prevention. Prevention and control of influenza with vaccines: recommendations of the Advisory Committee on Immunization Practices (ACIP)—United States, 2012-13 influenza season. MMWR Morb Mortal Wkly Rep. 2012;61(32):615.
Updates
VACCINATION IN CHILDREN SIX MONTHS TO EIGHT YEARS OF AGE
Children six months to eight years of age need two doses of vaccine, administered at least four weeks apart, during their first season for vaccination. For children who last received seasonal influenza vaccine before the 2010-2011 season but did not receive a vaccine containing 2009(H1N1) antigen (i.e., seasonal vaccine since July 2010 or monovalent 2009[H1N1] vaccine), it is recommended that they receive two doses this season, even if they received two doses of seasonal vaccine before the 2010-2011 influenza season (Figure 1). Children in this age group need only one dose of vaccine this year if they received two or more doses of seasonal vaccine since July 1, 2010; two or more doses of seasonal influenza vaccine before July 1, 2010, and one or more doses of monovalent 2009(H1N1) vaccine; or one or more doses of seasonal influenza vaccine before July 1, 2010, and one or more doses of seasonal influenza vaccine since July 1, 2010.
Figure 1. Influenza Vaccination Dosing in Children Six Months to Eight Years of Age
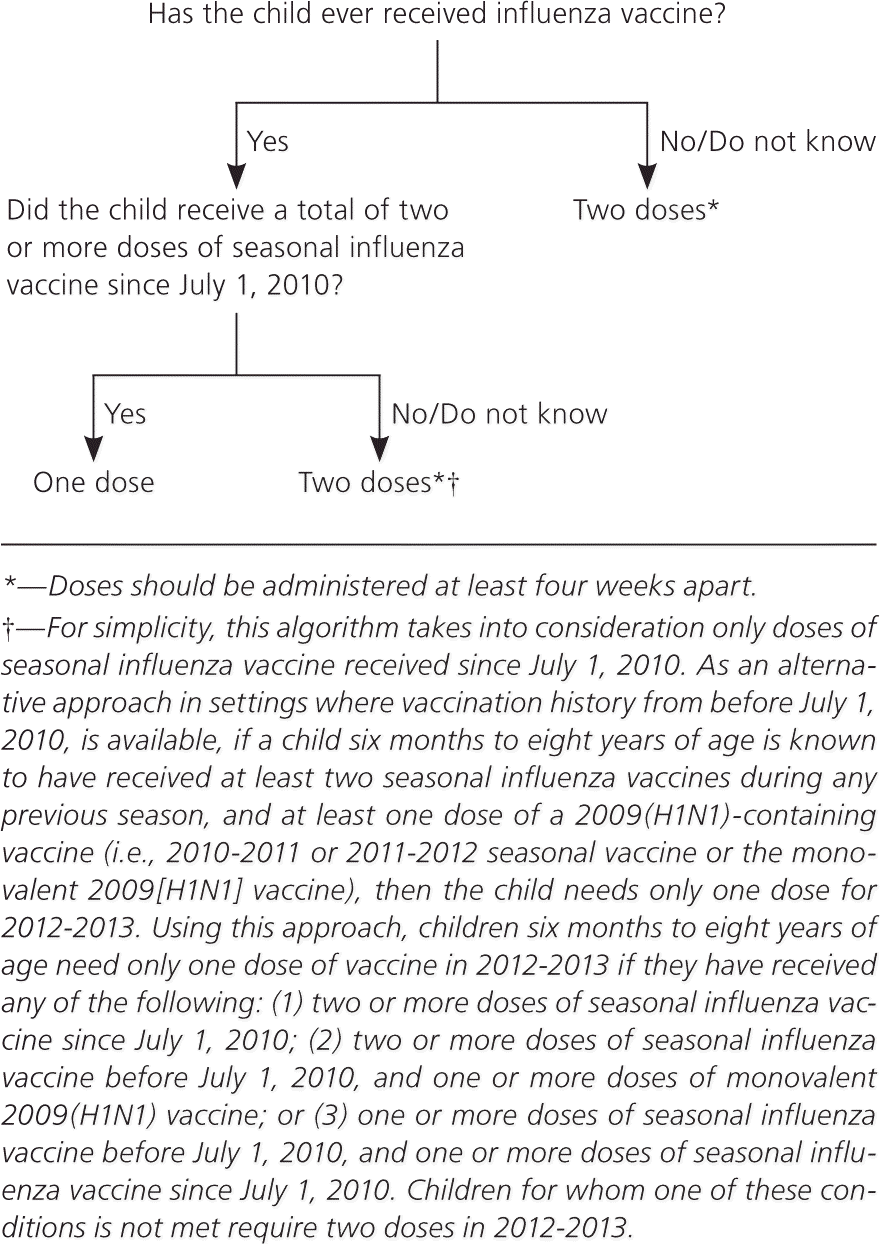
Algorithm for influenza vaccine dosing in children six months to eight years of age.
Adapted from Centers for Disease Control and Prevention. Prevention and control of influenza with vaccines: recommendations of the Advisory Committee on Immunization Practices (ACIP)—United States, 2012-13 influenza season. MMWR Morb Mortal Wkly Rep. 2012;61(32):614.
FEBRILE SEIZURES ASSOCIATED WITH TIV AND PCV13
After a report of increased risk of febrile seizure after influenza vaccination in Australia, the CDC and U.S. Food and Drug Administration enhanced monitoring during the 2010-2011 influenza season. In response to the findings in Australia, ACIP does not recommend Afluria for children younger than nine years. Increased risk for febrile seizure was noted in children six months to four years of age on the day of vaccination to the day after. Risk peaked at 16 months of age. The risk was higher when the seasonal influenza and PCV13 vaccines were administered on the same day. No increased risk was observed in children five years and older after vaccination with TIV or in children of any age who received live-attenuated influenza vaccine. Increased risk of febrile seizures in young children in the United States was less than one per 1,000 children vaccinated. No changes in the use of TIV or PCV13 are recommended for the 2012-2013 influenza season.
VACCINATION IN PATIENTS WITH EGG ALLERGY
For patients with a history of egg allergy who have experienced only hives, ACIP recommends vaccination with TIV rather than live-attenuated influenza vaccine, that the vaccine be administered by a health care professional proficient in potential manifestations of egg allergy, and observation for at least 30 minutes after vaccination (Figure 2). Persons who have had reactions to egg proteins that include angioedema, respiratory distress, lightheadedness, or recurrent emesis, or who required epinephrine or other emergency medical intervention, are more likely to have a systemic or anaphylactic reaction to the vaccination. These patients should be referred to a physician with expertise in management of allergic reactions for further risk assessment. A previous severe reaction to the influenza vaccine is a contraindication to future vaccination.
Figure 2. Influenza Vaccination in Patients with Egg Allergy
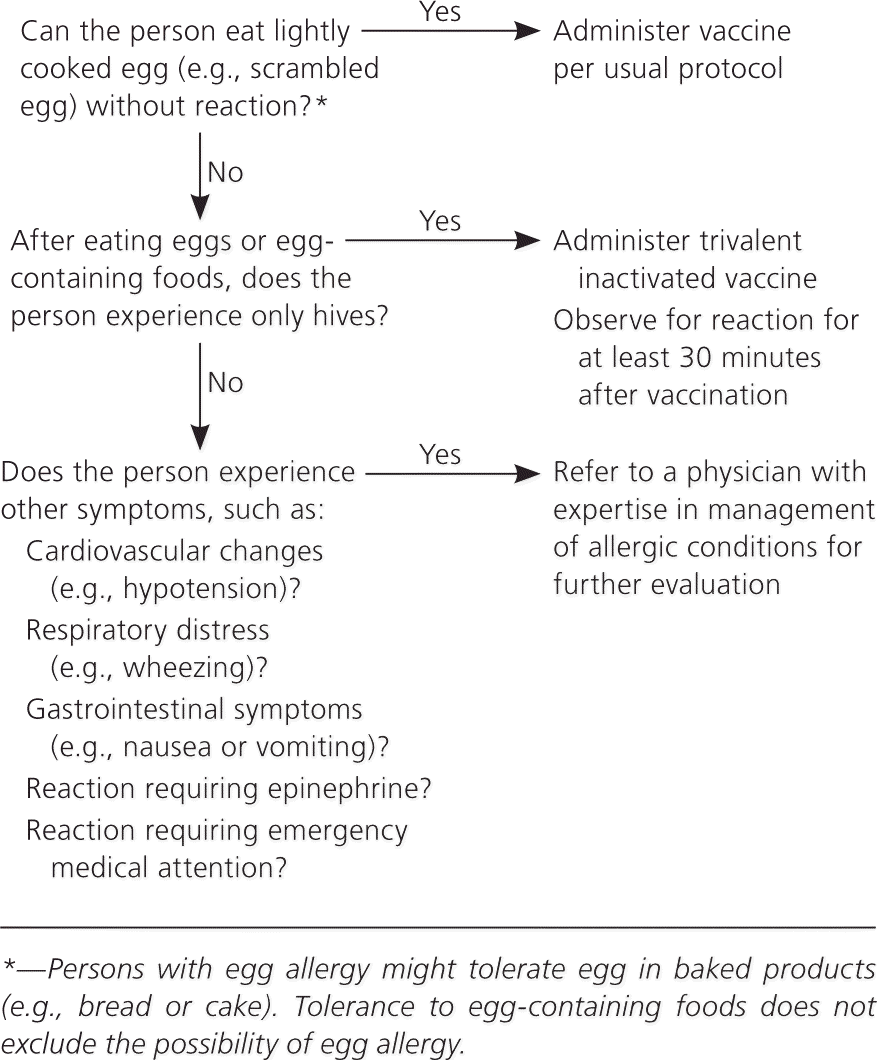
Algorithm for influenza vaccination in persons who report egg allergy.
Adapted from Centers for Disease Control and Prevention. Prevention and control of influenza with vaccines: recommendations of the Advisory Committee on Immunization Practices (ACIP)—United States, 2012-13 influenza season. MMWR Morb Mortal Wkly Rep. 2012;61(32):617.

