Most cases of urinary incontinence in women fall under one of three major subtypes: urge, stress, or mixed. A stepped-care approach that advances from least invasive (behavioral modification) to more invasive (surgery) interventions is recommended. Bladder retraining and pelvic floor muscle exercises are first-line treatments for persons without cognitive impairment who present with urge incontinence. Neuromodulation devices, such as posterior tibial nerve stimulators, are an option for urge incontinence that does not respond to behavioral therapy. Pharmacologic therapy with anticholinergic medications is another option for treating urge incontinence if behavioral therapy is unsuccessful; however, because of adverse effects, these agents are not recommended in older adults. Other medication options for urge incontinence include mirabegron and onabotulinumtoxinA. Sacral nerve stimulators, which are surgically implanted, have also been shown to improve symptoms of urge incontinence. Pelvic floor muscle exercises are considered first-line treatment for stress incontinence. Noninvasive electrical and magnetic stimulation devices are also available. Alternatives for treating stress incontinence include vaginal inserts, such as pessaries, and urethral plugs. Limited or conflicting evidence exists for the use of medications for stress incontinence; no medications are approved by the U.S. Food and Drug Administration for this condition. Minimally invasive procedures, including radiofrequency denaturation of the urethra and injection of periurethral bulking agents, can be used if stress incontinence does not respond to less invasive treatments. Surgical interventions, such as sling and urethropexy procedures, should be reserved for stress incontinence that has not responded to other treatments.
Urinary incontinence, defined as the involuntary leakage of urine, affects 20 million persons nationwide. 1 Estimations of prevalence range from 3 to 55 percent, depending on the definition and the population.2 Within nursing homes, 60 to 70 percent of patients experience the disorder.3 These estimates are thought to be conservative, because at least one-half of patients do not report incontinence to a physician.4
SORT: KEY RECOMMENDATIONS FOR PRACTICE
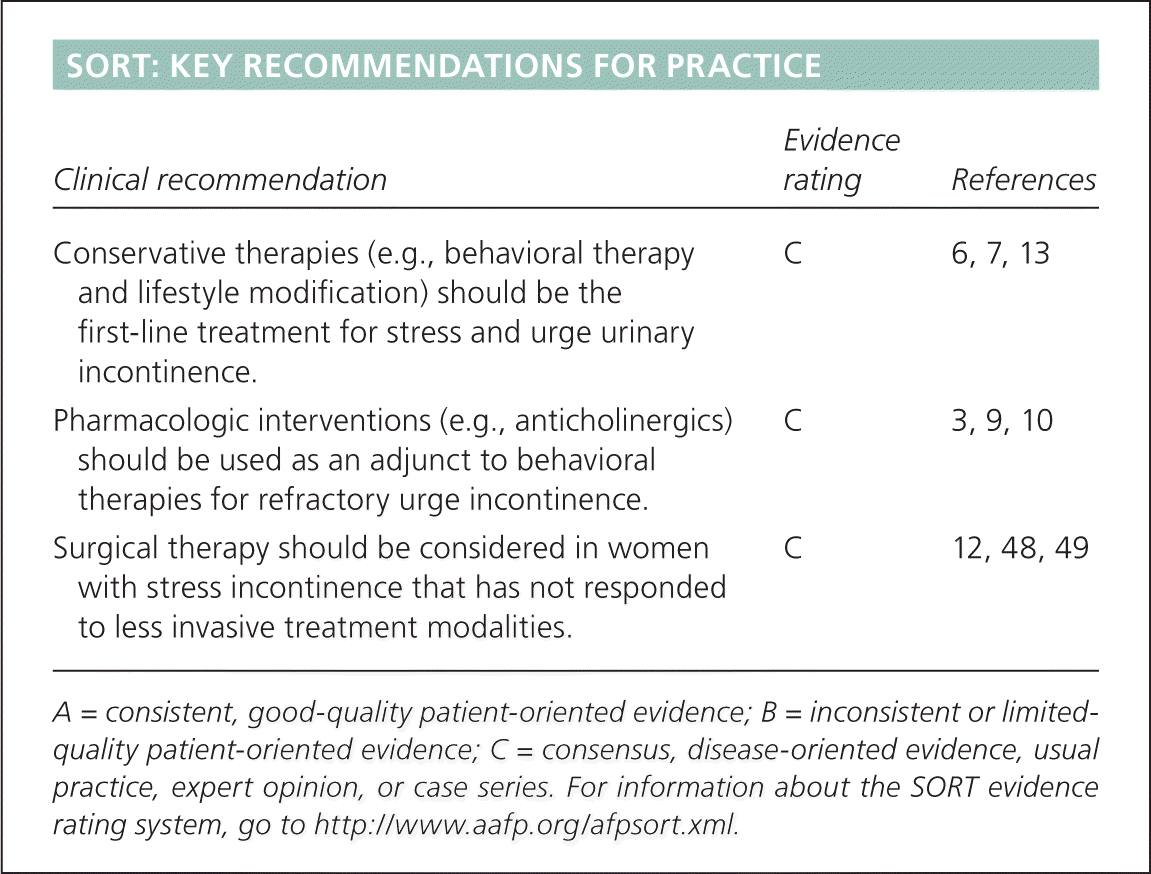
| Clinical recommendation | Evidence rating | References |
|---|---|---|
| Conservative therapies (e.g., behavioral therapy and lifestyle modification) should be the first-line treatment for stress and urge urinary incontinence. | C | 6, 7, 13 |
| Pharmacologic interventions (e.g., anticholinergics) should be used as an adjunct to behavioral therapies for refractory urge incontinence. | C | 3, 9, 10 |
| Surgical therapy should be considered in women with stress incontinence that has not responded to less invasive treatment modalities. | C | 12, 48, 49 |
A = consistent, good-quality patient-oriented evidence; B = inconsistent or limited-quality patient-oriented evidence; C = consensus, disease-oriented evidence, usual practice, expert opinion, or case series. For information about the SORT evidence rating system, go to https://www.aafp.org/afpsort.xml.
The diagnosis of urinary incontinence was reviewed in a recent issue of AFP.5 This article focuses on treatment for the most common types of incontinence in women: urge, stress, and mixed (Table 16,7). Two other types of incontinence, overflow and functional, also occur in women, but they are far less common and are not reviewed here in detail. Overflow incontinence (urinary retention) is most often caused by adverse effects of anticholinergic drugs or by impaired detrusor innervation from neurologic disease. If no reversible cause is found, treatment usually requires intermittent or indwelling catheter drainage of the bladder to avoid hydronephrosis and renal impairment. Functional incontinence is caused by an inability to get to the toilet because of physical limitations or cognitive impairment. Treatment is aimed at assisting the patient with toileting.
Table 1. Classification of Urinary Incontinence in Women
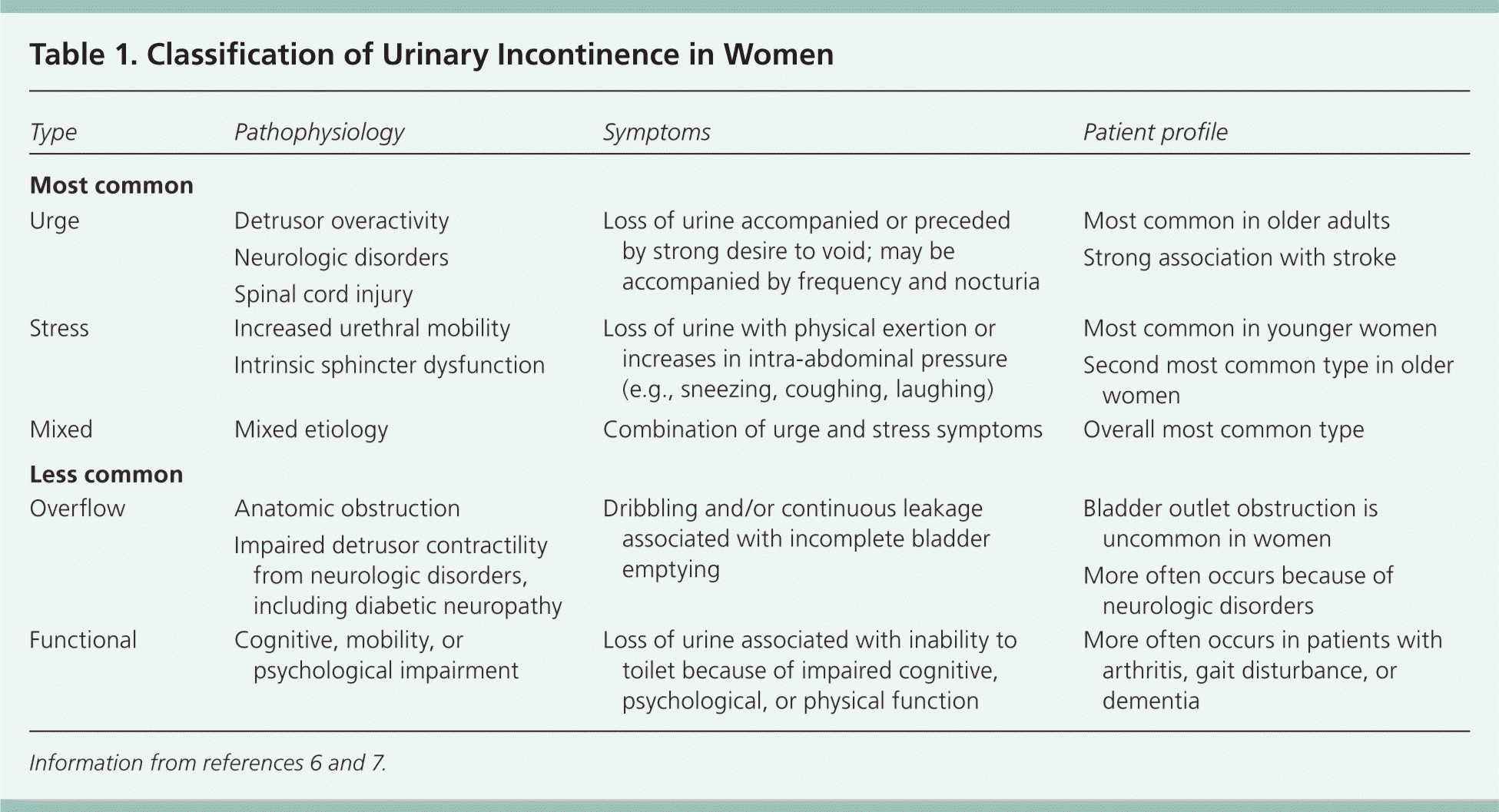
| Type | Pathophysiology | Symptoms | Patient profile |
|---|---|---|---|
| Most common | |||
| Urge |
|
|
|
| Stress |
|
|
|
| Mixed |
|
|
|
| Less common | |||
| Overflow |
|
|
|
| Functional |
|
|
|
General Treatment Considerations
Guidelines for diagnosis and treatment of urinary incontinence were published in 2012 by the American Urological Association.8 The usual approach to the treatment of urinary incontinence is a stepped-care plan starting with noninvasive behavioral modifications, followed by devices and pharmacologic interventions, and finally surgery in those whose symptoms do not respond to initial treatment. Treatment options are shown in Table 2.3,6,7,9–12 Lifestyle changes, including avoiding excessive fluid intake and limiting caffeinated or carbonated beverages, are also reasonable recommendations for all types of incontinence.13 When patients have mixed incontinence, treatment should be directed toward the predominant symptom.14
Table 2. Treatment Options for Urinary Incontinence
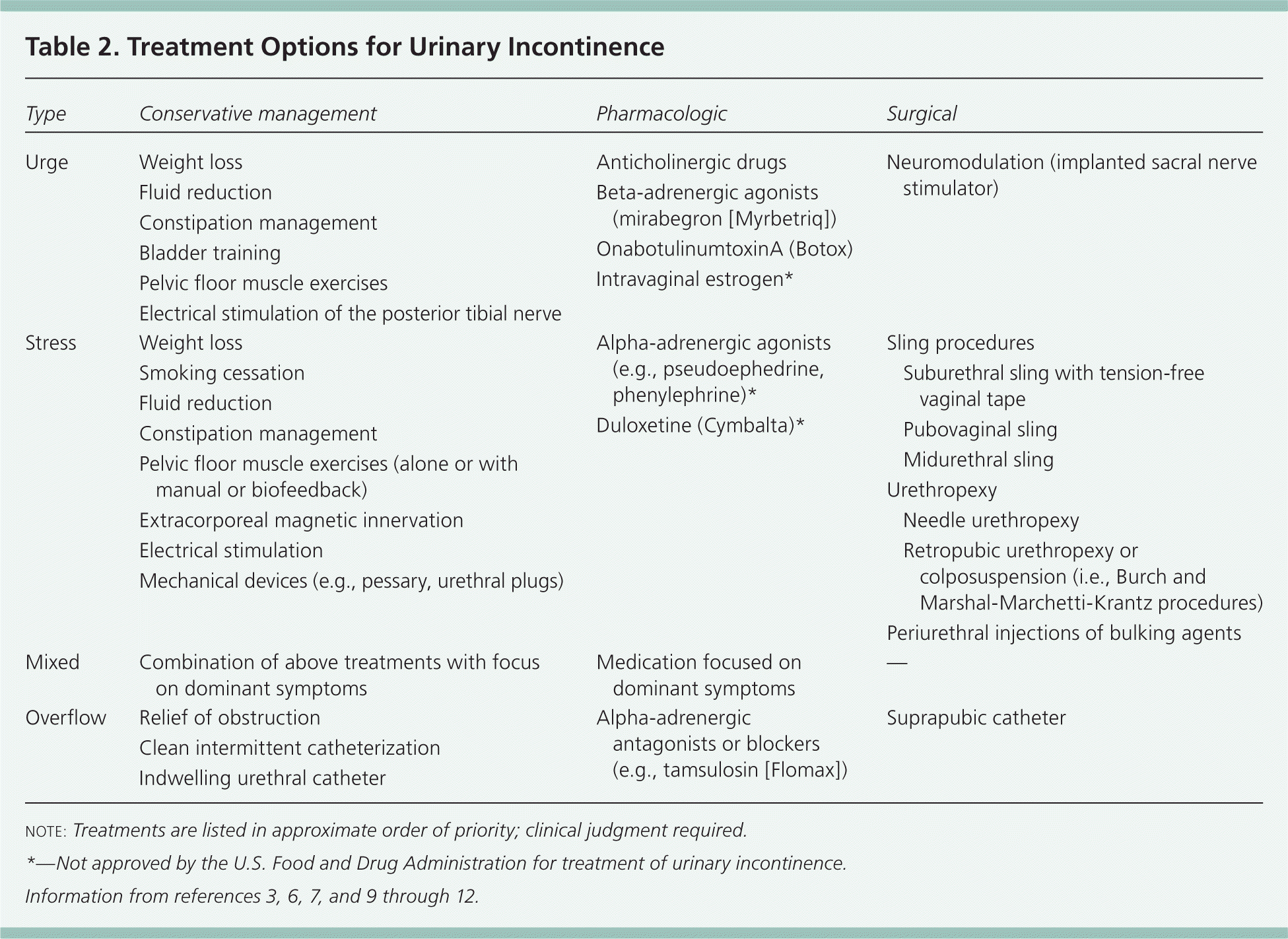
| Type | Conservative management | Pharmacologic | Surgical | |
|---|---|---|---|---|
| Urge |
|
|
| |
| Stress |
| Sling procedures
| ||
| Mixed |
|
|
| |
| Overflow |
|
|
| |
note: Treatments are listed in approximate order of priority; clinical judgment required.
*—Not approved by the U.S. Food and Drug Administration for treatment of urinary incontinence.
Urge Incontinence
Urge incontinence is characterized by an abrupt desire to void accompanied by an involuntary leakage of urine. This type of incontinence is common in older women.9 The broad diagnostic category of overactive bladder encompasses the symptoms of frequency, urgency, and nocturia; if these symptoms occur with incontinence, it is considered urge incontinence.6
BEHAVIORAL TREATMENTS
Behavioral strategies are first-line treatment for urge incontinence in persons with no cognitive impairment.13,15 Bladder training and pelvic floor muscle strengthening exercises can be used, and both are described in Table 3.13 Bladder training involves teaching the patient to suppress the urge to void.13 There is mixed evidence demonstrating improved outcomes with this strategy, stemming from studies of variable quality.14
Table 3. Common Behavioral Therapy for Urinary Incontinence
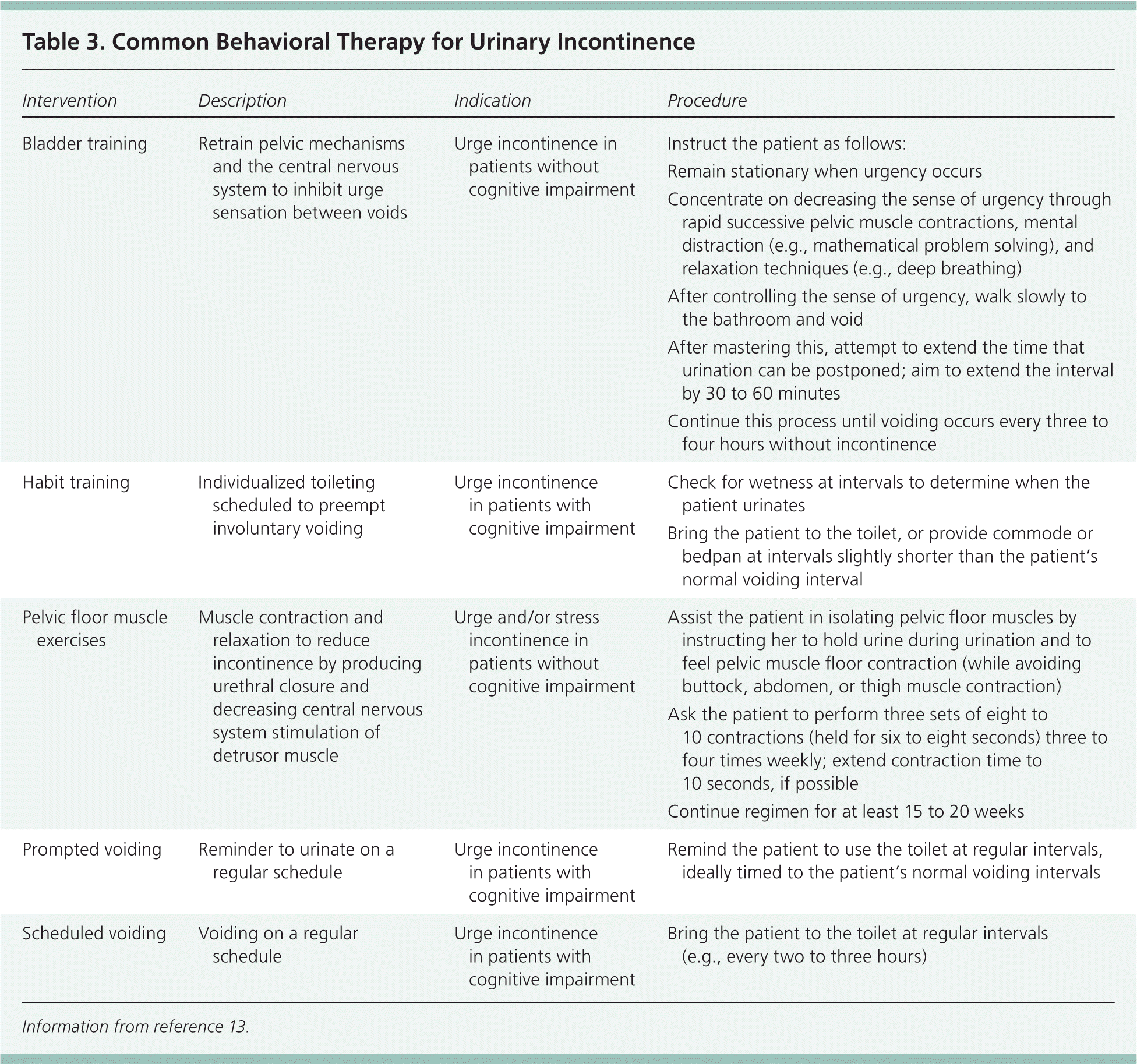
| Intervention | Description | Indication | Procedure |
|---|---|---|---|
| Bladder training | Retrain pelvic mechanisms and the central nervous system to inhibit urge sensation between voids | Urge incontinence in patients without cognitive impairment | Instruct the patient as follows: |
| Remain stationary when urgency occurs | |||
| Concentrate on decreasing the sense of urgency through rapid successive pelvic muscle contractions, mental distraction (e.g., mathematical problem solving), and relaxation techniques (e.g., deep breathing) | |||
| After controlling the sense of urgency, walk slowly to the bathroom and void | |||
| After mastering this, attempt to extend the time that urination can be postponed; aim to extend the interval by 30 to 60 minutes | |||
| Continue this process until voiding occurs every three to four hours without incontinence | |||
| Habit training | Individualized toileting scheduled to preempt involuntary voiding | Urge incontinence in patients with cognitive impairment | Check for wetness at intervals to determine when the patient urinates |
| Bring the patient to the toilet, or provide commode or bedpan at intervals slightly shorter than the patient's normal voiding interval | |||
| Pelvic floor muscle exercises | Muscle contraction and relaxation to reduce incontinence by producing urethral closure and decreasing central nervous system stimulation of detrusor muscle | Urge and/or stress incontinence in patients without cognitive impairment | Assist the patient in isolating pelvic floor muscles by instructing her to hold urine during urination and to feel pelvic muscle floor contraction (while avoiding buttock, abdomen, or thigh muscle contraction) |
| Ask the patient to perform three sets of eight to 10 contractions (held for six to eight seconds) three to four times weekly; extend contraction time to10 seconds, if possible | |||
| Continue regimen for at least 15 to 20 weeks | |||
| Prompted voiding | Reminder to urinate on a regular schedule | Urge incontinence in patients with cognitive impairment | Remind the patient to use the toilet at regular intervals, ideally timed to the patient's normal voiding intervals |
| Scheduled voiding | Voiding on a regular schedule | Urge incontinence in patients with cognitive impairment | Bring the patient to the toilet at regular intervals (e.g., every two to three hours) |
Information from reference 13.
Although many physicians think of pelvic floor muscle exercises, commonly known as Kegel exercises, as a treatment for only stress incontinence, they are also an effective treatment for urge incontinence. Properly performed pelvic floor muscle exercises are more effective than medications for reducing urge incontinence.16
For patients who have cognitive impairment and urge incontinence, several other behavioral strategies can be used, including habit training, scheduled voiding, and prompted voiding (Table 3).13 Studies indicate that scheduled voiding may be the most effective, with clear short-term benefits.17,18
DEVICES
Several electrical neuromodulation devices are approved by the U.S. Food and Drug Administration (FDA) for treating urge incontinence refractory to behavioral interventions. Although the precise mode of action is unknown, the effects can be explained by modulating reflex pathways. Techniques include the use of removable vaginal or anal stimulators and percutaneous stimulators of the posterior tibial nerve, which shares a common nerve root with the innervation of the bladder.
Posterior tibial nerve stimulators are the most widely used devices. In this office-based procedure, a needle electrode is applied near the medial malleolus, and electrical stimulation is administered in 30-minute sessions. Posterior tibial nerve stimulation works at least as well as medication, reducing urge incontinence in up to 75 percent of patients.19 A demonstration of the technique can be viewed at http://www.youtube.com/watch?v=0drKILgxhiM.
PHARMACOLOGIC THERAPY
Medications can be used to treat urge and mixed incontinence if behavioral therapy is unsuccessful. Cure is rarely achieved solely with drug therapy, however, and in many studies improvement over placebo is modest.6,7 Combination therapy with medication and behavioral treatments is more effective than either modality alone.6,7
Anticholinergic Drugs. Anticholinergics are the preferred agents for the treatment of urge incontinence14 (Table 43,9,10,20,21 ). They reduce detrusor overactivity by antagonizing M2/M3 muscarinic receptors in the bladder.3 M2/M3-selective agents are preferred over nonselective agents, which also bind M1 receptors in the brain and can unmask cognitive dysfunction, although both selective and nonselective agents may do so.22,23 A trial of four to eight weeks is recommended. Although no one drug is clearly superior, long-acting agents are generally preferred because of greater effectiveness and fewer adverse anticholinergic effects.3,24,25 However, anticholinergic effects may occur with any of these medications, and patients commonly stop taking them.3,6,24
Table 4. Anticholinergic Drugs for Treatment of Urge Incontinence

| Drug | Formulations | Unique factors |
|---|---|---|
| Nonselective agents | ||
| Fesoterodine (Toviaz) | Extended release |
|
| Oxybutynin (Ditropan) | Extended release |
|
| Immediate release | ||
| Topical gel | ||
| Transdermal patch | ||
| Tolterodine (Detrol) | Extended release |
|
| Immediate release | ||
| Trospium (Sanctura) | Extended release |
|
| Immediate release | ||
| M2/M3-selective agents | ||
| Darifenacin (Enablex) | Extended release |
|
| Solifenacin (Vesicare) | Extended release | |
note: Adverse effect profile of these drugs includes tachycardia, palpitations, edema, confusion, psychomotor retardation, nausea, vomiting, abdominal pain, constipation, urinary retention, blurry vision, and dry mouth.
Because of adverse effects, the American Geriatrics Society recommends avoiding these medications in older adults unless no alternatives are available.26 Furthermore, the anticholinergic action of these medications can counter the procholinergic effect of cholinesterase inhibitors used to treat dementia. Dementia is a contraindication to the use of anticholinergics, as are narrow-angle glaucoma and gastrointestinal obstruction/gastric retention.3
Beta-Adrenergic Agonists. Approved by the FDA in 2012, mirabegron (Myrbetriq) is from a new class of drugs used to treat urge incontinence. Mirabegron acts on beta3-adrenergic receptors to relax the detrusor.27 Studies have shown that use of mirabegron results in one to two fewer incontinence episodes per day, similar to sustained-release tolterodine (Detrol).28 Common adverse effects are nausea, diarrhea, constipation, dizziness, and headache.27 Increased blood pressure can also occur, and mirabegron should not be used in patients with uncontrolled hypertension.27 When used with an anticholinergic, the risk of urinary retention increases.27
OnabotulinumtoxinA. Also recently approved by the FDA, injection of onabotulinumtoxinA (Botox) into the detrusor muscle can be considered for treating urge incontinence that has not responded to conservative treatments.7,9,25,29 OnabotulinumtoxinA is superior to placebo in reducing incontinence as well as in improving quality of life.29 Symptom reduction lasts three to six months.9,29 Optimal doses for effectiveness and long-term safety have not yet been determined.14,29
Estrogen. Although intravaginal estrogen is sometimes used to treat urge incontinence, neither intravaginal nor systemic estrogens are FDA-approved for this. Systemic estrogen has been shown to worsen incontinence.10,30
SURGERY
Urge incontinence can be treated with surgically implanted devices that stimulate the sacral, paraurethral, and pudendal nerves.31 Sacral nerve stimulators are most commonly used, and up to two-thirds of patients experience improvement in symptoms, which is notable because these devices are used only for symptoms that are refractory to all other treatment.14 Implantable devices are costly and carry a risk of surgical complications.31
Stress Incontinence
Stress incontinence is characterized by the involuntary loss of urine with increases in intra-abdominal pressure. It is the most common type of incontinence in younger women, but also occurs in older women.9 Key risk factors include childbirth, medications that relax the urethral sphincter, obesity, lung disease (from chronic cough), and prior pelvic surgeries.9 Numerous treatments are available, although few studies compare one treatment with another.
WEIGHT LOSS
Women who are overweight or obese and who experience stress incontinence should be encouraged to lose weight, which has been shown to reduce the frequency of incontinence symptoms.32
BEHAVIORAL TREATMENTS
Pelvic floor muscle exercises are the mainstay of behavioral therapy for stress incontinence11,13 (Table 313 ). Up to 38 percent of patients with stress incontinence alone who follow a pelvic floor muscle exercise regimen for at least three months experience a cure.14,33 Increased effectiveness is demonstrated in women undergoing longer training and in those following comprehensive clinic-based training rather than self-help booklets.34,35
Manual feedback (palpating the pelvic muscles during the exercises) and biofeedback (using a vaginal or anal device that provides visual or audio feedback about pelvic muscle contraction) have been used to teach patients the correct technique.13,36 Weighted intravaginal cones have also been used for improving technique when women have difficulty identifying their pelvic floor muscles. Although these strategies may improve technique and, consequently, symptoms in the short term, there is no evidence that they result in higher rates of long-term improvement or cure than the exercises alone.37,38
ELECTRICAL AND MAGNETIC STIMULATION
Electrical stimulation of the pelvic floor muscles with a vaginal or anal electrode can be used in women who cannot voluntarily contract pelvic floor muscles.13 This can be done at home and typically consists of two 15-minute sessions daily for 12 weeks. Medicare has approved its use in patients who have incontinence that does not respond to structured pelvic floor muscle exercise programs.
Extracorporeal magnetic innervation involves a series of treatments in which the patient sits, fully clothed, on a chair that generates a low-power magnetic field. Patients typically undergo two or three treatments per week for six to eight weeks. One early study showed this method to be most effective for women who have mild stress incontinence (i.e., using three sanitary pads per day or fewer).39 A more recent study found it to be more effective than sham treatment for women who are unable to generate adequate pelvic floor muscle contractions.40
DEVICES
Vaginal inserts, including incontinence pessaries and incontinence tampons, can be used for treating stress incontinence in pregnant women, in those who are not surgical candidates, and in those whose symptoms have not responded to previous surgeries.41 Vaginal inserts compress the bladder neck and urethra, thus decreasing urine loss caused by stress incontinence. Although pessaries are not widely used, their associated risks and costs are low, and they achieve results quickly. There are few contraindications to pessary use (e.g., active pelvic infection, severe ulceration, allergy to product materials, noncompliance).41 Incontinence tampons, which also place pressure on the bladder neck, are available in Europe.
[ corrected] Urethral plugs are devices that are inserted into the urethra to prevent urine loss during activities that cause stress incontinence (e.g., running).42 They are available in two lengths: 3.5 cm and 4.5 cm. There is limited evidence promoting or discouraging their use,43 and they are associated with a number of adverse effects, including urinary tract infection (occurring in up to 31 percent of women over a two-year period), hematuria (3 percent), and migration into the bladder (1 percent).42 Despite this, multi-year studies indicate a high degree of patient satisfaction, and the likelihood of significant adverse effects diminishes with continued use.43
PHARMACOLOGIC THERAPY
No medications are FDA-approved for the treatment of stress incontinence. Alpha-adrenergic agonists, such as pseudoephedrine and phenylephrine, cause urethral constriction and, in theory, should decrease stress incontinence.3 However, there is only weak evidence to support their superiority over placebo.3,44 Adverse effects range from insomnia and anxiety to hypertension, arrhythmias, and stroke.44
Although it is not FDA-approved for the treatment of stress incontinence, the antidepressant duloxetine (Cymbalta) has been shown to reduce stress incontinence,45 but there is no evidence that it generates cure. Most women discontinue therapy within four weeks, citing adverse effects (45 percent) or lack of effectiveness (24 percent).10,35 Given some evidence of benefit, however, a patient who has stress incontinence and who is a candidate for antidepressant treatment may receive dual benefit from duloxetine. Other antidepressants demonstrate no objective improvement in incontinence.35
MINIMALLY INVASIVE PROCEDURES
Radiofrequency denaturation is a nonsurgical modality that uses a device inserted into the urethra to deliver radiofrequency energy. This energy denatures collagen in the bladder neck and proximal urethra, causing a reduction in compliance of those structures. The onetime procedure can be performed with local anesthesia in a physician's office. Cure rates range from 22 to 67 percent, and more than 50 percent of patients report symptom improvement up to three years after a single treatment.11,46,47 The procedure is safe, with the most common adverse effects including dysuria and urinary tract infections.11,47
Injection of bulking agents is a minimally invasive procedure for women with intrinsic weakness of the urethral sphincter whose symptoms do not respond to noninvasive treatments and who cannot undergo surgery. Agents such as autologous fat, collagen, or carbon beads are injected through a needle placed transurethrally or periurethrally. Cure or improvement rates range from 18 to 40 percent, with repeated injections required to maintain effectiveness. Adverse effects include urinary retention, urgency, dysuria, and urinary tract infections.11
SURGERY
Surgery is reserved for incontinence that does not respond to less invasive treatments.6 About 30 percent of women with stress incontinence ultimately elect to undergo surgery.48 Adverse outcomes include perioperative complications, development of urgency and urge incontinence, pelvic organ prolapse, and need for repeat surgery.
Surgical options include slings and urethropexy. Sling procedures include pubovaginal slings and midurethral slings (i.e., retropubic sling, single incision sling [mini-sling], tension-free vaginal tape, and transobturator sling). Urethropexy options include needle urethropexy and retropubic urethropexy (i.e., Burch and Marshall-Marchetti-Krantz procedures). There is no consensus on the best surgical approach. All methods aim to augment urethral closure, or support and stabilize the bladder neck and urethra.12,33,48,49 One of the most commonly performed approaches is the use of tension-free vaginal tape. In this outpatient procedure, with the patient under local anesthesia, the tape is inserted to create a sling around the urethra. Because of its acceptable adverse effect profile, demonstrated effectiveness, and durability, some are considering tension-free vaginal tape the new standard for surgical treatment of stress incontinence.12
Data Sources: We consulted general clinical review articles on the subject, and searched the Cochrane database, Agency for Healthcare Research and Quality, and the National Guideline Clearinghouse, as well as pertinent articles from reference bibliographies. Search date: August 1, 2012.

