Latent tuberculosis infection refers to an asymptomatic, nontransmissible infection with Mycobacterium tuberculosis, carrying a 5% to 10% lifetime risk of progressing to active disease. One-half of this risk occurs within the first two years after infection. High-risk groups include recent immigrants from high-incidence countries, health care professionals, persons living or working in institutional settings, and homeless persons. Risk factors for progression to active disease include immunodeficiency, recent exposure to tuberculosis, and chronic kidney disease requiring dialysis. Tuberculin skin testing has several limitations, including the need for multiple office visits and the potential for false-positive results in patients who have received the bacillus Calmette-Guérin vaccine or been exposed to environmental mycobacteria. Interferon-gamma release assays address these deficiencies but are limited by their cost and requirement for blood processing. Interferon-gamma release assays are preferred in immigrants exposed to bacillus Calmette-Guérin and in patients who are not likely to return for interpretation of skin test results. Tuberculin skin testing is preferred for children younger than five years. Active disease should be excluded before initiating treatment. The newest treatment option of 12 weekly doses of isoniazid and rifapentine has similar or better effectiveness than standard nine-month therapy with daily isoniazid. A four-month regimen of daily rifampin is another alternative.
Infection with Mycobacterium tuberculosis is transmitted by airborne droplets from patients with active respiratory disease.1 After the primary infection, tuberculosis (TB) can progress to active pulmonary disease (most common) or extra-pulmonary disease, or it can remain latent for part or all of the patient's life. About one-third of the world's population is thought to be infected with M. tuberculosis, including an estimated 10 to 15 million latent infections in the United States.2 There is a 5% to 10% lifetime risk of progression from latent TB to active disease.1 One-half of this progression occurs in the first two years after infection, with the remaining risk distributed over the rest of the life span.3 Risk factors for infection and for progression from latent to active disease are listed in Table 1.3–7 Because more than 80% of active TB cases in the United States arise from latent TB, prompt treatment is key to prevent active disease.3 Patients with latent TB are noninfectious and typically do not feel ill, so the only indication of latent infection is a positive screening test (Table 2).7
SORT: KEY RECOMMENDATIONS FOR PRACTICE
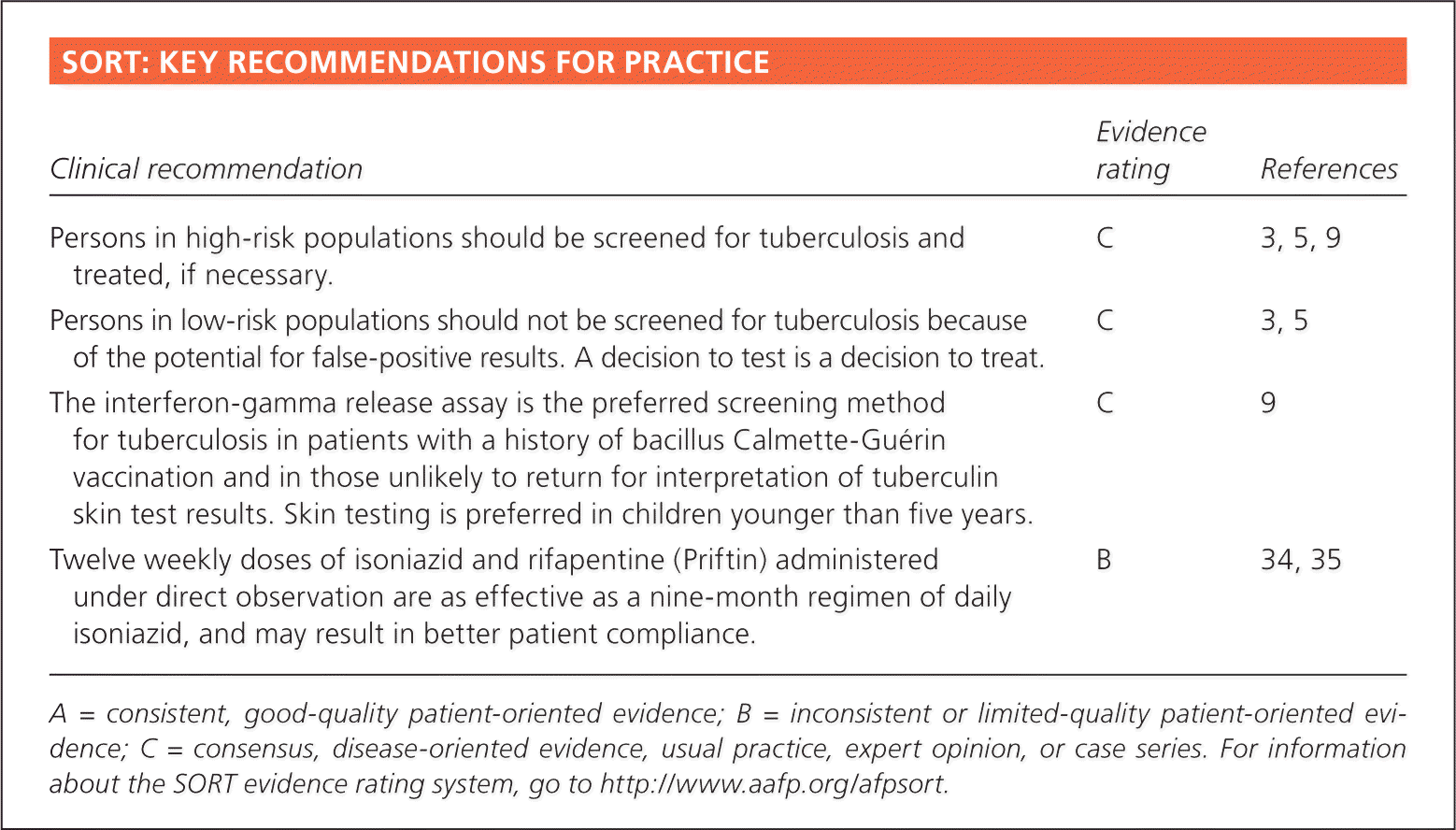
| Clinical recommendation | Evidence rating | References |
|---|---|---|
| Persons in high-risk populations should be screened for tuberculosis and treated, if necessary. | C | 3, 5, 9 |
| Persons in low-risk populations should not be screened for tuberculosis because of the potential for false-positive results. A decision to test is a decision to treat. | C | 3, 5 |
| The interferon-gamma release assay is the preferred screening method for tuberculosis in patients with a history of bacillus Calmette-Guérin vaccination and in those unlikely to return for interpretation of tuberculin skin test results. Skin testing is preferred in children younger than five years. | C | 9 |
| Twelve weekly doses of isoniazid and rifapentine (Priftin) administered under direct observation are as effective as a nine-month regimen of daily isoniazid, and may result in better patient compliance. | B | 34, 35 |
A = consistent, good-quality patient-oriented evidence; B = inconsistent or limited-quality patient-oriented evidence; C = consensus, disease-oriented evidence, usual practice, expert opinion, or case series. For information about the SORT evidence rating system, go to https://www.aafp.org/afpsort.
Table 1. Risk Factors for Tuberculosis and Rates of Active Disease by Ethnicity
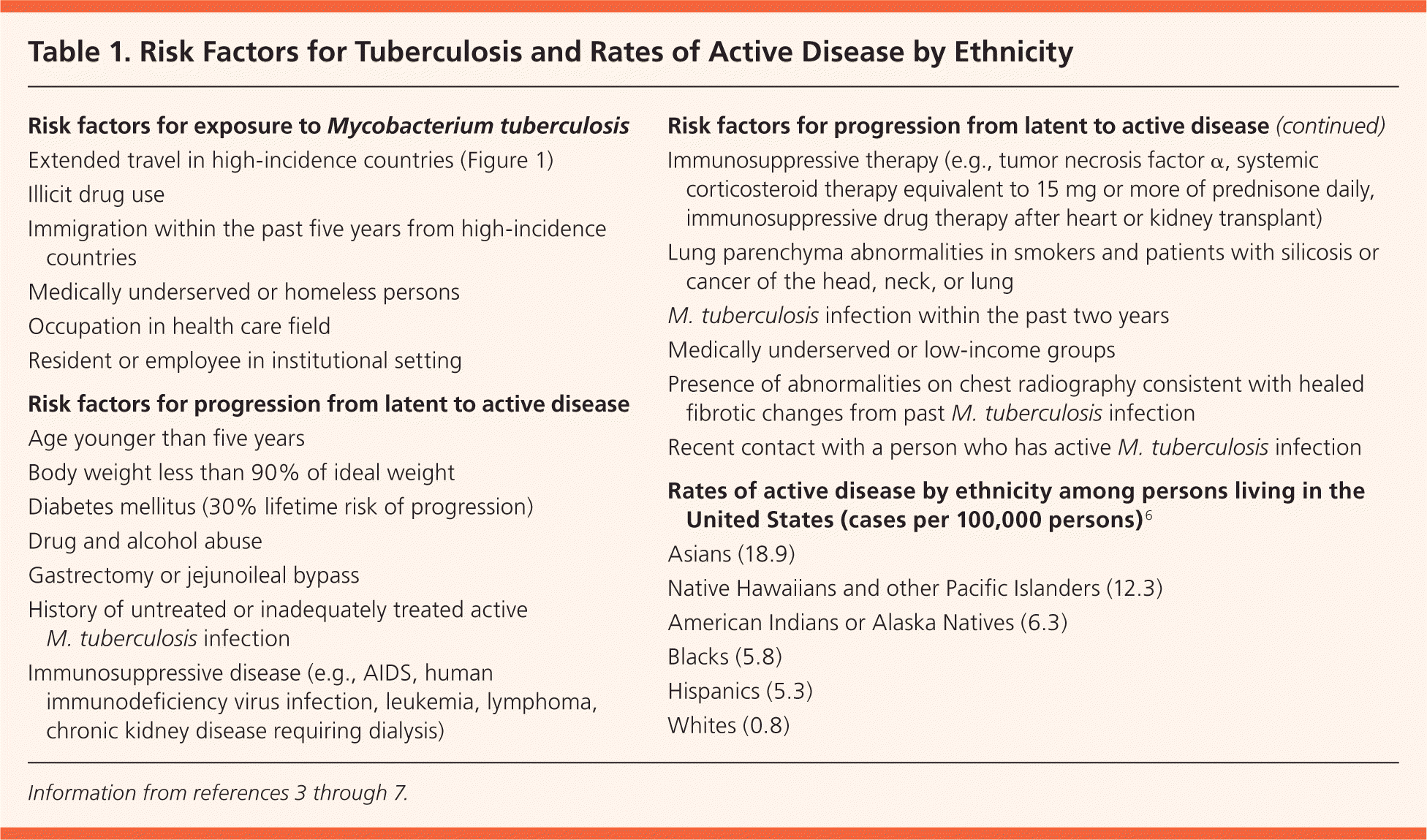
| Risk factors for exposure to |
| Extended travel in high-incidence countries (Figure 1) |
| Illicit drug use |
| Immigration within the past five years from high-incidence countries |
| Medically underserved or homeless persons |
| Occupation in health care field |
| Resident or employee in institutional setting |
| Risk factors for progression from latent to active disease |
| Age younger than five years |
| Body weight less than 90% of ideal weight |
| Diabetes mellitus (30% lifetime risk of progression) |
| Drug and alcohol abuse |
| Gastrectomy or jejunoileal bypass |
| History of untreated or inadequately treated active M. tuberculosis infection |
| Immunosuppressive disease (e.g., AIDS, human immunodeficiency virus infection, leukemia, lymphoma, chronic kidney disease requiring dialysis) |
| Immunosuppressive therapy (e.g., tumor necrosis factor α, systemic corticosteroid therapy equivalent to 15 mg or more of prednisone daily, immunosuppressive drug therapy after heart or kidney transplant) |
| Lung parenchyma abnormalities in smokers and patients with silicosis or cancer of the head, neck, or lung |
| M. tuberculosis infection within the past two years |
| Medically underserved or low-income groups |
| Presence of abnormalities on chest radiography consistent with healed fibrotic changes from past M. tuberculosis infection |
| Recent contact with a person who has active M. tuberculosis infection |
| Rates of active disease by ethnicity among persons living in the United States (cases per 100,000 persons)6 |
| Asians (18.9) |
| Native Hawaiians and other Pacific Islanders (12.3) |
| American Indians or Alaska Natives (6.3) |
| Blacks (5.8) |
| Hispanics (5.3) |
| Whites (0.8) |
Table 2. Comparison of Latent vs. Active Tuberculosis Infection
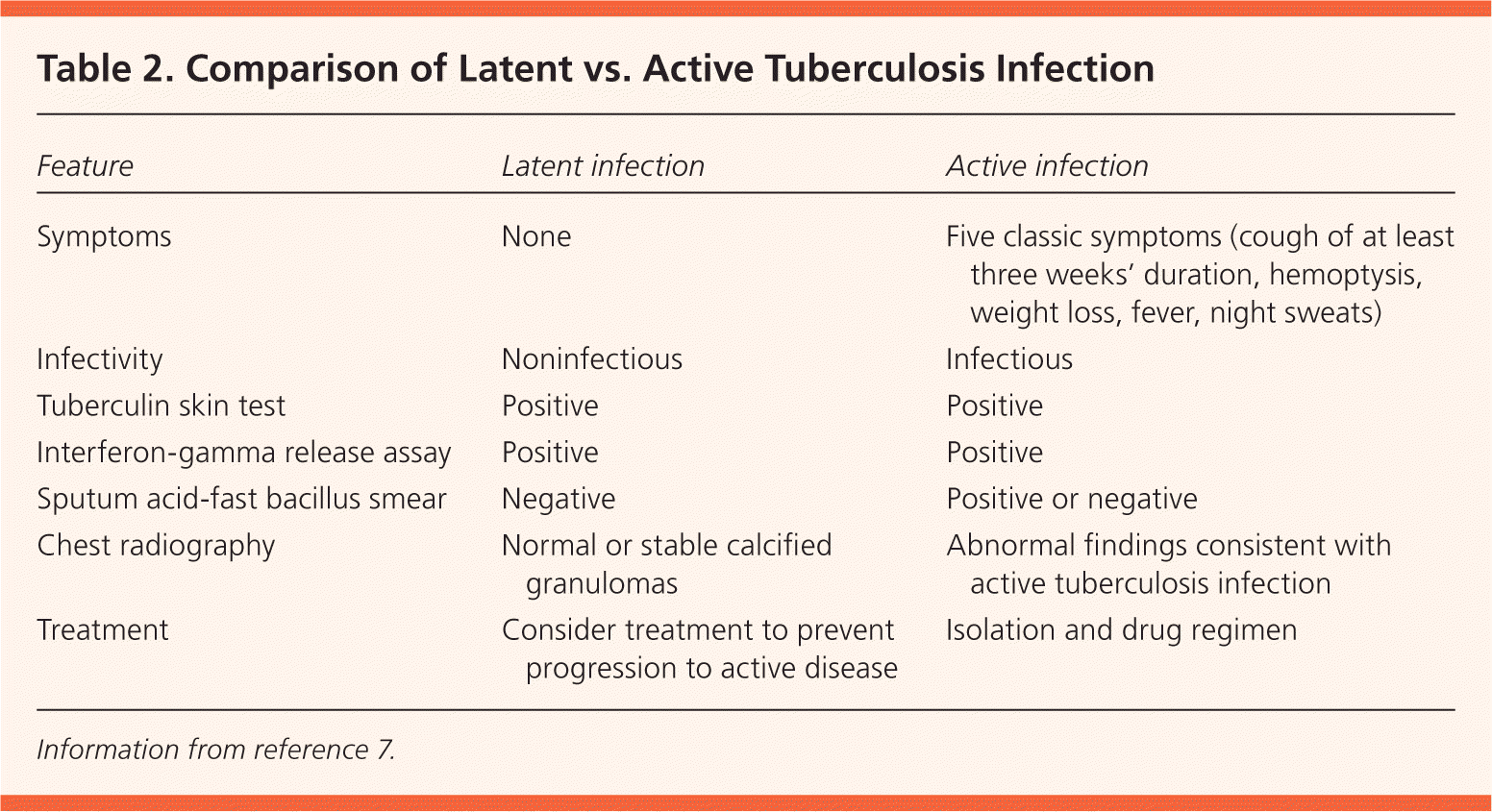
| Feature | Latent infection | Active infection |
|---|---|---|
| Symptoms | None | Five classic symptoms (cough of at least three weeks' duration, hemoptysis, weight loss, fever, night sweats) |
| Infectivity | Noninfectious | Infectious |
| Tuberculin skin test | Positive | Positive |
| Interferon-gamma release assay | Positive | Positive |
| Sputum acid-fast bacillus smear | Negative | Positive or negative |
| Chest radiography | Normal or stable calcified granulomas | Abnormal findings consistent with active tuberculosis infection |
| Treatment | Consider treatment to prevent progression to active disease | Isolation and drug regimen |
Information from reference 7.
Risk Factors Warranting Testing
Targeted screening is recommended only for individuals and groups at increased risk of TB infection.5 Routine testing outside of high-risk groups leads to more false-positive results, creates needless anxiety, and wastes resources.5 High-risk groups include recent immigrants (within the past five years) from high-incidence countries, health care professionals, persons living or working in institutional settings, and homeless persons.4 High-incidence areas include most of the countries in Africa, Asia, eastern Europe, Central America, and South America (Figure 1).8 Asian immigrants in the United States are at significant risk, with 25 times the rate of active TB compared with non-Hispanic whites.9 The only low-risk persons who should be screened are those who will be entering a high-risk group in the future, such as for work or travel.5
Figure 1.
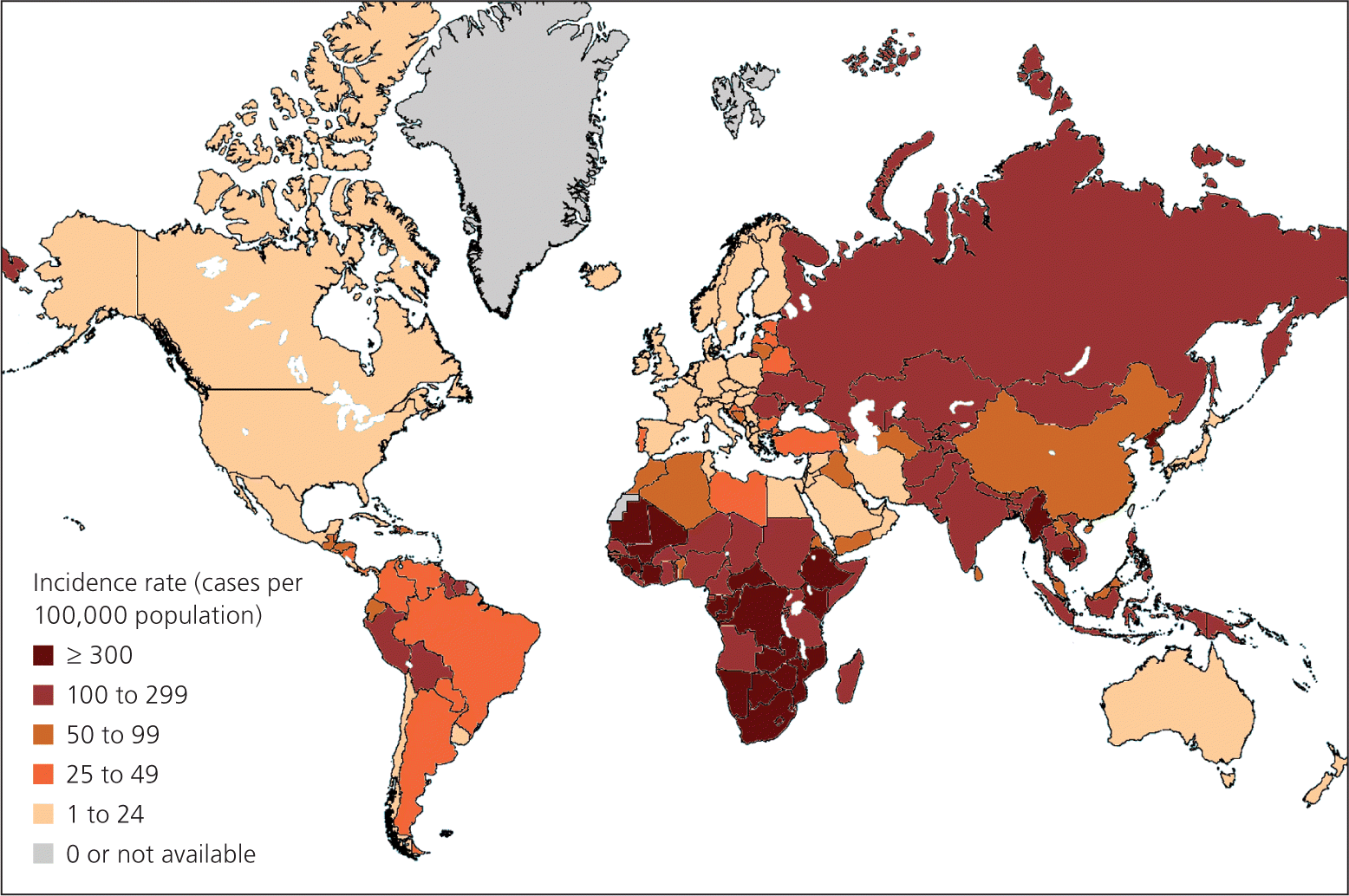
World map of estimated tuberculosis incidence rates.
Reprinted from LoBue P; Centers for Disease Control and Prevention. Travelers' health: chapter 3. Infectious diseases related to travel: tuberculosis. http://wwwnc.cdc.gov/travel/yellowbook/2012/chapter-3-infectious-diseases-related-to-travel/tuberculosis.htm. Accessed January 29, 2014.
Screening and Testing Options
Screening for TB is facilitated by questionnaires designed to identify high-risk persons. Because the decision to screen for latent TB infection is also a decision to treat,3 screening should be discouraged in low-risk populations and in those likely to refuse treatment.5
Screening options for TB include the tuberculin skin test (TST) and interferon-gamma release assay (IGRA; Table 3).9,10 Although the IGRA is more specific and less sensitive than the TST, it does not eliminate the need for targeted testing in high-risk populations.11 Interpretation of both tests must be based on the patient's immune status, history of exposure to TB and bacillus Calmette-Guérin (BCG), and other risk factors.9 With the TST, an induration of 15 mm or more is considered positive in persons without risk factors, 10 mm or more is positive for those at higher risk, and 5 mm or more is positive for certain high-risk persons (e.g., immunocompromised patients, those exposed to active TB).12,13
Table 3. Centers for Disease Control and Prevention Recommendations for Use of the IGRA and TST

| IGRA preferred; TST acceptable |
| Groups with historically low rates of returning for TST result interpretation (e.g., homeless persons, illicit drug users) |
| Persons who have received bacillus Calmette-Guérin vaccine |
| TST preferred; IGRA acceptable |
| Children younger than five years* |
| Either test acceptable |
| Persons who have occupational exposure to Mycobacterium tuberculosis and are undergoing periodic screening |
| Recent contacts of persons with known or suspected active tuberculosis† |
| Both tests may be considered |
| Persons in whom the initial IGRA result is indeterminate, borderline, or invalid, but a reason for testing persists‡ |
| Persons with negative initial test results in whom the risk of infection, progression, or a poor outcome is high (e.g., immunocompromised persons, children younger than five years)§ |
| Persons with negative initial test results in whom clinical suspicion exists for active tuberculosis (e.g., those with symptoms, signs, or radiographic evidence of active disease)§ |
| Persons with positive initial test results in whom additional evidence of infection is needed to encourage compliance with treatment |
| Persons with positive initial test results who are healthy and at low risk of progression |
IGRA = interferon-gamma release assay; TST = tuberculin skin test.
*—Other recommendations regarding the use of IGRAs in children have been published by the American Academy of Pediatrics.10
†—If test results are negative in the first eight weeks after initial exposure, repeat testing should be performed eight to 10 weeks after the end of exposure.
‡—Repeat testing with another blood sample may provide interpretable results.
§—If initial test results are negative, a positive result from a second test increases detection sensitivity. However, multiple negative results from any combination of tests do not exclude infection.
Use of the IGRA is preferred for persons who have received the BCG vaccine and those who are less likely to return for interpretation of TST results (e.g., homeless persons).9 Despite previous recommendations that TST results be interpreted without regard for BCG vaccination status, about 20% of positive TST results in this population may represent false positives rather than latent or active TB infection. False-positive results are more likely if the interval between BCG vaccination and use of the TST is less than 10 years.14 Therefore, the IGRA is preferred for immigrants who have received the BCG vaccine.9
The TST is preferred for children younger than five years9 because of reported variability and decreased sensitivity of IGRA testing in this age group.15 Children younger than five years are at higher risk of progression to more severe active disease, probably because of a lower interferon-gamma and T-cell response compared with older children and adults.16
Screening with the TST or IGRA may be performed after exposure to active TB. Either test may not show conversion for eight to 12 weeks in infected individuals, so “window prophylaxis” should be initiated in exposed high-risk patients, such as immunosuppressed persons or young children5 (Table 41,2,17–20 ). If repeat testing at 12 weeks is negative, medications can be stopped, although further clinical evaluation is indicated for immunosuppressed patients.9
Table 4. Treatment Options for Latent Tuberculosis Infection*
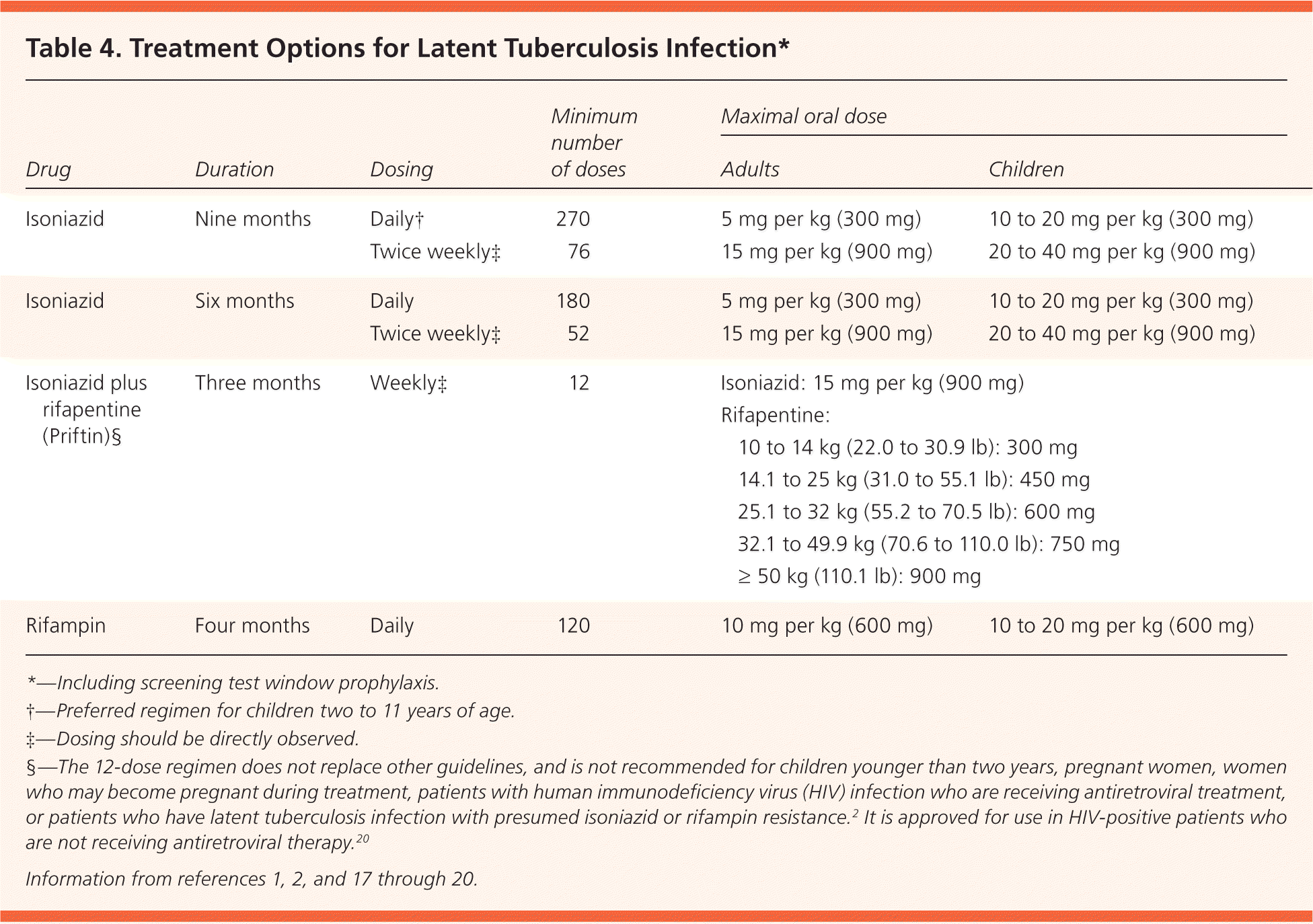
| Drug | Duration | Dosing | Minimum number of doses | Maximal oral dose | ||
|---|---|---|---|---|---|---|
| Adults | Children | |||||
| Isoniazid | Nine months | Daily† | 270 | 5 mg per kg (300 mg) | 10 to 20 mg per kg (300 mg) | |
| Twice weekly‡ | 76 | 15 mg per kg (900 mg) | 20 to 40 mg per kg (900 mg) | |||
| Isoniazid | Six months | Daily | 180 | 5 mg per kg (300 mg) | 10 to 20 mg per kg (300 mg) | |
| Twice weekly‡ | 52 | 15 mg per kg (900 mg) | 20 to 40 mg per kg (900 mg) | |||
| Isoniazid plus rifapentine (Priftin)§ | Three months | Weekly‡ | 12 | Isoniazid: 15 mg per kg (900 mg) | ||
Rifapentine:
| ||||||
| Rifampin | Four months | Daily | 120 | 10 mg per kg (600 mg) | 10 to 20 mg per kg (600 mg) | |
*—Including screening test window prophylaxis.
†—Preferred regimen for children two to 11 years of age.
‡—Dosing should be directly observed.
§—The 12-dose regimen does not replace other guidelines, and is not recommended for children younger than two years, pregnant women, women who may become pregnant during treatment, patients with human immunodeficiency virus (HIV) infection who are receiving antiretroviral treatment, or patients who have latent tuberculosis infection with presumed isoniazid or rifampin resistance.2 It is approved for use in HIV-positive patients who are not receiving antiretroviral therapy.20
The TST or IGRA may be used for periodic screening of persons at risk of occupational exposure to M. tuberculosis. IGRA results are more likely to be positive in recently infected persons who are at highest risk of progression to active disease.21 IGRA results can be obtained after a single visit. Disadvantages of IGRA use include the processing requirements and higher cost.22 Tests can range from up to $25 at local health departments to $140 to $260 at a private reference laboratory.
The two-step TST involves an initial test and then a repeat test in one to three weeks to stimulate a reaction or booster effect in persons infected with M. tuberculosis in the distant past.13 When results of both the initial test and the repeat test are negative, a negative baseline is established. This process is often used for persons who may need frequent screening, such as health care professionals. If the first test is negative but the subsequent test is positive, infection should be assumed.
Discordant Test Results
Clinical judgment should be used when assessing discordant test results, taking into consideration the patient's risk of infection, of developing active disease, and of poor outcomes. The Centers for Disease Control and Prevention supports the use of the IGRA in addition to the TST in specific circumstances.9 It does not advocate confirmatory testing (IGRA to confirm positive TST results), unlike the United Kingdom, Switzerland, France, and Canada.23–25 Despite this, some U.S. health departments have adopted confirmatory IGRA. There is a 78.9% concordance rate between TST and IGRA results in healthy populations.17 Agreement is much more likely when TST results are negative (90.6% vs. 44.4% when positive).26 When results are discordant, TB risk factors and potential causes of false-positive and false-negative results should be considered (Table 5).5,9,12,17,22 The TST and IGRA measure different aspects of the immune response, with different interpretation criteria9 (Table 65,9,17,20,22,27 ). Most discordance in low-prevalence populations is related to these differences.28
Table 5. Causes of False-Positive and False-Negative IGRA and TST Results
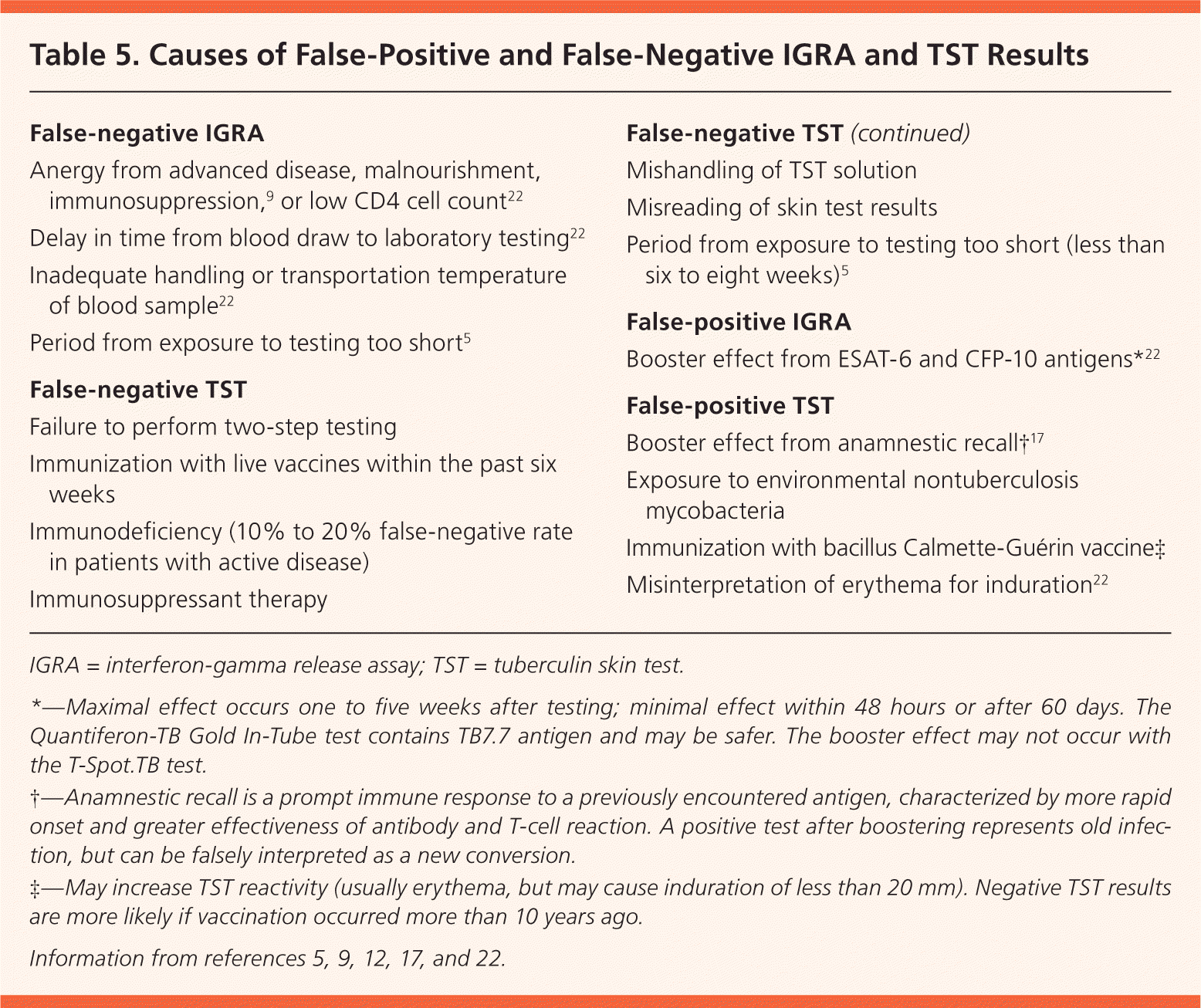
| False-negative IGRA |
| Anergy from advanced disease, malnourishment, immunosuppression,9 or low CD4 cell count22 |
| Delay in time from blood draw to laboratory testing22 |
| Inadequate handling or transportation temperature of blood sample22 |
| Period from exposure to testing too short5 |
| False-negative TST |
| Failure to perform two-step testing |
| Immunization with live vaccines within the past six weeks |
| Immunodeficiency (10% to 20% false-negative rate in patients with active disease) |
| Immunosuppressant therapy |
| Mishandling of TST solution |
| Misreading of skin test results |
| Period from exposure to testing too short (less than six to eight weeks)5 |
| False-positive IGRA |
| Booster effect from ESAT-6 and CFP-10 antigens*22 |
| False-positive TST |
| Booster effect from anamnestic recall†17 |
| Exposure to environmental nontuberculosis mycobacteria |
| Immunization with bacillus Calmette-Guérin vaccine‡ |
| Misinterpretation of erythema for induration22 |
IGRA = interferon-gamma release assay; TST = tuberculin skin test.
*—Maximal effect occurs one to five weeks after testing; minimal effect within 48 hours or after 60 days. The Quantiferon-TB Gold In-Tube test contains TB7.7 antigen and may be safer. The booster effect may not occur with the T-Spot.TB test.
†—Anamnestic recall is a prompt immune response to a previously encountered antigen, characterized by more rapid onset and greater effectiveness of antibody and T-cell reaction. A positive test after boostering represents old infection, but can be falsely interpreted as a new conversion.
‡—May increase TST reactivity (usually erythema, but may cause induration of less than 20 mm). Negative TST results are more likely if vaccination occurred more than 10 years ago.
Table 6. Comparison of Tuberculosis Tests

| Test characteristic | Tuberculin skin test | Interferon-gamma release assays | |
|---|---|---|---|
| Quantiferon-TB Gold In-Tube test | T-Spot.TB test | ||
| Format22 | Purified protein derivative injected intradermally; patient must return in 48 to 72 hours for results [ | Enzyme-linked immunosorbent assay using whole blood; processed within 16 hours | Enzyme-linked immunosorbent spot test using peripheral blood mononuclear cells; processed in eight to 24 hours (up to 30 hours if T-Cell Xtend is used) |
| Antigens27 | Tuberculin proteins5 | ESAT-6, CFP-10, TB7.7 | Mixtures of synthetic peptides representing ESAT-6 and CFP-10 |
| Measurement27 | Size of skin induration | Interferon-gamma level | Interferon-gamma–producing cell count |
| Sensitivity* | 80% to 95%9 | 70% to 91%9 | 84% to 91%9 |
| Specificity* | 80%9 | 95% to 99%20 | 95% to 97%20 |
| Results affected by bacillus Calmette-Guérin vaccination? | Yes | No | No |
*—Sensitivity and specificity of tests vary because of the lack of standard for comparison, and are based on the population studied.17
Screening tests cannot detect the presence of M. tuberculosis; instead, they measure the host's immune response to past or current infection.9 The TST is based on delayed hypersensitivity to TB antigens. The current generation of IGRAs measures interferon-gamma released upon stimulation of T cells in response to the ESAT-6 and CFP-10 antigens of M. tuberculosis.5,9,29 Although sensitive, the TST has poor specificity, partly because of cross reactivity and interpretation errors.9 Estimating the sensitivity of the TST and IGRA is problematic because of the lack of a preferred test for latent TB infection.17 The IGRA is more specific (greater than 95%) with fewer false-positive results because the test antigens are not shared by BCG or environmental mycobacteria (except Mycobacterium marinum, Mycobacterium szulgai, and Mycobacterium kansasii).17,29 It should be noted that the TST contains ESAT-6 and CFP-10 antigens, which may boost IGRA results.22
Confirmatory testing may be considered in high-risk patients. Because of impaired immune response in these patients, negative TST results may be confirmed with an IGRA.9 Conversely, in low-risk patients, positive TST (or IGRA) results should not be considered proof of infection. Relying solely on positive TST results in low-risk patients could lead to unnecessary treatment, whereas additional IGRA testing could result in fewer treatment recommendations.22 IGRA confirmation of positive TST results may persuade reluctant patients to consider treatment.9
Treatment
Neither the TST nor the IGRA can distinguish between latent and active disease.17,20 At-risk patients with positive TB test results should be offered treatment after active disease is ruled out.5 A patient history should be obtained, and a physical examination and chest radiography should be performed. Any radiographic abnormalities should be followed with three sputum acid-fast bacillus smears to exclude active TB.
There are four approved treatment regimens for latent TB infection (Table 4).1,2,17–20 The standard nine-month isoniazid regimen reduces TB risk by 90% in patients who are fully compliant; however, only 64% of patients complete at least six months of therapy.3,30 A six-month regimen of isoniazid reduces risk by 60% to 80%.3 The less-studied four-month rifampin regimen is less expensive, has better patient compliance (69% to 78%), and has less liver toxicity31,32; four months of treatment provides 60% protection.33 A three-month, 12-dose regimen of isoniazid and rifapentine (Priftin), given once weekly under direct observation to ensure compliance and safety, is more expensive but as effective as nine months of isoniazid in persons 12 years and older.34 Its shorter course may result in better patient compliance.35 Monitoring recommendations and adverse effects of available regimens are listed in Table 7.1,5 As more evidence-based recommendations become available, the evaluation and treatment of latent TB infection will continue to evolve.
Table 7. Monitoring and Adverse Effects of Drugs Used in the Treatment of Tuberculosis

| Drug | Clinical monitoring* | Adverse effects | Precautions |
|---|---|---|---|
| Isoniazid5 | Baseline and monthly liver function testing; repeat if abnormal† | Rash, increased liver transaminase levels, peripheral neuropathy, mild central nervous system effects, potential drug interactions with phenytoin (Dilantin) and disulfiram (Antabuse) | Hepatitis risk increases with age and alcohol consumption; vitamin B6 may be added to prevent peripheral neuropathy and central nervous system effects; active hepatitis and end-stage liver disease are relative contraindications |
| Rifampin5 | Complete blood and platelet counts, baseline liver function testing; repeat if abnormal† or if patient has signs or symptoms of tuberculosis | Rash, hepatitis, fever, thrombocytopenia, influenza-like symptoms, orange body fluids | Contraindicated or should be used with caution in HIV-positive patients receiving protease inhibitors or nonnucleoside reverse transcriptase inhibitors; decreases many drug levels |
| Rifapentine (Priftin)1 | Baseline and monthly clinical assessment and liver function testing; repeat if abnormal† or if patient has signs or symptoms of tuberculosis | Red secretions, staining of contact lenses, drug hypersensitivity reactions (particularly hypotension or thrombocytopenia); rarely, neutropenia and increased liver transaminase levels | Decreases many drug levels, particularly those metabolized by cytochrome P450 3A; safety in pregnancy is unknown; contraindicated in children younger than two years, HIV-positive patients receiving antiretrovirals, and patients who have latent tuberculosis infection with presumed resistance to isoniazid or rifampin |
HIV = human immunodeficiency virus.
*—Baseline laboratory testing is not routinely indicated for all patients (including older persons). It is indicated for patients with HIV infection or a history of chronic liver disease (e.g., hepatitis B or C, alcoholic hepatitis, cirrhosis), women who are pregnant or no more than three months postpartum, persons who consume alcohol regularly, and persons at risk of chronic liver disease. Testing may be considered on an individual basis, particularly for patients taking medications for chronic conditions.
†—Some experts recommend that isoniazid be withheld if liver transaminase levels are more than three times the upper limit of normal in symptomatic patients, or more than five times the upper limit of normal in asymptomatic patients.
Data Sources: A PubMed search was completed in Clinical Queries using the key terms tuberculosis, exposure to tuberculosis, latent tuberculosis infection, TST and IGRA testing for tuberculosis, and treatment of tuberculosis. The search included meta-analyses, randomized controlled trials, and reviews. Also searched were the National Guideline Clearinghouse, Cochrane Database of Systematic Reviews, Centers for Disease Control and Prevention website, and Essential Evidence Plus. References from within those sources, as well as from UpToDate, were also searched. Search dates: January 20, 2012, to March 20, 2012, and January 24 to 30, 2014.

