Epistaxis is a common emergency encountered by primary care physicians. Up to 60% of the general population experience epistaxis, and 6% seek medical attention for it. More than 90% of cases arise from the anterior nasal circulation, and most treatments can be easily performed in the outpatient setting. Evaluation of a patient presenting with epistaxis should begin with assessment of vital signs, mental status, and airway patency. When examining the nose, a nasal speculum and a good light source, such as a headlamp, can be useful. Compressive therapy is the first step to controlling anterior epistaxis. Oxymetazoline nasal spray or application of cotton soaked in oxymetazoline or epinephrine 1:1,000 may be useful adjuncts to compressive therapy. Directive nasal cautery, most commonly using silver nitrate, can be used to control localized continued bleeding or prominent vessels that are the suspected bleeding source. Finally, topical therapy and nasal packing can be used if other methods are unsuccessful. Compared with anterior epistaxis, posterior epistaxis is more likely to require hospitalization and twice as likely to need nasal packing. Posterior nasal packing is often associated with pain and a risk of aspiration if it is dislodged. After stabilization, patients with posterior packing often require referral to otolaryngology or the emergency department for definitive treatments.
Epistaxis is one of the most common otolaryngologic emergencies, occurring in up to 60% of the general population, with one in 10 of those affected seeking medical attention. It accounts for one in 200 emergency department visits.1,2 Epistaxis has a bimodal age distribution, peaking in children younger than 10 years and in adults between 70 and 79 years of age.1,3 Males are slightly more likely to experience epistaxis than females.4
SORT: KEY RECOMMENDATIONS FOR PRACTICE

| Clinical recommendation | Evidence rating | References |
|---|---|---|
| Compressive therapy should be the first intervention to stop anterior epistaxis. | C | 12, 13 |
| Silver nitrate and electrical desiccation are effective at stopping anterior epistaxis in patients when compressive therapy is unsuccessful. | C | 10, 14 |
| When available, endoscopic artery ligation may be the best initial treatment for posterior epistaxis because it is more effective than packing and less costly than endovascular embolization. | B | 20, 22, 23 |
A = consistent, good-quality patient-oriented evidence; B = inconsistent or limited-quality patient-oriented evidence; C = consensus, disease-oriented evidence, usual practice, expert opinion, or case series. For information about the SORT evidence rating system, go to https://www.aafp.org/afpsort.
Anatomy
Approximately 90% of epistaxis cases arise from the anterior part of the nasal septum; this is known as anterior epistaxis.5–7 Anterior bleeding most commonly occurs from the rich vascular supply at the Kiesselbach plexus (Figure 1).8 This plexus is formed by terminal branches of the internal carotid artery (anterior and posterior ethmoidal arteries) and external carotid artery (sphenopalatine, superior labial, and greater palatine arteries).3,6,9
FIGURE 1.
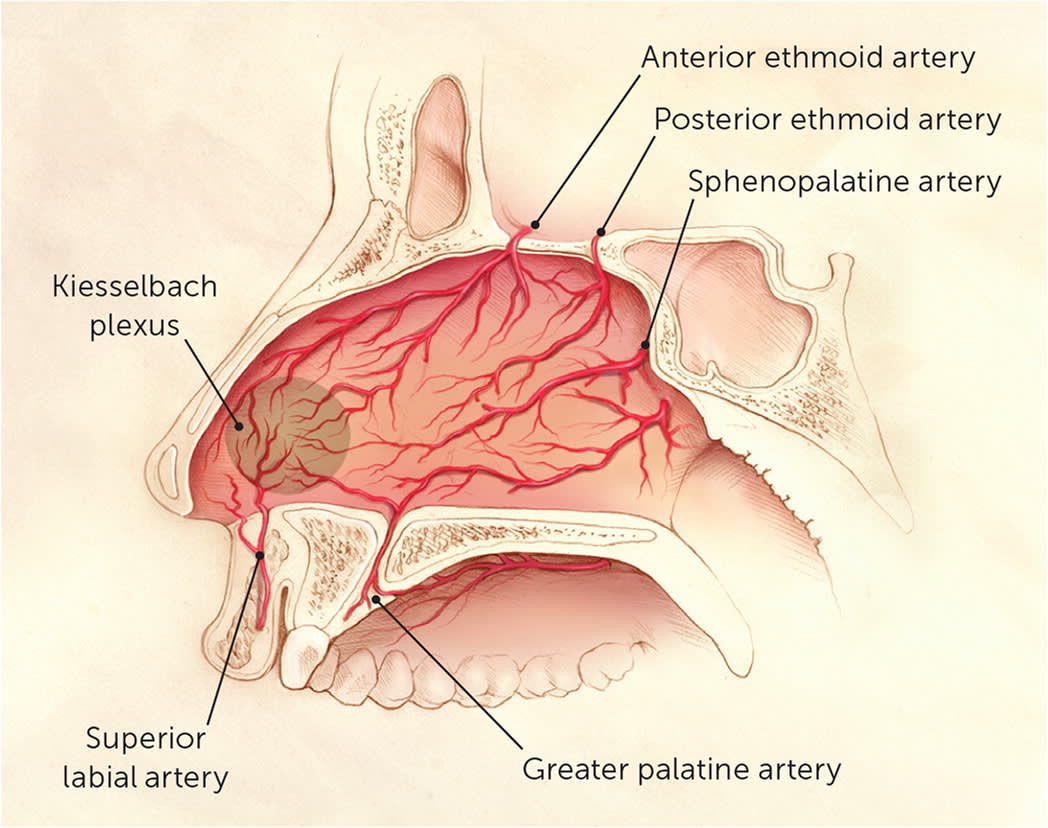
Vascular anatomy of the nasal cavity.
Illustration by Christy Krames
Reprinted with permission from Kucik CJ, Clenney T. Management of epistaxis. Am Fam Physician. 2005;71(2):305.
Posterior epistaxis typically occurs along the nasal septum or lateral nasal wall, and originates from branches of the internal maxillary, sphenopalatine, and descending palatine arteries.3,10 The posterior ethmoid artery provides a small contribution.10 Because hemostasis is more difficult to achieve with posterior bleeding, the distinction between anterior and posterior epistaxis guides management.11
Etiology
A focused history and physical examination identify most causes of epistaxis (Table 1).3,6 At the initial presentation of bleeding, the physician should determine the side of the bleeding as well as inquire about previous bleeding episodes and treatment, comorbid conditions, and medication use.6 The differential diagnosis should include local and systemic etiologies. In children, repeated digital trauma (e.g., nose picking) is the most common cause. There are no known specific conditions or risk factors associated with posterior epistaxis.3,6 It is unclear whether seasonal changes or hypertension has a direct role.4,6,11
TABLE 1. Causes of Epistaxis

| Local | |
| Inflammatory | |
| Chronic sinusitis | |
| Environmental irritants | |
| Granulomatous disease | |
| Pyogenic granuloma | |
| Viral illness | |
| Structural | |
| Septal deviation or perforation | |
| Traumatic | |
| Cocaine use | |
| Foreign body | |
| Nasal fracture | |
| Nasal intubation | |
| Nasal oxygen | |
| Nose picking | |
| Surgical procedure | |
| Topical medications (e.g., intranasal steroids) | |
| Tumors and vascular malformations | |
| Systemic | |
| Anticoagulants | |
| Coagulopathy | |
| Hemophilia | |
| Leukemia | |
| Liver disease | |
| Thrombocytopenia | |
| Vitamin deficiencies (A, C, D, E, K) | |
Physical Examination
The physical examination should begin with assessment of vital signs, mental status, and airway patency. When examining the nose, a nasal speculum and good light source, such as a head-lamp, are useful. The Kiesselbach plexus should be examined first for bleeding, followed by the vestibule, septum, and turbinates. If a bleeding source cannot be identified in these areas, there is concern for posterior bleeding. If bleeding persists after attempts to control anterior bleeding with compression and packing, management of a possible posterior source should be initiated.
Management
ANTERIOR EPISTAXIS
Outpatient management of anterior epistaxis is a stepwise process beginning with conservative measures to control bleeding and moving toward more invasive means to achieve hemostasis. An initial assessment of airway patency is necessary. A brisk bleed can lead to a large amount of blood entering the posterior pharynx, potentially causing airway obstruction. Any concern for airway obstruction should be immediately referred for emergency evaluation.
Once airway patency has been determined, compressive therapy should be applied to stop bleeding in the anterior nasal plexus. Firm pressure is placed on the bilateral nostrils, below the nasal bones, for 10 to 15 minutes without interruption. Simple manual pinching may be used, or a nasal clip can be fashioned using tongue depressors taped together (Figure 2). To aid compressive therapy, direct spray of oxymetazoline (Afrin) or application of cotton soaked in oxymetazoline or epinephrine 1:1,000 may be useful to abate or slow the bleeding.12,13 Clinicians should be aware of the adverse effects of systemic epinephrine absorption, such as elevated blood pressure and tachycardia.
FIGURE 2.

Nasal compressive device using tongue depressors.
After compressive therapy, the nares should be inspected for any sign of continued bleeding. Any hematoma must be evacuated for proper inspection. This can be accomplished through the patient blowing the nose, suction, irrigation, or direct forceps evacuation. Proper lighting is crucial; a headlamp provides adequate lighting and leaves the hands free to maneuver. A nasal speculum is useful to increase the field of vision during inspection.13
The next step is directive therapy. If there continues to be bleeding or a prominent vessel that is suspected to be the source of the bleeding, direct vasoocclusive therapy to the area is warranted. In the outpatient setting, the use of silver nitrate sticks is convenient and effective. Silver nitrate creates a chemical cautery when it comes in contact with a moist mucous membrane. The silver nitrate should be applied in a circumferential pattern around the site of bleeding before it is applied to the bleeding site itself. Brisk bleeding will wash silver nitrate away before cauterization, so relative hemostasis is needed for this approach to be successful.14 Electrical desiccation in the same pattern is also an effective way to control bleeding in the nares.10 Blind cauterization is not recommended, because excessive destruction of the nasal mucosa with silver nitrate or electrical desiccation can lead to ulceration and septal perforation.
If compressive therapy is inadequate and directive therapy is ineffective or impossible because of continued brisk bleeding, topical therapy and nasal packing are the next options. Traditional nasal packing involves placing cotton stripping impregnated with petroleum jelly into the base of the nasal cavity, and layering until the nares are completely compressed (Figure 3).8 This is an effective measure for controlling nasal bleeding, although rebleeding occurs in about 15% of patients.15 Nasal tampons and nasal balloon packing may be easier to use14; however, unless the practice treats a large number of patients with epistaxis, it may not be feasible if these materials are not readily available.
FIGURE 3.
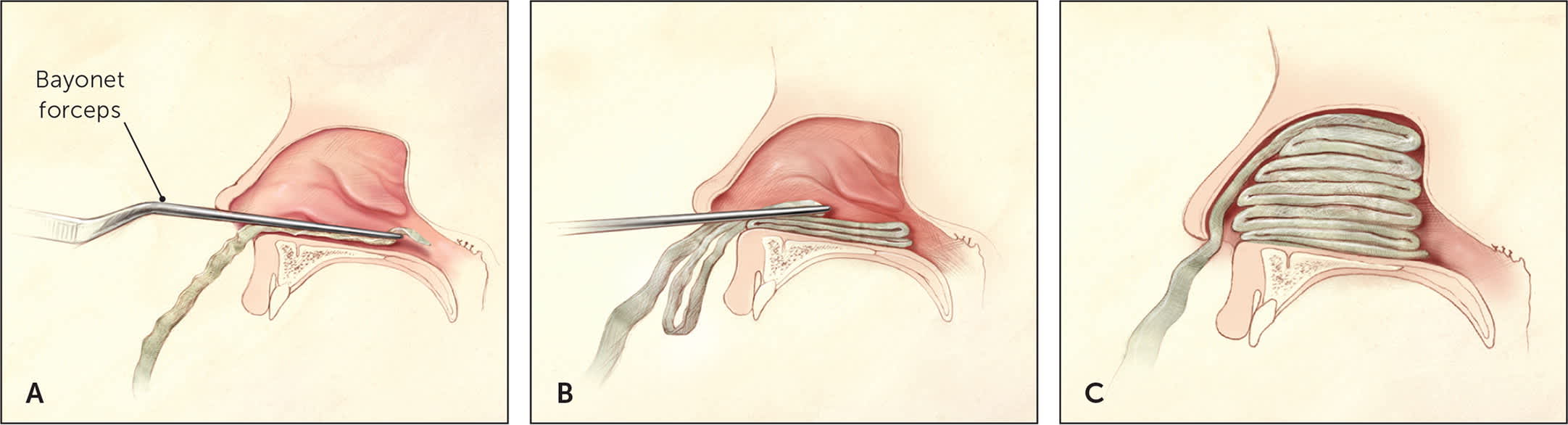
Packing of the anterior nasal cavity using gauze strip impregnated with petroleum jelly. (A) Gauze is gripped with bayonet forceps and inserted into the anterior nasal cavity. (B) With a nasal speculum (not shown) used for exposure, the first packing layer is inserted along the floor of the anterior nasal cavity. Forceps and speculum then are withdrawn. (C) Additional layers of packing are added in an accordion-fold fashion, with the nasal speculum used to hold the positioned layers down while a new layer is inserted. Packing is continued until the anterior nasal cavity is filled.
Illustration by Christy Krames
Reprinted with permission from Kucik CJ, Clenney T. Management of epistaxis. Am Fam Physician. 2005;71(2):309.
Nasal packing should be left in place for 48 hours. The use of oral and topical antibiotics in patients with nasal packing is common to prevent infectious complications such as staphylococcus-induced toxic shock syndrome and sinusitis, but there is little evidence to support antibiotic use.16
Topical hemostatic agents such as Floseal and Surgicel may be effective for managing epistaxis, but are often unavailable in the outpatient setting.14
POSTERIOR EPISTAXIS
Posterior epistaxis is often brisk, and given the location of these vessels, it is usually difficult to visualize the site of bleeding. Compared with anterior epistaxis, patients with posterior epistaxis are more likely to require hospitalization and are twice as likely to require nasal packing.17
As with anterior epistaxis, the physician should evaluate and clear the airway, and provide intravenous access and fluid resuscitation, if needed. Patients with posterior epistaxis generally require referral to an otolaryngologist after stabilization.
Chemical cautery is usually not possible for posterior epistaxis because the source of bleeding is rarely identified.18,19 Newer products that can adhere to an irregular moist surface, such as gelatin-thrombin matrix,20 are still being tested, and there is no evidence to support their blind application.
Posterior nasal packing may be attempted by a physician trained in this procedure. It is up to 70% effective at treating posterior epistaxis when performed by trained physicians21; however, it is not as successful as endoscopic or surgical management, and may be less cost-effective as an initial management technique.20,22,23 Nonetheless, it is a common procedure that may be attempted in the outpatient setting or en route to an emergency department.
Posterior packing is performed using a balloon catheter, Foley catheter, or red rubber catheter with cotton packing. The catheter is passed through the nostril, down the nasopharynx, and into the oropharynx (Figure 4).8 The balloon is inflated with 8 to 10 mL of water and gently retracted until it sits in the posterior choana. If cotton packing is used, the rubber catheter is drawn out of the mouth after it is visualized in the oropharynx. The packing is secured to the end of the catheter and then pulled back through the mouth to sit in the choana. In each case, traction is maintained by clamping the area outside of the nostril, making sure to provide padding between the clamp and the nasal ala to minimize the risk of alar necrosis.21
FIGURE 4.
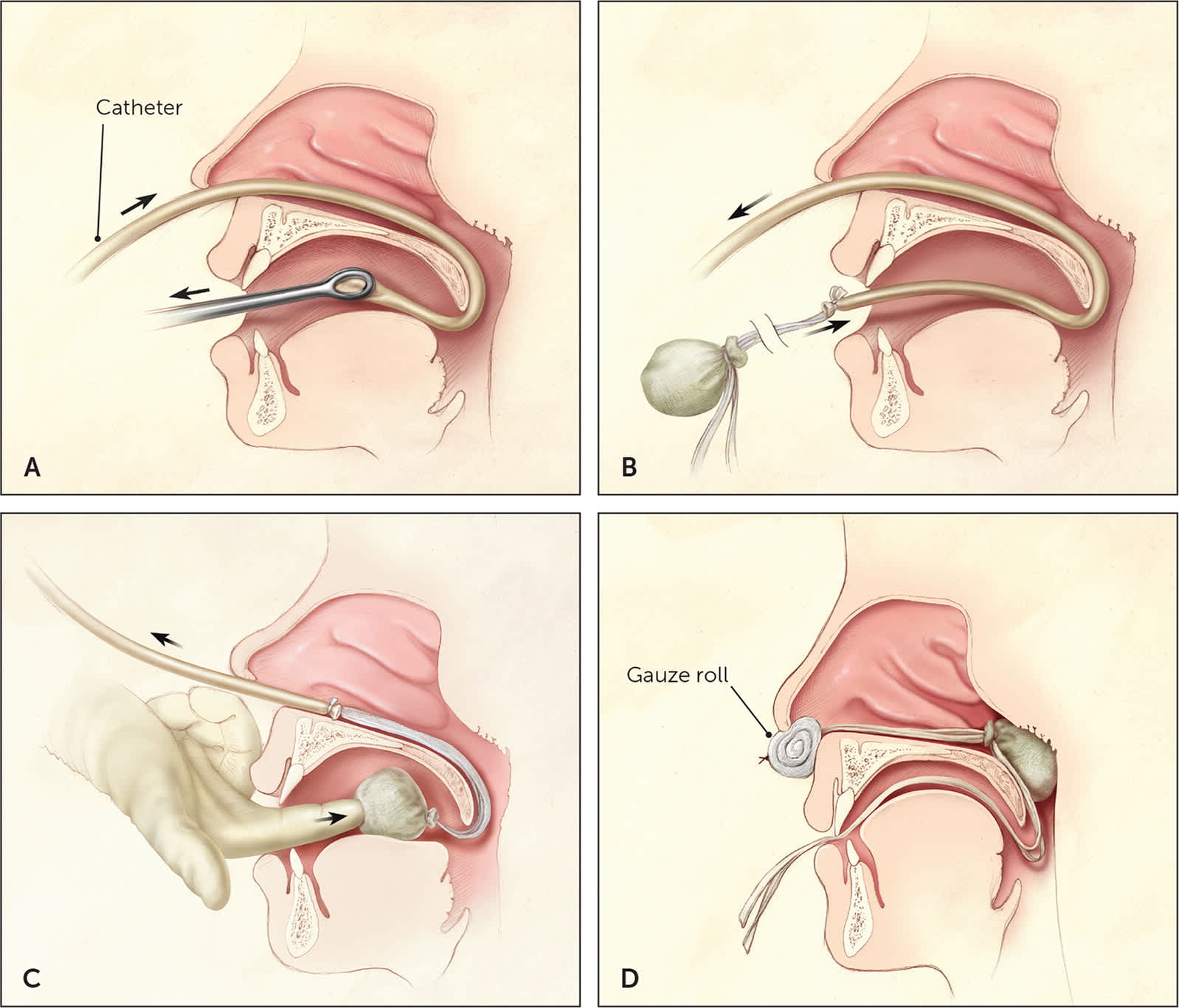
Posterior nasal packing. (A) After adequate anesthesia has been administered, a catheter is passed through the affected nostril and through the nasopharynx, and drawn out the mouth with the aid of ring forceps. (B) A gauze pack is secured to the end of the catheter using umbilical tape or suture material, with long tails left to protrude from the mouth. (C) The gauze pack is guided through the mouth and around the soft palate using a combination of careful traction on the catheter and pushing with a gloved finger. This is the most uncomfortable (and most dangerous) part of the procedure; it should be completed smoothly and with the aid of a bite block (not shown) to protect the physician's finger. (D) The gauze pack should come to rest in the posterior nasal cavity. It is secured in position by maintaining tension on the catheter with a padded clamp or firm gauze roll placed anterior to the nostril. The ties protruding from the mouth, which will be used to remove the pack, are taped to the patient's cheek.
Illustration by Christy Krames
Reprinted with permission from Kucik CJ, Clenney T. Management of epistaxis. Am Fam Physician. 2005;71(2):310.
Posterior packing is often associated with pain, and there is a risk of aspiration if it is dislodged. Patients are commonly monitored in the hospital while packing is in place. There is up to a 50% chance of rebleed with posterior epistaxis.18 Telemetry may be considered given the possibility of a vasovagal reflex, which can cause cardiac abnormalities and respiratory arrest.
If clinicians with appropriate expertise are available, endoscopic artery ligation and endovascular embolization are more effective than packing.20,22,23 Endoscopic treatment may be the best initial treatment, because it is less costly than embolization and more effective than packing.20
This article updates a previous article on this topic by Kucik and Clenney.8
Data Sources: Literature search included the use of medical databases PubMed, Ovid, and Essential Evidence Plus. Keywords used included epistaxis, anterior epistaxis, posterior epistaxis, epistaxis management. Search dates: February 1 to May 15, 2017.

