Clinical Question
Is smectite safe and effective for the treatment of acute infectious diarrhea in children?
Evidence-Based Answer
When compared with placebo, smectite decreases the duration of diarrhea by about a day, with a mean difference (MD) of 24.4 fewer hours of diarrhea (95% confidence interval [CI], 17.9 to 30.9 hours). Smectite also decreases stool output by 11.4 g per kg (95% CI, 0.8 to 22 g per kg). Children using smectite are more likely to have resolution of diarrhea by day 3 (number needed to treat = 2.7). There is no difference in the number of children requiring intravenous rehydration therapy or hospitalization. No serious adverse effects or deaths have been reported in children using smectite.1 (Strength of Recommendation: C, based on consensus, disease-oriented evidence, usual practice, expert opinion, or case series.)
Practice Pointers
Acute diarrhea is the passage of three or more loose stools per day.2 In the United States, diarrhea accounts for 1% of deaths in patients younger than five years.3 Diarrhea is commonly caused by infection from contaminated food and water, or transmitted via fecal-oral route.3 Smectite is a natural, medicinal clay formed from sheets of aluminum and magnesium silicate.1 It is an oral adjuvant for diarrhea that is used outside of—and not commonly available in—the United States.1 It typically comes as a powder that is dissolved in water and is available without a prescription. Smectite is proposed to reduce gut inflammation, reduce toxin penetration by acting as a barrier, and increase water absorption.1
This Cochrane review included 18 trials with 2,616 children. Studies were conducted in high-income and low- to middle-income countries (the United States was not represented) and included hospital and ambulatory settings. Most studies involved infants and children one to 24 months of age, although nine studies also included children up to 12 years of age. Both breastfed and nonbreastfed children were represented. Rotavirus was the most commonly reported etiology of diarrhea. Smectite administration varied from 1 to 6 g per dose, at intervals of one to four times per day, with varying durations (i.e., from three to six days or until recovery).
The primary outcome for the Cochrane review was duration of diarrhea. Fifteen studies (2,209 participants) reported duration with varying definitions for resolution (i.e., time to last loose stool, time to first formed stool, time to first soft or formed stool, and time to normalization of stool). The mean duration of diarrhea in the smectite group was 24.4 fewer hours than in the control group (95% CI, 17.9 to 30.9). More participants in the smectite group achieved resolution of diarrhea by day 3 (number needed to treat = 2.7; 95% CI, 1.5 to 9.8).
The secondary outcomes evaluated were stool frequency, volume of stool output, the need for hospitalization, and the need for intravenous access for rehydration. Children stooled less frequently when treated with smectite (MD = 1.3 fewer episodes per day; 95% CI, 0.4 to 2.3 fewer episodes) and, when they did, had decreased stool output (MD = 11.4 g per kg; 95% CI, 0.8 to 21.9 g per kg). There was no difference in the need for hospitalization or the need for intravenous access for rehydration.
No serious adverse effects or deaths were reported in any of the studies. Constipation was the most commonly reported adverse effect of smectite, although there were few events.
Current National Institute for Health and Care Excellence guidelines focus on preventing and managing dehydration for children with diarrhea and vomiting and specifically recommend against using antidiarrheal medications.4 Smectite could be of benefit in countries with limited availability of oral or intravenous rehydration therapy, especially in patients with preexisting malnutrition.
SUMMARY TABLE: SMECTITE VS. CONTROL FOR ACUTE INFECTIOUS DIARRHEA IN CHILDREN
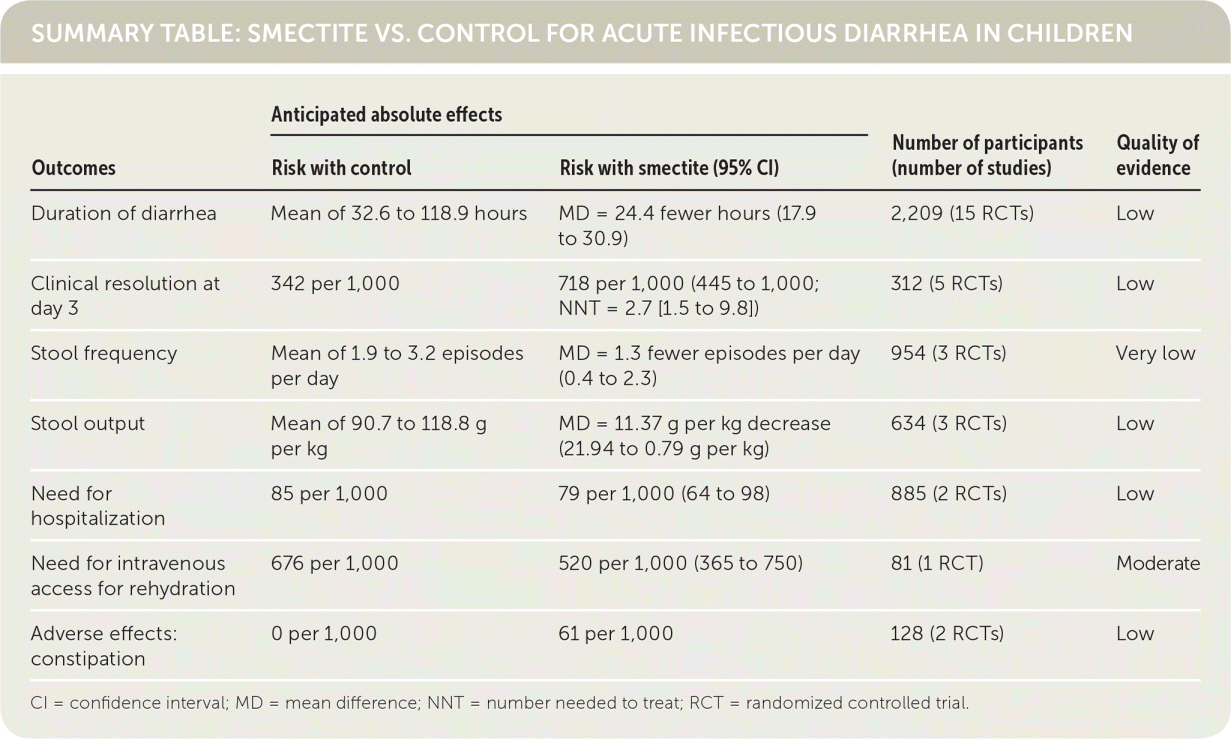
| Outcomes | Anticipated absolute effects | Number of participants (number of studies) | Quality of evidence | |
|---|---|---|---|---|
| Risk with control | Risk with smectite (95% CI) | |||
| Duration of diarrhea | Mean of 32.6 to 118.9 hours | MD = 24.4 fewer hours (17.9 to 30.9) | 2,209 (15 RCTs) | Low |
| Clinical resolution at day 3 | 342 per 1,000 | 718 per 1,000 (445 to 1,000; NNT = 2.7 [1.5 to 9.8]) | 312 (5 RCTs) | Low |
| Stool frequency | Mean of 1.9 to 3.2 episodes per day | MD = 1.3 fewer episodes per day (0.4 to 2.3) | 954 (3 RCTs) | Very low |
| Stool output | Mean of 90.7 to 118.8 g per kg | MD = 11.37 g per kg decrease (21.94 to 0.79 g per kg) | 634 (3 RCTs) | Low |
| Need for hospitalization | 85 per 1,000 | 79 per 1,000 (64 to 98) | 885 (2 RCTs) | Low |
| Need for intravenous access for rehydration | 676 per 1,000 | 520 per 1,000 (365 to 750) | 81 (1 RCT) | Moderate |
| Adverse effects: constipation | 0 per 1,000 | 61 per 1,000 | 128 (2 RCTs) | Low |
CI = confidence interval; MD = mean difference; NNT = number needed to treat; RCT = randomized controlled trial.
The practice recommendations in this activity are available at http://www.cochrane.org/CD011526.
Editor's Note: The numbers needed to treat and confidence intervals reported in this Cochrane for Clinicians were calculated by the authors based on raw data provided in the original Cochrane review.
The views expressed in this article are those of the authors and do not necessarily reflect the official policy or position of the Department of the Navy, Uniformed Services University of the Health Sciences, Department of Defense, or the U.S. government.
We are military service members. This work was prepared as part of our official duties. Title 17 U.S.C. 105 provides that “Copyright protection under this title is not available for any work of the United States Government.” Title 17 U.S.C. 101 defines United States Government work as a work prepared by a military service member or employee of the United States Government as part of that person's official duties.

