Clinical Question
Are progestogens a safe and effective treatment option for patients with threatened miscarriage?
Evidence-Based Answer
Progestogens reduce the risk of miscarriage when compared with placebo in patients with threatened miscarriage (number needed to treat [NNT] = 10.) Use of progestogens poses no significant risks to mother or baby.1 (Strength of Recommendation: A, based on consistent, good-quality patient-oriented evidence.)
Practice Pointers
Miscarriage occurs in 15% to 20% of pregnancies and is associated with significant physical and psychological morbidity. Threatened miscarriage is defined as any vaginal bleeding, with or without abdominal pain, with a closed cervix and a viable fetus inside the uterine cavity. Effective treatment options to increase the chance of a successful pregnancy are lacking. Bed rest, pelvic rest, vitamins, uterine relaxants, and administration of beta subunit of human chorionic gonadotropin are not recommended.2 Progesterone has physiologic roles in maintaining pregnancy by inducing changes in the endometrium, suppressing uterine contractions, and modulating the maternal immune system. Progestogens—medications that mimic the activity of progesterone—are therefore a rational therapeutic option to treat threatened miscarriage. Early studies had less rigorous methodology, so the authors of this Cochrane review sought to evaluate progestogen using newer data.1
This Cochrane review included seven randomized controlled trials and 696 patients from lower middle- to high-income countries, although none of the trials took place in North America.1 Three of these trials investigated oral progestogens and four trials evaluated vaginal progesterone. The authors assessed the body of evidence to be of moderate quality for the primary outcome of miscarriage vs. successful pregnancy, mainly because of limitations in study design. For example, only three of the seven studies were deemed to be at low risk of selection bias. The authors analyzed only studies that included pregnant women at 23 weeks of gestation or less, presenting with a concern of threatened miscarriage, and in which the viability of the pregnancy had been confirmed before starting treatment. Secondary outcomes were maternal (pain, preterm birth) and fetal (preterm birth, stillbirth, neonatal death, congenital abnormalities, low birth weight, or any other adverse neonatal outcomes reported). Maternal pain was reported in two trials; one double-blind, randomized, placebo-controlled trial included 50 patients and demonstrated significantly decreased maternal pain with use of progesterone. The other single-blind, randomized trial included 60 patients and demonstrated no pain relief with progesterone vs. no treatment.
SUMMARY TABLE Outcomes of Progesterone vs. Placebo or No Treatment for Threatened Miscarriage
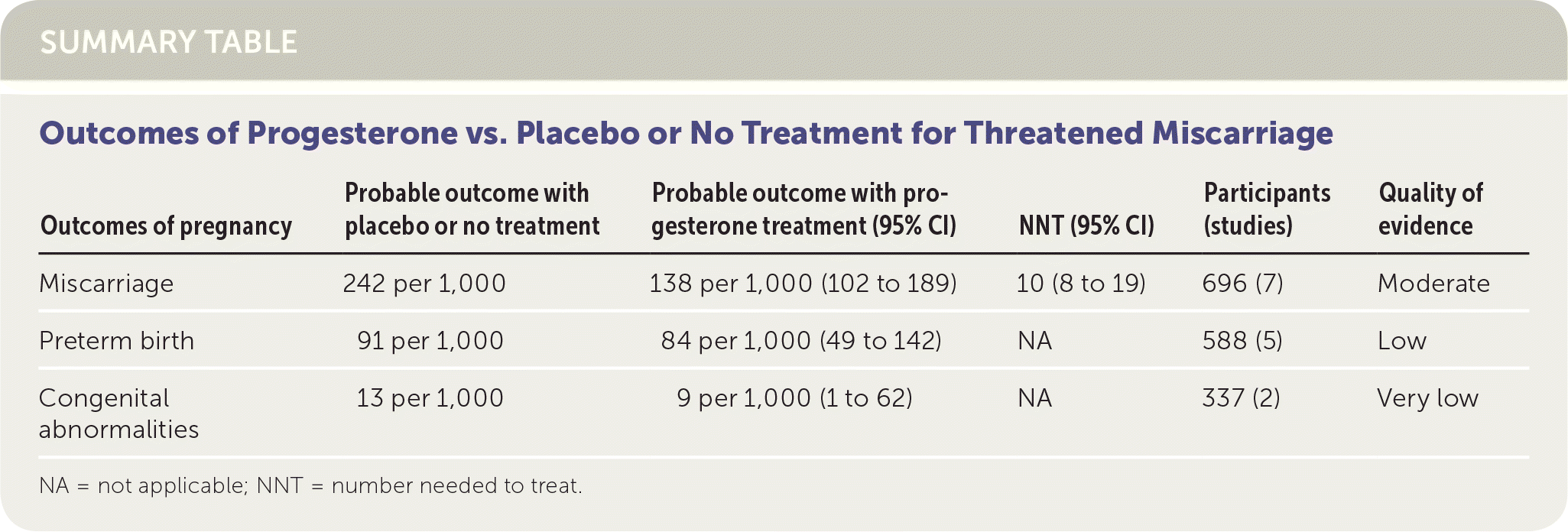
| Outcomes of pregnancy | Probable outcome with placebo or no treatment | Probable outcome with progesterone treatment (95% CI) | NNT (95% CI) | Participants (studies) | Quality of evidence |
|---|---|---|---|---|---|
| Miscarriage | 242 per 1,000 | 138 per 1,000 (102 to 189) | 10 (8 to 19) | 696 (7) | Moderate |
| Preterm birth | 91 per 1,000 | 84 per 1,000 (49 to 142) | NA | 588 (5) | Low |
| Congenital abnormalities | 13 per 1,000 | 9 per 1,000 (1 to 62) | NA | 337 (2) | Very low |
NA = not applicable; NNT = number needed to treat.
Although progestogens decreased the risk of miscarriage overall, this was significant only with orally—not vaginally—administered therapies. Women receiving any progestogen therapy had a lower risk of miscarriage than those receiving placebo (absolute risk reduction = 10.4%; 95% CI, 5.3% to 14.0%; NNT = 10). Two of the three trials employing the oral treatment route used dydrogesterone, which is not available in the United States. The third trial (n = 60) used oral micronized progesterone, 400 mg daily for four weeks, starting with the diagnosis of threatened miscarriage and continuing even if vaginal bleeding stopped. These demonstrated consistent effects and reached statistical significance when the data were pooled.
Progestogens as an intervention did not cause any significant harms. When progestogens were compared with placebo, there were no differences in pregnancy-induced hypertension, antepartum hemorrhage, stillbirth, low birth weight, or respiratory distress syndrome. No significant differences were found for preterm birth or congenital abnormalities, although these analyses were limited by low- and very low-quality evidence, respectively. Adverse effects of progestogens are usually mild and include breast tenderness, bloating, and headache. Progestogens should be used with caution in patients who have cardiovascular disease or impaired liver function, including cholestasis.
A guideline from the National Institute for Health and Care Excellence is currently in development.3 The guideline supports offering women treatment while counseling them that the evidence is not strong. More research is needed to define the most effective preparation, dosing, route and frequency of administration, and duration of therapy.
The practice recommendations in this activity are available at http://www.cochrane.org/CD005943.
Editor's Note: The numbers needed to treat and absolute risk reductions reported in this Cochrane for Clinicians were calculated by the author based on raw data provided in the original Cochrane review.
The opinions and assertions contained herein are the private views of the author and do not reflect the official policy or position of the U.S. Air Force, the Department of Defense, or the U.S. government.

