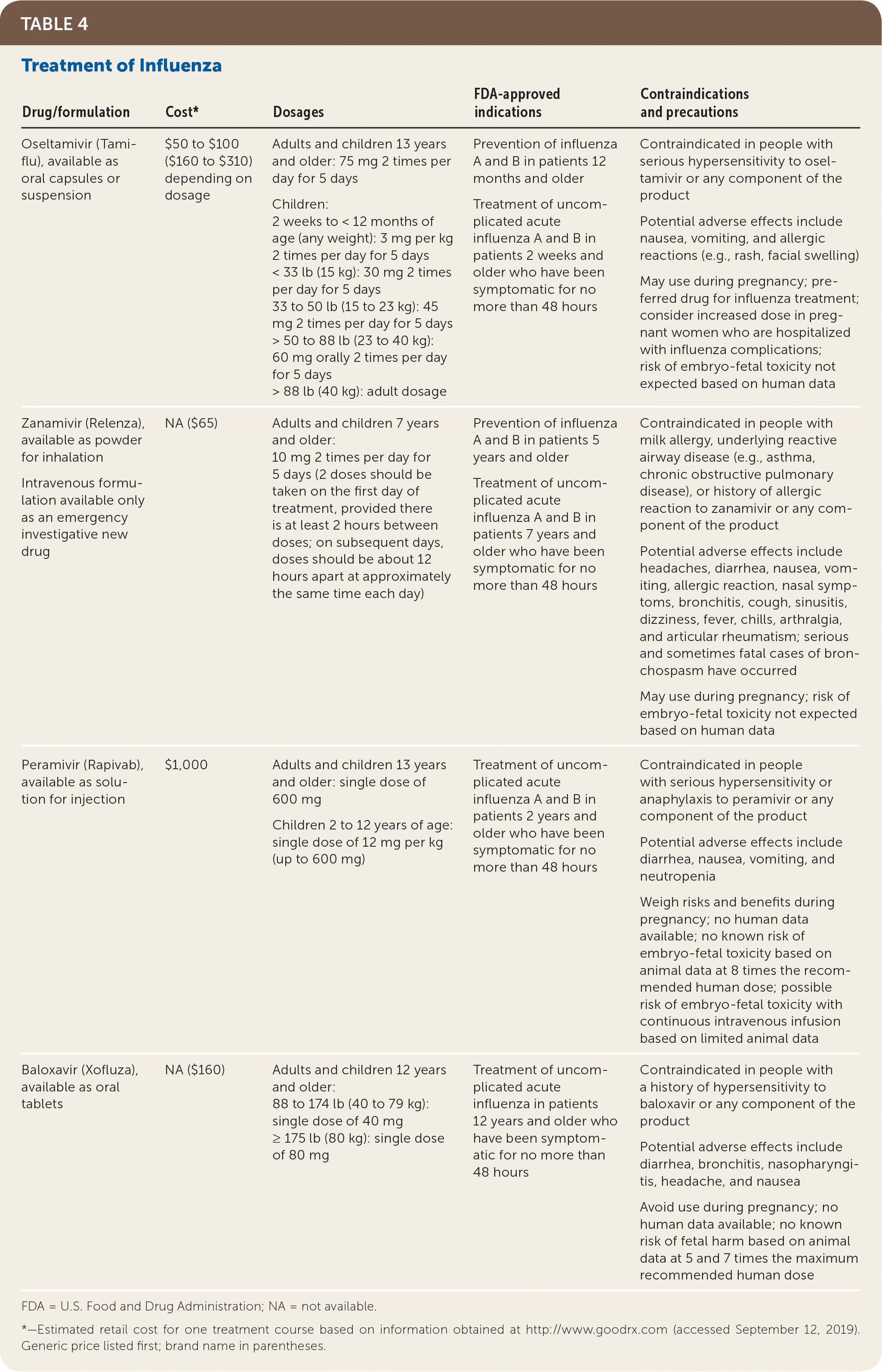
| Drug/formulation | Cost* | Dosages | FDA-approved indications | Contraindications and precautions |
|---|---|---|---|---|
| Oseltamivir (Tamiflu), available as oral capsules or suspension | $50 to $100 ($160 to $310) depending on dosage | Adults and children 13 years and older: 75 mg 2 times per day for 5 days Children: 2 weeks to < 12 months of age (any weight): 3 mg per kg 2 times per day for 5 days < 33 lb (15 kg): 30 mg 2 times per day for 5 days 33 to 50 lb (15 to 23 kg): 45 mg 2 times per day for 5 days > 50 to 88 lb (23 to 40 kg): 60 mg orally 2 times per day for 5 days > 88 lb (40 kg): adult dosage | Prevention of influenza A and B in patients 12 months and older Treatment of uncomplicated acute influenza A and B in patients 2 weeks and older who have been symptomatic for no more than 48 hours | Contraindicated in people with serious hypersensitivity to oseltamivir or any component of the product Potential adverse effects include nausea, vomiting, and allergic reactions (e.g., rash, facial swelling) May use during pregnancy; preferred drug for influenza treatment; consider increased dose in pregnant women who are hospitalized with influenza complications; risk of embryo-fetal toxicity not expected based on human data |
| Zanamivir (Relenza), available as powder for inhalation Intravenous formulation available only as an emergency investigative new drug | NA ($65) | Adults and children 7 years and older: 10 mg 2 times per day for 5 days (2 doses should be taken on the first day of treatment, provided there is at least 2 hours between doses; on subsequent days, doses should be about 12 hours apart at approximately the same time each day) | Prevention of influenza A and B in patients 5 years and older Treatment of uncomplicated acute influenza A and B in patients 7 years and older who have been symptomatic for no more than 48 hours | Contraindicated in people with milk allergy, underlying reactive airway disease (e.g., asthma, chronic obstructive pulmonary disease), or history of allergic reaction to zanamivir or any component of the product Potential adverse effects include headaches, diarrhea, nausea, vomiting, allergic reaction, nasal symptoms, bronchitis, cough, sinusitis, dizziness, fever, chills, arthralgia, and articular rheumatism; serious and sometimes fatal cases of bronchospasm have occurred May use during pregnancy; risk of embryo-fetal toxicity not expected based on human data |
| Peramivir (Rapivab), available as solution for injection | $1,000 | Adults and children 13 years and older: single dose of 600 mg Children 2 to 12 years of age: single dose of 12 mg per kg (up to 600 mg) | Treatment of uncomplicated acute influenza A and B in patients 2 years and older who have been symptomatic for no more than 48 hours | Contraindicated in people with serious hypersensitivity or anaphylaxis to peramivir or any component of the product Potential adverse effects include diarrhea, nausea, vomiting, and neutropenia Weigh risks and benefits during pregnancy; no human data available; no known risk of embryo-fetal toxicity based on animal data at 8 times the recommended human dose; possible risk of embryo-fetal toxicity with continuous intravenous infusion based on limited animal data |
| Baloxavir (Xofluza), available as oral tablets | NA ($160) | Adults and children 12 years and older: 88 to 174 lb (40 to 79 kg): single dose of 40 mg ≥ 175 lb (80 kg): single dose of 80 mg | Treatment of uncomplicated acute influenza in patients 12 years and older who have been symptomatic for no more than 48 hours | Contraindicated in people with a history of hypersensitivity to baloxavir or any component of the product Potential adverse effects include diarrhea, bronchitis, nasopharyngitis, headache, and nausea Avoid use during pregnancy; no human data available; no known risk of fetal harm based on animal data at 5 and 7 times the maximum recommended human dose |