A 39-year-old man with a history of mild intermittent asthma and limited medical care presented for evaluation of shortness of breath and bilateral lower extremity edema. He reported three weeks of symptoms, including cough, congestion, rhinorrhea, and subjective fevers. Over that time, he had increasing shortness of breath, dyspnea on exertion, and an inability to button his work pants. His social history included sexual activity with multiple female partners and weekly methamphetamine use through a pipe. He reported no intravenous drug use or alcohol use.
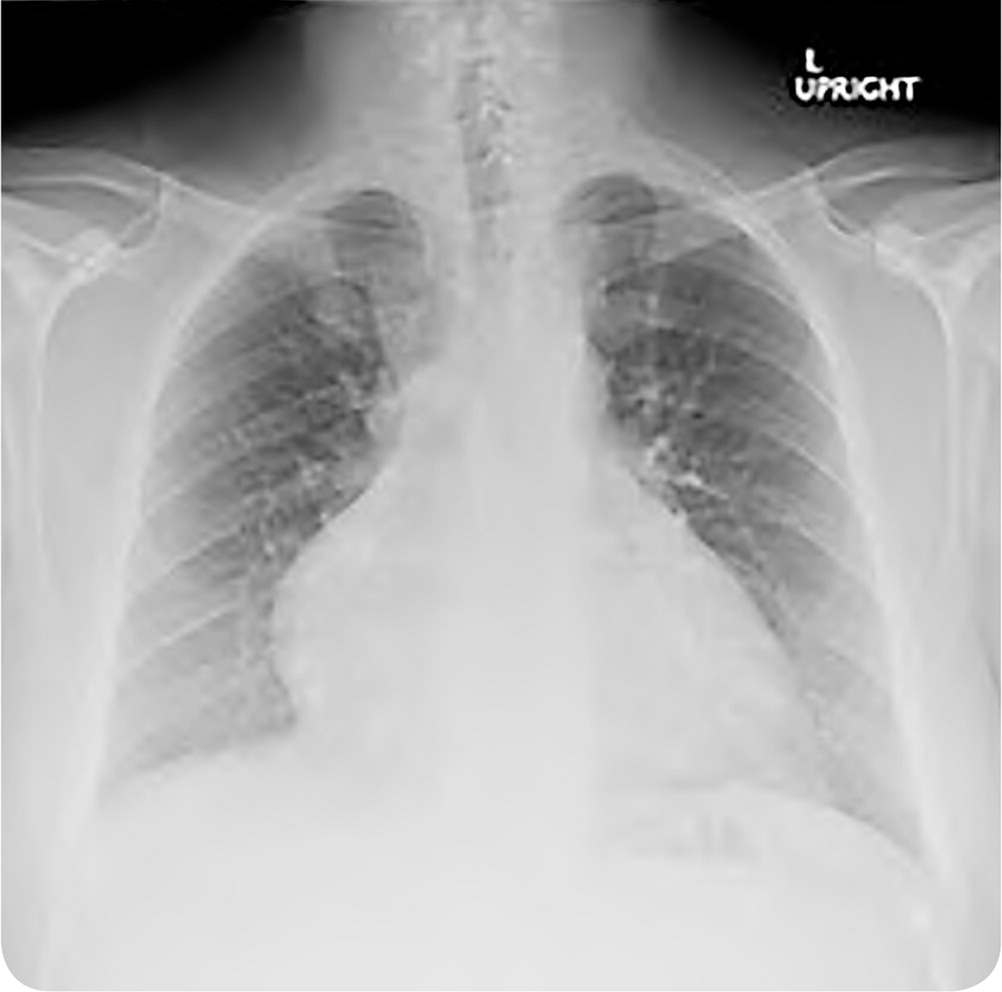
Physical examination revealed jugular venous distention to the level of the mandible, normal findings on a cardiac examination, crackles at the lung bases, and 3+ pitting edema in the bilateral lower extremities to the midthigh. An electrocardiogram showed normal sinus rhythm, prolonged corrected QT interval, and no evidence of ischemia. Serum troponins were not elevated. A transthoracic echocardiogram revealed severely reduced left ventricular systolic function with an ejection fraction of 21%. The left ventricle showed global hypokinesis. Chest radiography was performed (Figure 1).
FIGURE 1
Question
Based on the patient's history, physical examination, and preliminary study findings, which one of the following is the most likely diagnosis?
A. HIV-associated cardiomyopathy.
B. Hypertrophic cardiomyopathy.
C. Ischemic cardiomyopathy.
D. Methamphetamine-associated cardiomyopathy.
E. Viral myocarditis.
Discussion
The answer is D: methamphetamine-associated cardiomyopathy. The chest radiograph demonstrates cardiomegaly with associated pulmonary edema, which is concerning for dilated cardiomyopathy. Cardiomyopathy with a markedly reduced ejection fraction in a young methamphetamine user is highly suggestive of methamphetamine-associated cardiomyopathy. Patients with methamphetamine-associated cardiomyopathy have a significantly reduced ejection fraction, with one study demonstrating a mean left ventricular ejection fraction of 19%.1–3 Echocardiography demonstrates global hypokinesia from the systemic effects of catecholamines, as well as the direct toxic effects of methamphetamine.4 The prognosis for recovery is good but is dependent on cessation of methamphetamine use.1,5 One study reported that the ejection fraction returned to normal within six weeks in one-third of patients who abstained from methamphetamine use.1
Methamphetamine, which is known by street names such as crank, crystal, ice, speed, and go, is one of the most common illicit substances worldwide. It is structurally similar to amphetamine and indirectly increases the release of dopamine, norepinephrine, epinephrine, and serotonin. This results in euphoria, increased alertness, and decreased appetite. Palpitations and cardiac arrhythmias are common, with 27% of patients exhibiting a prolonged corrected QT interval on electrocardiography.6 Prolonged methamphetamine use can lead to myocardial infarction, as well as cardiomyopathy.
Patients with HIV can develop cardiomyopathy because of the increased risk of ischemic disease, myocarditis, altered immune response, or toxic effects of medications. In resource-rich areas with access to antiretroviral therapy, patients with HIV more commonly develop traditional coronary artery disease. Although this patient is at risk of HIV infection, his young age and significantly reduced ejection fraction are more suggestive of methamphetamine-associated cardiomyopathy.
Hypertrophic cardiomyopathy is an autosomal dominant genetic disorder affecting cardiac muscle, resulting in left ventricular hypertrophy and outflow obstruction. The presentation ranges from chest pain and symptoms of heart failure to dangerous arrhythmias and sudden cardiac death. A crescendo-decrescendo systolic murmur classically changes with the position of the patient.
Ischemic cardiomyopathy is the most common cause of heart failure in the United States.7 It generally affects patients with known cardiac disease or traditional cardiac risk factors. An echocardiogram may show regional wall motion abnormalities, although global hypokinesia may be seen in instances of multivessel disease.
Viral myocarditis is a common cause of acute heart failure following a respiratory or gastrointestinal tract infection. Patients may present with chest pain and symptoms of new-onset or worsening heart failure. ST-T wave changes and cardiac biomarker elevations may occur. Although this patient had a recent respiratory tract illness, his lack of chest pain, electrocardiogram changes, and elevation in troponin level makes viral myocarditis less likely.
SUMMARY TABLE
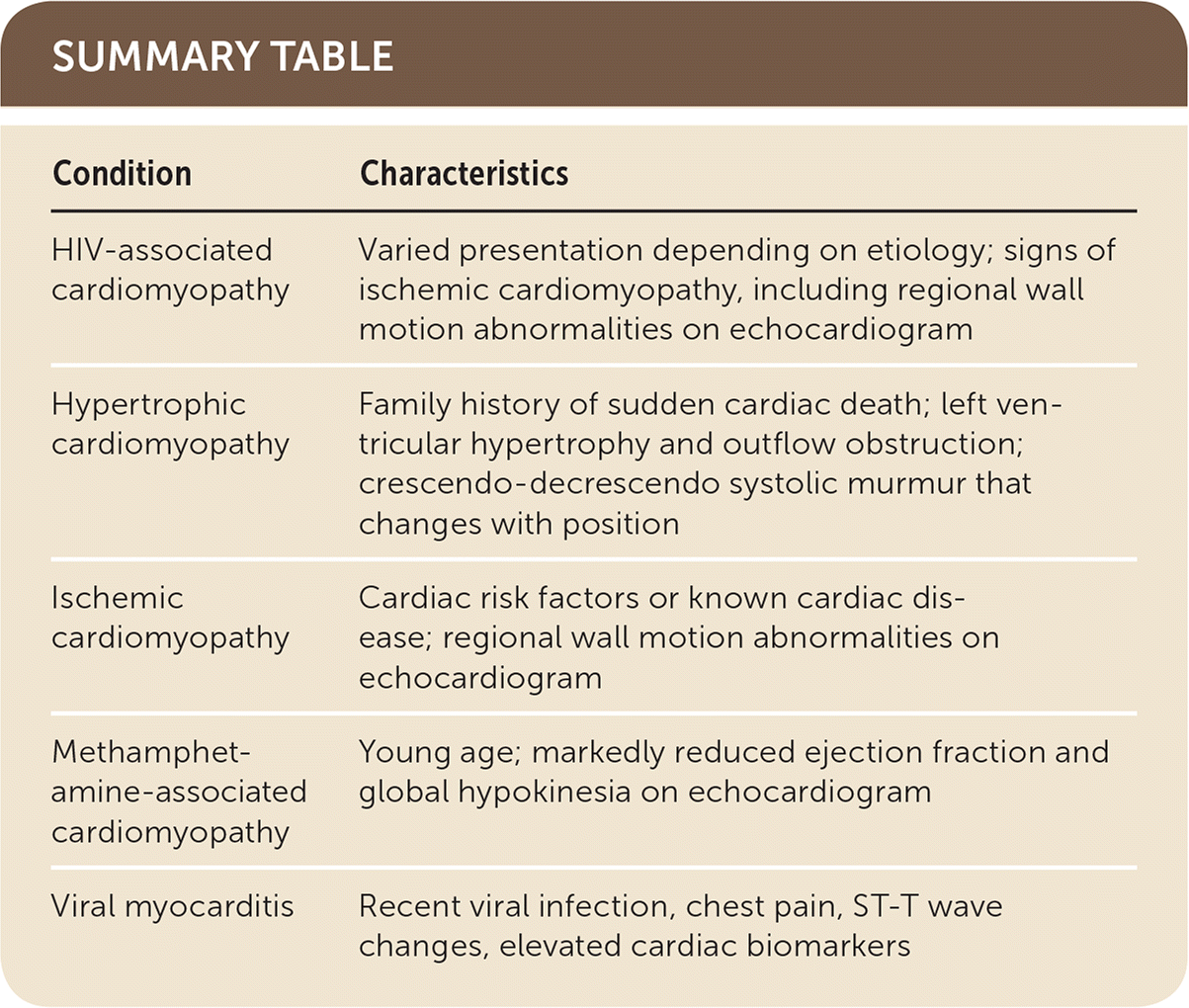
| Condition | Characteristics |
|---|---|
| HIV-associated cardiomyopathy | Varied presentation depending on etiology; signs of ischemic cardiomyopathy, including regional wall motion abnormalities on echocardiogram |
| Hypertrophic cardiomyopathy | Family history of sudden cardiac death; left ventricular hypertrophy and outflow obstruction; crescendo-decrescendo systolic murmur that changes with position |
| Ischemic cardiomyopathy | Cardiac risk factors or known cardiac disease; regional wall motion abnormalities on echocardiogram |
| Methamphetamine-associated cardiomyopathy | Young age; markedly reduced ejection fraction and global hypokinesia on echocardiogram |
| Viral myocarditis | Recent viral infection, chest pain, ST-T wave changes, elevated cardiac biomarkers |

