Guidelines published in 2016 provide a revised definition of sepsis: life-threatening organ dysfunction caused by a dysregulated host response to infection. The guidelines define septic shock as sepsis with circulatory, cellular, and metabolic dysfunction that is associated with a higher risk of mortality. The measurement of serum lactate has been incorporated into the latest septic shock definition. The guidelines recommend the Sequential Organ Failure Assessment (original and quick versions) as an important tool for early diagnosis. Respiratory, gastrointestinal, genitourinary, and skin and soft tissue infections are the most common sources of sepsis. Pneumonia is the most common cause of sepsis. Although many patients with sepsis have fever, the clinical manifestation can be subtle, particularly in older patients and those who are immunocompromised. Initial evaluation of patients with suspected sepsis includes basic laboratory tests, cultures, imaging studies as indicated, and sepsis biomarkers such as procalcitonin and lactate levels. Fluid resuscitation is the priority in early management, including administering an intravenous crystalloid at 30 mL per kg within the first three hours. Antimicrobial therapy should also be initiated early. Most research indicates that antimicrobial therapy should be started within three hours of presentation. The latest guidelines recommend starting antimicrobials within one hour, but this is controversial. Vasopressor therapy is indicated if hypotension persists despite fluid administration. Future trials of sepsis management are focusing on improving long-term rates of readmission and death, physical disability, cognitive impairment, and quality of life.
Sepsis is a substantial global health burden and is the leading cause of death among adults in intensive care units (ICUs).1 It affects more than 900,000 people annually in the United States, with an incidence of 535 cases per 100,000 person-years.2 Medical advances over the past decade, standardized protocols, and physician awareness have significantly improved survival, but mortality rates remain between 20% and 36%, with approximately 270,000 deaths annually in the United States.3–5
WHAT'S NEW ON THIS TOPIC
Sepsis
2016 guidelines provide a new definition for sepsis: a life-threatening organ dysfunction caused by a dysregulated host response to infection. The guidelines define septic shock as sepsis with circulatory, cellular, and metabolic dysfunction that is associated with a higher risk of mortality; the presence of hypotension is no longer required.*
Medical advances over the past decade, standardized protocols, and physician awareness have significantly improved survival in patients with sepsis, but mortality rates remain between 20% and 36%, with approximately 270,000 deaths annually in the United States.
Lactate-guided resuscitation (i.e., measuring lactate every four to six hours until levels have normalized) reduces overall mortality compared with no lactate monitoring.
SIRS = systemic inflammatory response syndrome; SOFA = Sequential Organ Failure Assessment.
*—The 2016 guidelines include the SOFA and quick SOFA to aid in diagnosis. Online calculators are available at https://www.mdcalc.com/sequential-organ-failure-assessment-sofa-score and https://www.mdcalc.com/qsofa-quick-sofa-score-sepsis. Although the SIRS criteria (https://www.mdcalc.com/sirs-sepsis-septic-shock-criteria) are no longer endorsed in the guidelines, they still have a role in the identification of acute infection.
SORT: KEY RECOMMENDATIONS FOR PRACTICE
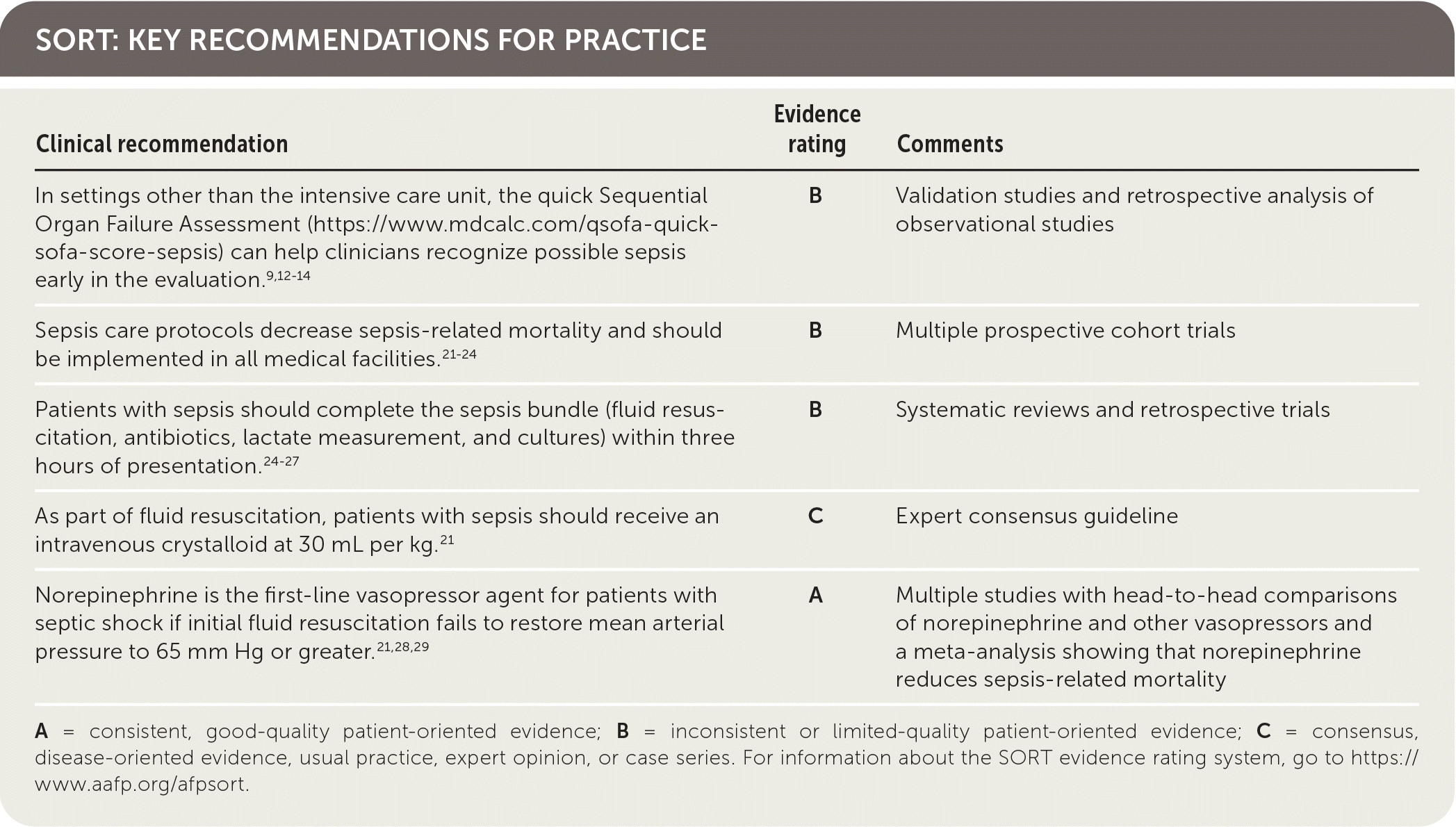
A = consistent, good-quality patient-oriented evidence; B = inconsistent or limited-quality patient-oriented evidence; C = consensus, disease-oriented evidence, usual practice, expert opinion, or case series. For information about the SORT evidence rating system, go to https://www.aafp.org/afpsort.
Of patients with sepsis, 80% are initially treated in an emergency department, and the remainder develop sepsis during hospitalization for other conditions.5 Major risk factors for developing sepsis are age of 65 years or older, malnutrition, chronic illness, immunosuppression, recent surgery or hospitalization, and indwelling devices.3 Approximately one-third of sepsis cases occur in the postoperative period.6
Although an increasing number of patients admitted for sepsis become well enough to be discharged from the hospital, these patients have higher rates of readmission and of death within 12 months and significantly reduced physical and cognitive function compared with matched controls.7
Definition
The Third International Consensus Definitions for Sepsis and Septic Shock (Sepsis-3) were published by the European Society of Intensive Care Medicine and the Society of Critical Care Medicine and incorporated into the Surviving Sepsis Campaign (SSC) International guidelines in 2016.8,9 The terminology was simplified, and sepsis and septic shock are now the only recognized terms.8,9 Sepsis is now defined as a life-threatening organ dysfunction caused by a dysregulated host response to infection. Septic shock is defined as sepsis with circulatory, cellular, and metabolic dysfunction that is associated with a higher risk of mortality.8 Previously, septic shock was identified by the presence of hypotension. It is now recognized that hypotension can be a late manifestation, and tissue hypoperfusion precedes hypotension. Lactate level, an indirect marker of tissue perfusion, has been incorporated in the diagnosis of septic shock in addition to the need for vasopressor therapy required to maintain mean arterial pressure of greater than 65 mm Hg. In this article, use of the term sepsis includes both sepsis and septic shock unless otherwise specified.
SIRS CRITERIA
The systemic inflammatory response syndrome (SIRS) criteria (https://www.mdcalc.com/sirs-sepsis-septic-shock-criteria) are no longer part of Sepsis-3. However, SIRS criteria still have a role in the identification of acute infections.
SIRS is defined as the presence of at least two of the following four criteria: temperature greater than 100.4°F (38°C) or less than 96.8°F (36°C); heart rate greater than 90 beats per minute; respiratory rate greater than 20 breaths per minute or partial pressure of carbon dioxide less than 32 mm Hg; and white blood cell count greater than 12,000 per μL (12 × 109 per L), less than 4,000 per μL (4 × 109 per L), or greater than 10% immature forms.
The overall sensitivity of the SIRS criteria for detecting sepsis is only about 50% to 60%, and one in eight patients admitted to the ICU with sepsis does not meet SIRS criteria.10,11 A limitation of the SIRS criteria is that SIRS may be present with noninfectious conditions such as autoimmune disorders, vasculitis, pancreatitis, burns, trauma, or recent surgery.
SOFA AND qSOFA
Sepsis-3 includes the full Sequential Organ Failure Assessment (SOFA) and a quick version (qSOFA) to aid in diagnosis. The qSOFA (https://www.mdcalc.com/qsofa-quick-sofa-score-sepsis) was designed to help clinicians recognize possible sepsis in settings other than the ICU.9,12–14 Sepsis should be suspected in patients meeting at least two of the three qSOFA criteria: respiratory rate of 22 breaths per minute or greater, altered mental status, and systolic blood pressure of 100 mm Hg or less. These patients should undergo additional evaluation.
The qSOFA is limited by its low sensitivity (approximately 50%) and because it typically identifies patients who present late in the course of sepsis.14 Nonetheless, Sepsis-3 includes qSOFA because it requires no laboratory testing and can be rapidly administered.12,13 Until additional diagnostic tools emerge, sepsis should be suspected in patients with a positive score on the SIRS criteria or qSOFA.15
The full SOFA (https://www.mdcalc.com/sequential-organ-failure-assessment-sofa-score) has been endorsed by the Society of Critical Care Medicine12 and is used in the ICU to predict in-hospital mortality. It assesses the severity of dysfunction for six organ systems in critically ill patients. The score is calculated at the time of ICU admission and then every 48 hours. An increase in the SOFA score by at least two points from baseline (assumed to be 0 before sepsis in patients with unknown preexisting organ dysfunction) indicates acute organ dysfunction with a presumptive diagnosis of sepsis and an increase in mortality rate of greater than 20%.13,16
Etiology
Respiratory, gastrointestinal, genitourinary, and skin or soft tissue infections are the most common sources of sepsis, accounting for more than 80% of all sepsis cases.17 Indwelling devices, endocarditis, and meningitis or encephalitis each account for 1% of sepsis cases.18 Pneumonia is the most common cause of sepsis.19
Bacterial microbes (gram-negative [62%] or gram-positive [47%]) are the most common causative organisms for sepsis.18 Some patients with sepsis are infected with multiple microbial organisms. A small number of patients with sepsis have fungal, viral, or parasitic infections.
The source will not be determined in approximately 50% of patients treated for sepsis, which is termed culture-negative sepsis.17,20 Respiratory tract infections are more likely to be culture-negative, whereas urinary tract infections are likely to be culture-positive.20
Overall Approach
Despite scientific advances over the past 20 years, the management of sepsis remains largely unchanged (eFigure A). The main difference is the concept of bundles, which are multiple interventions that should be completed within a specified time frame. Use of such sepsis care protocols has been shown to decrease sepsis mortality and should be implemented in all medical facilities.21–24
eFIGURE A

Algorithm for the treatment of sepsis and septic shock.
Information from:
Levy MM, Evans LE, Rhodes A. The surviving sepsis campaign bundle: 2018 update. Intensive Care Med. 2018;44(6):925–928.
Rhodes A, Evans LE, Alhazzani W, et al. Surviving Sepsis Campaign. Crit Care Med. 2017;45(3):486–552.
Seymour CW, Rosengart MR. Septic shock. Advances in diagnosis and treatment [published correction appears in JAMA. 2015;314(13):1404]. JAMA. 2015;314(7):708–717.
Thompson K, Venkatesh B, Finfer S. Sepsis and septic shock: current approaches to management. Intern Med J. 2019;49(2):160–170.
After initial airway and respiratory stabilization, patients with sepsis should complete the sepsis bundle (fluid resuscitation, antibiotics, lactate measurement, and cultures) within three hours of presentation.24–27 Vasopressor therapy is initiated if the patient is hypotensive despite fluid resuscitation.21,28,29 Infection source control with early surgical consultation should be obtained for suspected infections requiring operative or other interventional treatment (e.g., abdominal, gallbladder or biliary, urinary, joint, skin and soft tissue infections).
Diagnosis
Sepsis has a variable presentation depending on the source of the initial infection and may not be apparent until late in the course of illness, when signs and symptoms are obvious. There are several medical conditions that mimic sepsis and should be considered in the differential diagnosis (e.g., acute pulmonary embolus, acute myocardial infarction, acute pancreatitis, acute transfusion reaction, adrenal crisis, acute alcohol withdrawal, thyrotoxicosis).30 To improve the diagnosis of sepsis, clinicians must obtain historical, clinical, laboratory, and radiographic data supportive of infection and organ dysfunction. Table 1 summarizes the clinical manifestations of sepsis and septic shock.31,32
TABLE 1. Clinical Manifestations of Sepsis and Septic Shock
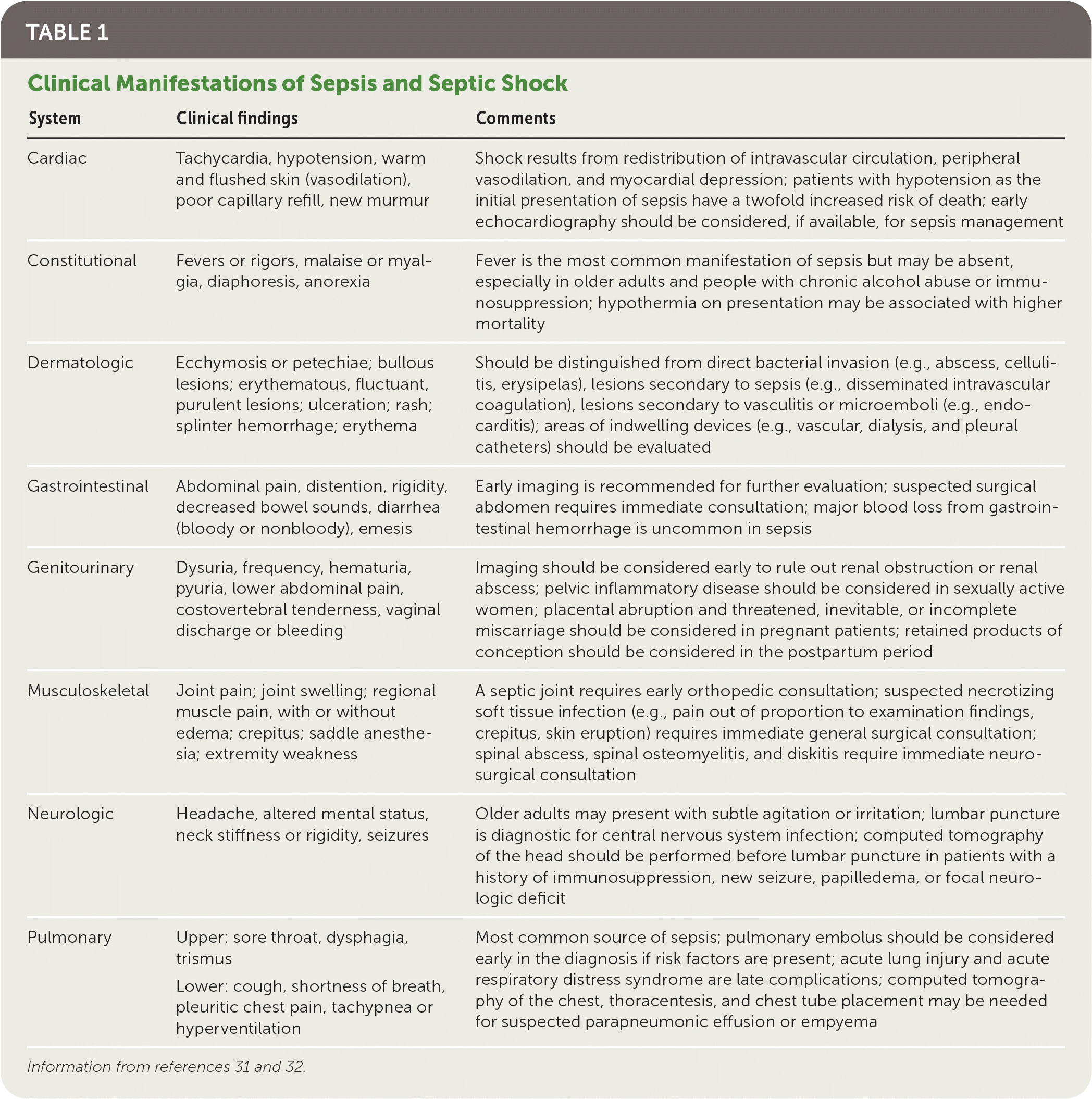
| System | Clinical findings | Comments |
|---|---|---|
| Cardiac | Tachycardia, hypotension, warm and flushed skin (vasodilation), poor capillary refill, new murmur | Shock results from redistribution of intravascular circulation, peripheral vasodilation, and myocardial depression; patients with hypotension as the initial presentation of sepsis have a twofold increased risk of death; early echocardiography should be considered, if available, for sepsis management |
| Constitutional | Fevers or rigors, malaise or myalgia, diaphoresis, anorexia | Fever is the most common manifestation of sepsis but may be absent, especially in older adults and people with chronic alcohol abuse or immunosuppression; hypothermia on presentation may be associated with higher mortality |
| Dermatologic | Ecchymosis or petechiae; bullous lesions; erythematous, fluctuant, purulent lesions; ulceration; rash; splinter hemorrhage; erythema | Should be distinguished from direct bacterial invasion (e.g., abscess, cellulitis, erysipelas), lesions secondary to sepsis (e.g., disseminated intravascular coagulation), lesions secondary to vasculitis or microemboli (e.g., endocarditis); areas of indwelling devices (e.g., vascular, dialysis, and pleural catheters) should be evaluated |
| Gastrointestinal | Abdominal pain, distention, rigidity, decreased bowel sounds, diarrhea (bloody or nonbloody), emesis | Early imaging is recommended for further evaluation; suspected surgical abdomen requires immediate consultation; major blood loss from gastrointestinal hemorrhage is uncommon in sepsis |
| Genitourinary | Dysuria, frequency, hematuria, pyuria, lower abdominal pain, costovertebral tenderness, vaginal discharge or bleeding | Imaging should be considered early to rule out renal obstruction or renal abscess; pelvic inflammatory disease should be considered in sexually active women; placental abruption and threatened, inevitable, or incomplete miscarriage should be considered in pregnant patients; retained products of conception should be considered in the postpartum period |
| Musculoskeletal | Joint pain; joint swelling; regional muscle pain, with or without edema; crepitus; saddle anesthesia; extremity weakness | A septic joint requires early orthopedic consultation; suspected necrotizing soft tissue infection (e.g., pain out of proportion to examination findings, crepitus, skin eruption) requires immediate general surgical consultation; spinal abscess, spinal osteomyelitis, and diskitis require immediate neurosurgical consultation |
| Neurologic | Headache, altered mental status, neck stiffness or rigidity, seizures | Older adults may present with subtle agitation or irritation; lumbar puncture is diagnostic for central nervous system infection; computed tomography of the head should be performed before lumbar puncture in patients with a history of immunosuppression, new seizure, papilledema, or focal neurologic deficit |
| Pulmonary | Upper: sore throat, dysphagia, trismus Lower: cough, shortness of breath, pleuritic chest pain, tachypnea or hyperventilation | Most common source of sepsis; pulmonary embolus should be considered early in the diagnosis if risk factors are present; acute lung injury and acute respiratory distress syndrome are late complications; computed tomography of the chest, thoracentesis, and chest tube placement may be needed for suspected parapneumonic effusion or empyema |
SYMPTOMS
Fever is the most common manifestation of sepsis.31 The absence of fever, however, does not exclude sepsis. Sepsis-induced hypothermia and the absence of fever are more likely in older adults and in people with chronic alcohol abuse or immunosuppression.33 Hypotension is the presenting abnormality in approximately 40% of patients with sepsis.34 In older adults, generalized weakness, agitation or irritation, or altered mental status may be the only manifestation.
LABORATORY TESTING
Laboratory testing should include a complete blood count with differential; basic metabolic panel; lactate, procalcitonin, and liver enzyme measurements; coagulation studies; and urinalysis. Arterial or venous blood sampling can determine the degree of acid-base abnormalities, which are common in sepsis and are likely secondary to tissue hypoperfusion (lactic acidosis) and renal dysfunction.35
Clinicians should obtain two sets of peripheral blood cultures (including a set from a central venous catheter, if present), as well as cultures of urine, stool (for diarrhea or recent antibiotic use), sputum (for respiratory symptoms), and skin and soft tissue (for skin abscess, ulceration, or drainage). Cerebrospinal, joint, pleural, and peritoneal fluid cultures are obtained as clinically indicated.2,36
IMAGING
Imaging studies should include chest radiography, with additional studies as indicated (e.g., echocardiography for suspected endocarditis, computed tomography of the chest for empyema or parapneumonic effusion, computed tomography of the abdomen/pelvis for renal or abdominal abscess).
SEPSIS BIOMARKERS
Procalcitonin is a marker for inflammation produced by cytokines and bacterial endotoxins and is widely used as an indicator for bacterial sepsis. Serum lactate level is also integral to the diagnosis, treatment, and prognosis of sepsis.
A procalcitonin value of less than 0.05 ng per mL is considered normal, and patients with levels less than 0.25 ng per mL have a low likelihood of bacterial sepsis.37 Procalcitonin levels rise within four hours after onset of an infection and peak at 12 to 48 hours.38 Procalcitonin levels have a statistically significant relationship with the severity of sepsis. For example, in one study, a mean procalcitonin level of 32.7 ng per mL was observed in patients with septic shock requiring vasopressors compared with a mean level of 9.6 ng per mL in patients with sepsis and no shock.39 Because of its short half-life, procalcitonin levels are also useful to monitor response to therapy and to provide guidance for antibiotic discontinuation, particularly in bacterial pneumonia. The inability to clear procalcitonin by at least 80% within 72 hours is associated with higher sepsis-related mortality in hospitalized patients.40
Increased lactate levels in patients with sepsis are the result of tissue hypoxia, aerobic glycolysis, or decreased clearance (e.g., hepatic dysfunction). A lactate level greater than 18 mg per dL (2 mmol per L) is a diagnostic criterion for septic shock in Sepsis-3. Elevated lactate levels should not be dismissed in a patient with sepsis, even with normal blood pressures. Lactate measurements should be obtained every four to six hours until levels have normalized. Lactate-guided fluid resuscitation reduces overall mortality compared with no lactate monitoring.41,42 The inability to clear lactate during sepsis management should prompt reevaluation of adequate source control.21
Treatment
FLUID RESUSCITATION
The priorities in early sepsis management are establishing vascular access and initiating fluid resuscitation. Patients with sepsis should receive an intravenous crystalloid at 30 mL per kg within the first three hours.21 Infusing an initial 1-L bolus over the first 30 minutes is an accepted approach. The remainder of fluid resuscitation should be given by repeat bolus infusions.43 Infusion of intravenous fluids in this manner enhances preload and cardiac output, thereby improving oxygen delivery. However, the hemodynamic effects of fluid boluses in sepsis last only 60 minutes.44
Several individual trials showed no difference in 28-day mortality benefit between resuscitation using a colloid (e.g., albumin) and a crystalloid (e.g., normal saline, Ringer lactate); however, a subsequent meta-analysis suggested a marginal mortality benefit with the use of albumin.26,45–47 Despite these findings, crystalloids are recommended for fluid resuscitation because of availability and lower cost.
Crystalloid formulations are unbalanced (e.g., 0.9% normal saline) or balanced (e.g., Ringer lactate, Plasma-Lyte A). Large-volume resuscitation with an unbalanced crystalloid can result in hyperchloremic acidosis, coagulopathy, and acute kidney injury. Because of these concerns, there has been an increased interest in the use of balanced crystalloids. In a recent trial comparing balanced crystalloid with normal saline in critically ill adults, the balanced crystalloid led to marginal improvement in mortality (10.3% vs. 11.1%; not statistically significant) and lower incidence of renal dysfunction (14.3% vs. 15.4%; statistically significant). This trial, however, had a heterogenous population with sepsis as the admitting diagnosis in only 15% of patients.48
Regardless of the fluid used, frequent reassessment of fluid balance beyond initial resuscitation is recommended to avoid under- or overhydration. Dynamic blood pressure response, tissue perfusion (lactate clearance), and most importantly urine output (should be 0.5 mL per kg per hour or greater) can be used to help avoid volume overload, particularly in patients with chronic renal disease, heart failure, or acute lung injury. Additional modalities can be used to determine fluid tolerance and fluid responsiveness (increase in stroke volume) for assessment of optimal fluid management. These include ultrasonography to assess inferior vena cava collapsibility or distensibility, pulse pressure variation (change in the difference between systolic and diastolic pressure), and passive leg raise test (assesses whether cardiac output and blood pressure increase when legs are raised from supine to 45 degrees).49
Fluid administration should be restricted in the latter phases of sepsis management. At 72 hours, the net fluid balance goal should be close to zero (i.e., patient ultimately voids an amount equal to the fluids given) or slightly negative (i.e., patient voids slightly more than the fluids given). Each 1-L increase in net positive balance at 72 hours is associated with increased risk of death.50–52
ANTIMICROBIAL THERAPY
Multiple studies indicate that early initiation of appropriate antibiotic therapy is associated with improved clinical outcomes.4,53–56 The precise timing is controversial. SSC guidelines recommend administration of antibiotics within the first hour.21 However, such a short time frame may be difficult in most clinical settings.21,28 More importantly, this one-hour goal has not been clearly validated by evidence-based protocols. In January 2019, the Society of Critical Care Medicine and the American College of Emergency Physicians issued a joint statement recommending against the one-hour goal.57
A meta-analysis of 11 trials including 16,178 patients assessed the timing of antibiotic administration.27 Patients were 18 years or older and presented to an emergency department with an admission diagnosis of severe sepsis or septic shock. There was no difference in mortality between patients who received antibiotics within three hours of triage in the emergency department and those who received antibiotics within the one hour after severe sepsis or septic shock was recognized. This meta-analysis does not minimize the importance of early antibiotic administration; however, it could not confirm the exact timing for maximal benefit.
A more recent retrospective study evaluated outcomes in more than 10,000 patients presenting to an emergency department with sepsis.58 One-year mortality rates were higher for patients receiving antibiotics after three hours compared with those receiving antibiotics within less than three hours. There was no statistically significant difference in mortality rates between patients receiving antibiotics within less than one hour vs. after one hour, supporting the position that the one-hour goal may be unnecessary. The study concluded that one death per 61 patients could be prevented if antibiotics are administered within 90 minutes of sepsis presentation.
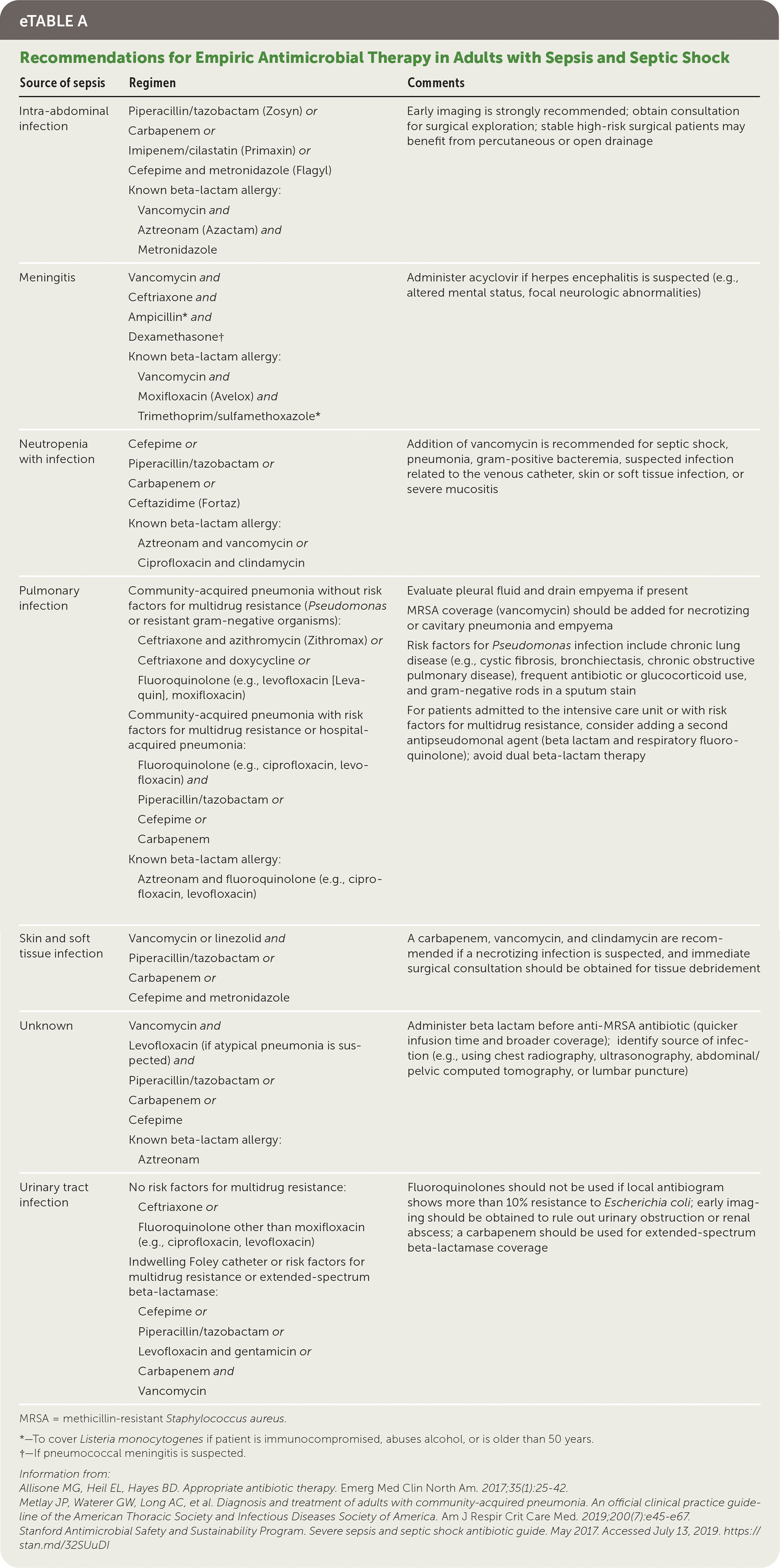
Initial antibiotic therapy should be broad and started empirically based on the suspected infection site, likely pathogen, clinical context (community vs. hospital acquired), and local resistance patterns.59–62 The use of inappropriate antibiotics is associated with up to a 34% increase in mortality.63,64 Antibiotic therapy should be narrowed or redirected once culture results are available and the causative organism has been identified. This approach reduces the risk of antimicrobial resistance, drug toxicity, and overall treatment cost. eTable A summarizes recommendations for empiric antimicrobial therapy in adults with sepsis and septic shock.
eTABLE A Recommendations for Empiric Antimicrobial Therapy in Adults with Sepsis and Septic Shock
| Source of sepsis | Regimen | Comments |
|---|---|---|
| Intra-abdominal infection | Piperacillin/tazobactam (Zosyn) or | Early imaging is strongly recommended; obtain consultation for surgical exploration; stable high-risk surgical patients may benefit from percutaneous or open drainage |
| Carbapenem or | ||
| Imipenem/cilastatin (Primaxin) or | ||
| Cefepime and metronidazole (Flagyl) | ||
| Known beta-lactam allergy: | ||
| Vancomycin and | ||
| Aztreonam (Azactam) and | ||
| Metronidazole | ||
| Meningitis | Vancomycin and | Administer acyclovir if herpes encephalitis is suspected (e.g., altered mental status, focal neurologic abnormalities) |
| Ceftriaxone and | ||
| Ampicillin* and | ||
| Dexamethasone† | ||
| Known beta-lactam allergy: | ||
| Vancomycin and | ||
| Moxifloxacin (Avelox) and | ||
| Trimethoprim/sulfamethoxazole* | ||
| Neutropenia with infection | Cefepime or | Addition of vancomycin is recommended for septic shock, pneumonia, gram-positive bacteremia, suspected infection related to the venous catheter, skin or soft tissue infection, or severe mucositis |
| Piperacillin/tazobactam or | ||
| Carbapenem or | ||
| Ceftazidime (Fortaz) | ||
| Known beta-lactam allergy: | ||
| Aztreonam and vancomycin or | ||
| Ciprofloxacin and clindamycin | ||
| Pulmonary infection | Community-acquired pneumonia without risk factors for multidrug resistance (Pseudomonas or resistant gram-negative organisms): | Evaluate pleural fluid and drain empyema if present MRSA coverage (vancomycin) should be added for necrotizing or cavitary pneumonia and empyema |
| Ceftriaxone and azithromycin (Zithromax) or | Risk factors for Pseudomonas infection include chronic lung disease (e.g., cystic fibrosis, bronchiectasis, chronic obstructive pulmonary disease), frequent antibiotic or glucocorticoid use, and gram-negative rods in a sputum stain | |
| Ceftriaxone and doxycycline or | ||
| Fluoroquinolone (e.g., levofloxacin [Levaquin], moxifloxacin) | ||
| Community-acquired pneumonia with risk factors for multidrug resistance or hospital-acquired pneumonia: | For patients admitted to the intensive care unit or with risk factors for multidrug resistance, consider adding a second antipseudomonal agent (beta lactam and respiratory fluoroquinolone); avoid dual beta-lactam therapy | |
| Fluoroquinolone (e.g., ciprofloxacin, levofloxacin) and | ||
| Piperacillin/tazobactam or | ||
| Cefepime or | ||
| Carbapenem | ||
| Known beta-lactam allergy: | ||
| Aztreonam and fluoroquinolone (e.g., ciprofloxacin, levofloxacin) | ||
| Skin and soft tissue infection | Vancomycin or linezolid and | A carbapenem, vancomycin, and clindamycin are recommended if a necrotizing infection is suspected, and immediate surgical consultation should be obtained for tissue debridement |
| Piperacillin/tazobactam or | ||
| Carbapenem or | ||
| Cefepime and metronidazole | ||
| Unknown | Vancomycin and | Administer beta lactam before anti-MRSA antibiotic (quicker infusion time and broader coverage); identify source of infection (e.g., using chest radiography, ultrasonography, abdominal/pelvic computed tomography, or lumbar puncture) |
| Levofloxacin (if atypical pneumonia is suspected) and | ||
| Piperacillin/tazobactam or | ||
| Carbapenem or | ||
| Cefepime | ||
| Known beta-lactam allergy: | ||
| Aztreonam | ||
| Urinary tract infection | No risk factors for multidrug resistance: | Fluoroquinolones should not be used if local antibiogram shows more than 10% resistance to Escherichia coli; early imaging should be obtained to rule out urinary obstruction or renal abscess; a carbapenem should be used for extended-spectrum beta-lactamase coverage |
| Ceftriaxone or | ||
| Fluoroquinolone other than moxifloxacin (e.g., ciprofloxacin, levofloxacin) | ||
| Indwelling Foley catheter or risk factors for multidrug resistance or extended-spectrum beta-lactamase: | ||
| Cefepime or | ||
| Piperacillin/tazobactam or | ||
| Levofloxacin and gentamicin or | ||
| Carbapenem and | ||
| Vancomycin |
MRSA = methicillin-resistant Staphylococcus aureus.
*—To cover Listeria monocytogenes if patient is immunocompromised, abuses alcohol, or is older than 50 years.
†—If pneumococcal meningitis is suspected.
Information from:
Allisone MG, Heil EL, Hayes BD. Appropriate antibiotic therapy. Emerg Med Clin North Am. 2017;35(1):25–42.
Metlay JP, Waterer GW, Long AC, et al. Diagnosis and treatment of adults with community-acquired pneumonia. An official clinical practice guideline of the American Thoracic Society and Infectious Diseases Society of America. Am J Respir Crit Care Med. 2019;200(7):e45–e67.
Stanford Antimicrobial Safety and Sustainability Program. Severe sepsis and septic shock antibiotic guide. May 2017. Accessed July 13, 2019. http://med.stanford.edu/bugsanddrugs/guidebook/_jcr_content/main/panel_builder_1454513702/panel_0/download_695325257/file.res/Sepsis%20ABX%202017-05-25.pdf
Currently, there is no consensus on de-escalation of combination antibiotic therapy, particularly in culture-negative sepsis. Factors to consider include clinical progress during treatment, use of biomarkers (e.g., decreasing procalcitonin levels) to monitor antibiotic response, and fixed duration of combination therapy.
Antibiotic therapy for seven to 10 days is sufficient for most infections associated with sepsis, including culture-negative sepsis.21 Specific infections, such as endocarditis, osteomyelitis, or colonized endovascular devices or orthopedic hardware that cannot be removed, require a longer duration of antibiotic therapy.
Vasopressor Therapy
Norepinephrine is the first-line vasopressor agent for patients with septic shock if initial fluid resuscitation fails to restore mean arterial pressure to 65 mm Hg or greater (https://www.mdcalc.com/mean-arterial-pressure-map).21,28,29 Vasopressor therapy clearly improves survival in these patients and should be started within the first hour following initial fluid resuscitation.25,29 Failure to initiate early vasopressor therapy in patients with septic shock increases mortality rates by 5% per hour of delay.65
Norepinephrine should be initiated at 2 to 5 mcg per minute and titrated up to 35 to 90 mcg per minute to achieve a mean arterial pressure of 65 mm Hg or greater.28 If norepinephrine fails to restore the mean arterial pressure to this level, vasopressin (up to 0.03 units per minute) can be added as a second-line agent, followed by the addition of epinephrine (20 to 50 mcg per minute) if needed.66
Vasopressor therapy is infused through a central venous catheter with dynamic blood pressure monitoring through an arterial line. Peripheral administration of norepinephrine can be initiated if placement of a central venous catheter is delayed (e.g., pending a consultation, failed attempt at inserting a central venous catheter). This is favored over continued fluid administration if septic shock is unresponsive to fluid resuscitation.67 The risk of tissue necrosis from short-term use of vasopressors through a peripheral venous catheter is low.68 Vasopressor therapy should be titrated to maintain adequate hemodynamic status and should be used for the shortest duration possible.
Other Interventions
STANDARD THERAPIES
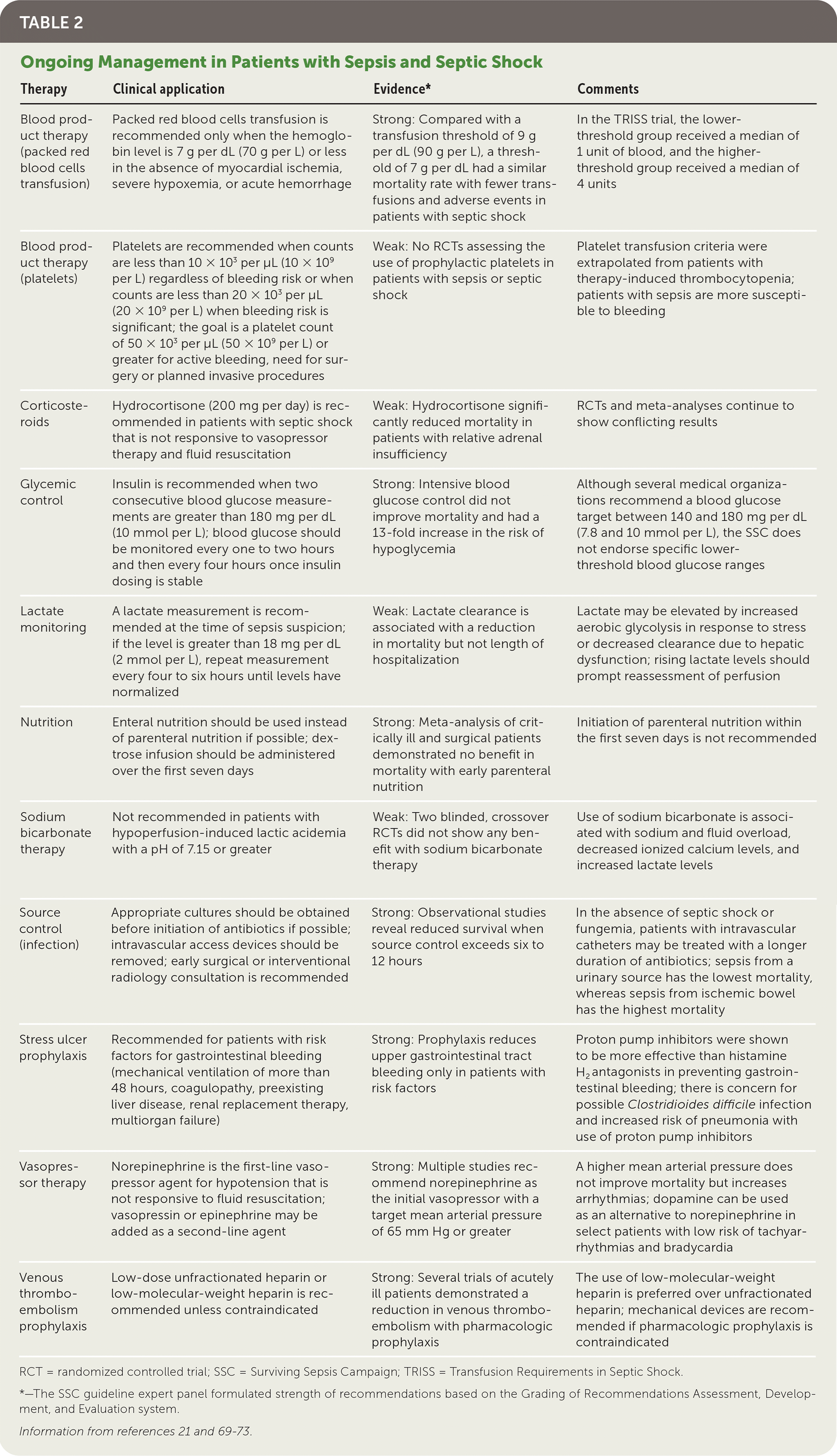
Additional therapeutic interventions designed to improve survival include corticosteroids (although evidence is mixed), blood product therapy, glycemic control, nutrition, and source control (Table 2).21,69–73
TABLE 2 Ongoing Management in Patients with Sepsis and Septic Shock
| Therapy | Clinical application | Evidence* | Comments |
|---|---|---|---|
| Blood product therapy (packed red blood cells transfusion) | Packed red blood cells transfusion is recommended only when the hemoglobin level is 7 g per dL (70 g per L) or less in the absence of myocardial ischemia, severe hypoxemia, or acute hemorrhage | Strong: Compared with a transfusion threshold of 9 g per dL (90 g per L), a threshold of 7 g per dL had a similar mortality rate with fewer transfusions and adverse events in patients with septic shock | In the TRISS trial, the lower-threshold group received a median of 1 unit of blood, and the higher-threshold group received a median of 4 units |
| Blood product therapy (platelets) | Platelets are recommended when counts are less than 10 × 103 per μL (10 × 109 per L) regardless of bleeding risk or when counts are less than 20 × 103 per μL (20 × 109 per L) when bleeding risk is significant; the goal is a platelet count of 50 × 103 per μL (50 × 109 per L) or greater for active bleeding, need for surgery or planned invasive procedures | Weak: No RCTs assessing the use of prophylactic platelets in patients with sepsis or septic shock | Platelet transfusion criteria were extrapolated from patients with therapy-induced thrombocytopenia; patients with sepsis are more susceptible to bleeding |
| Corticosteroids | Hydrocortisone (200 mg per day) is recommended in patients with septic shock that is not responsive to vasopressor therapy and fluid resuscitation | Weak: Hydrocortisone significantly reduced mortality in patients with relative adrenal insufficiency | RCTs and meta-analyses continue to show conflicting results |
| Glycemic control | Insulin is recommended when two consecutive blood glucose measurements are greater than 180 mg per dL (10 mmol per L); blood glucose should be monitored every one to two hours and then every four hours once insulin dosing is stable | Strong: Intensive blood glucose control did not improve mortality and had a 13-fold increase in the risk of hypoglycemia | Although several medical organizations recommend a blood glucose target between 140 and 180 mg per dL (7.8 and 10 mmol per L), the SSC does not endorse specific lower-threshold blood glucose ranges |
| Lactate monitoring | A lactate measurement is recommended at the time of sepsis suspicion; if the level is greater than 18 mg per dL (2 mmol per L), repeat measurement every four to six hours until levels have normalized | Weak: Lactate clearance is associated with a reduction in mortality but not length of hospitalization | Lactate may be elevated by increased aerobic glycolysis in response to stress or decreased clearance due to hepatic dysfunction; rising lactate levels should prompt reassessment of perfusion |
| Nutrition | Enteral nutrition should be used instead of parenteral nutrition if possible; dextrose infusion should be administered over the first seven days | Strong: Meta-analysis of critically ill and surgical patients demonstrated no benefit in mortality with early parenteral nutrition | Initiation of parenteral nutrition within the first seven days is not recommended |
| Sodium bicarbonate therapy | Not recommended in patients with hypoperfusion-induced lactic acidemia with a pH of 7.15 or greater | Weak: Two blinded, crossover RCTs did not show any benefit with sodium bicarbonate therapy | Use of sodium bicarbonate is associated with sodium and fluid overload, decreased ionized calcium levels, and increased lactate levels |
| Source control (infection) | Appropriate cultures should be obtained before initiation of antibiotics if possible; intravascular access devices should be removed; early surgical or interventional radiology consultation is recommended | Strong: Observational studies reveal reduced survival when source control exceeds six to 12 hours | In the absence of septic shock or fungemia, patients with intravascular catheters may be treated with a longer duration of antibiotics; sepsis from a urinary source has the lowest mortality, whereas sepsis from ischemic bowel has the highest mortality |
| Stress ulcer prophylaxis | Recommended for patients with risk factors for gastrointestinal bleeding (mechanical ventilation of more than 48 hours, coagulopathy, preexisting liver disease, renal replacement therapy, multiorgan failure) | Strong: Prophylaxis reduces upper gastrointestinal tract bleeding only in patients with risk factors | Proton pump inhibitors were shown to be more effective than histamine H2 antagonists in preventing gastrointestinal bleeding; there is concern for possible Clostridioides difficile infection and increased risk of pneumonia with use of proton pump inhibitors |
| Vasopressor therapy | Norepinephrine is the first-line vasopressor agent for hypotension that is not responsive to fluid resuscitation; vasopressin or epinephrine may be added as a second-line agent | Strong: Multiple studies recommend norepinephrine as the initial vasopressor with a target mean arterial pressure of 65 mm Hg or greater | A higher mean arterial pressure does not improve mortality but increases arrhythmias; dopamine can be used as an alternative to norepinephrine in select patients with low risk of tachyarrhythmias and bradycardia |
| Venous thromboembolism prophylaxis | Low-dose unfractionated heparin or low-molecular-weight heparin is recommended unless contraindicated | Strong: Several trials of acutely ill patients demonstrated a reduction in venous thromboembolism with pharmacologic prophylaxis | The use of low-molecular-weight heparin is preferred over unfractionated heparin; mechanical devices are recommended if pharmacologic prophylaxis is contraindicated |
RCT = randomized controlled trial; SSC = Surviving Sepsis Campaign; TRISS = Transfusion Requirements in Septic Shock.
*—The SSC guideline expert panel formulated strength of recommendations based on the Grading of Recommendations Assessment, Development, and Evaluation system.
ADJUNCTIVE THERAPIES
A recent small retrospective observational study evaluated the effect of usual care vs. the addition of intravenous ascorbic acid (1,500 mg every six hours), hydrocortisone (50 mg every six hours), and thiamine (200 mg every 12 hours) in patients with sepsis.74,75 Patients in the treatment group had decreased mortality (8.5% vs. 40%; P < .001) and decreased time on vasopressor therapy compared with the control group. The major limitation of this trial was sample size (94 patients), but it has generated considerable interest.76 A larger clinical trial is ongoing to better assess the effectiveness of this therapy.77
What Is New
A recent article analyzed a dataset of more than 20,000 patients to identify those at greatest risk of dying from sepsis using clinical phenotypes. Four phenotypes were derived based on 27 biomarkers.78 Patients with the lowest mortality (5%) were taking the lowest doses of vasopressors and patients with the highest mortality (40%) had liver dysfunction and septic shock. This study highlights the clinical heterogeneity of sepsis; however, further research is needed before these clinical phenotypes can be used in clinical practice.
Data Sources: We searched OvidSP, PubMed, UptoDate, Google Scholar, and http://www.survivingsepsis.org. Key words were sepsis, septic shock, Surviving Sepsis Campaign, pathophysiology, antimicrobial therapy, fluid resuscitation, mortality, epidemiology, hospitalization, long-term follow-up, intensive glucose control, corticosteroids, vasopressor therapy, fluid resuscitation, fluid balance, norepinephrine, procalcitonin, lactate, early goal-directed therapy, systemic inflammatory response syndrome, sequential organ failure assessment, venous thromboembolism prophylaxis, and source control. Search dates: December 7, 2018; January to June 30, 2019; and October 12, 2019.
The views expressed herein are those of the authors and do not reflect the official policy of the Department of the Army, Department of Defense, or the U.S. government.

