Key Points for Practice
• The BMJ/MAGIC Group recommends no screening for individuals with an estimated 15-year risk of CRC less than 3%.
• Colonoscopy, flexible sigmoidoscopy, annual FIT, and biennial FIT similarly reduce CRC mortality.
• A key difference between recommended CRC screening methods is the probability of colonoscopy, which introduces risks of cardiovascular events, major bleeding, and perforation.
From the AFP Editors
Colorectal cancer (CRC) is a common cancer with 65% overall five-year survival. CRC incidence increases by 0.5% to 1.5% every five years after 50 years of age. Because removal of adenomas can prevent cancer and earlier stage survival is higher, screening can reduce cancer incidence and mortality. CRC screening recommendations vary because the underlying evidence is low quality, with few screening methods evaluated by randomized trials. The BMJ/MAGIC Group performed a systematic review based on 15-year CRC risk models to compare screening options.
Recommendations apply to average-risk asymptomatic adults 50 to 79 years of age with at least 15 years of life expectancy. Recommendations do not apply to people with a history of polyps, CRC, inflammatory bowel disease, or hereditary syndromes that increase CRC risk.
Estimating Colorectal Cancer Risk
The BMJ/MAGIC Group recommends calculating estimated CRC risk and avoiding screening for low-risk patients. Several risk estimate calculators have been developed, but the QCancer calculator (https://qcancer.org/15yr/colorectal/) is recommended based on proven accuracy, use of routinely available information, and 15-year risk estimation. QCancer has not been validated in U.S. patients. If estimated 15-year risk is less than 3%, no screening is recommended because CRC mortality approximates risks of cardiovascular events, serious bleeding, and colon perforation from screening.
Comparing Screening Methods
MORTALITY
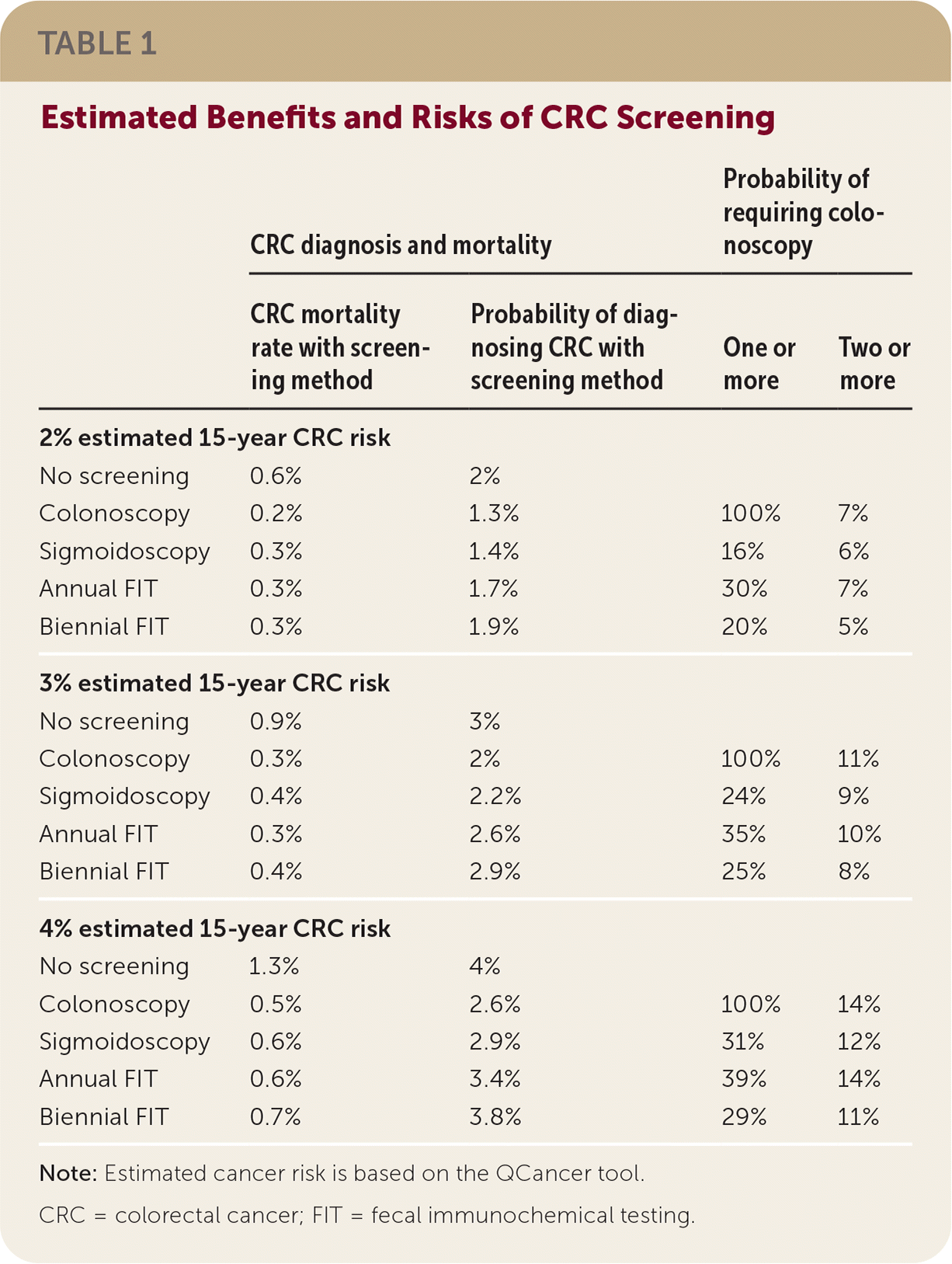
Systematic review demonstrates similar reductions in 15-year CRC mortality for four screening options, although evidence for all-cause mortality is lacking. Reductions in 15-year CRC mortality compared with no screening are 63% with colonoscopy, 59% with annual fecal immunochemical testing (FIT), 52% for flexible sigmoidoscopy, and 50% with biennial FIT. CRC mortality risks by QCancer risk and screening method are shown in Table 1.
TABLE 1. Estimated Benefits and Risks of CRC Screening
| CRC diagnosis and mortality | Probability of requiring colonoscopy | |||
|---|---|---|---|---|
| CRC mortality rate with screening method | Probability of diagnosing CRC with screening method | One or more | Two or more | |
| 2% estimated 15-year CRC risk | ||||
| No screening | 0.6% | 2% | ||
| Colonoscopy | 0.2% | 1.3% | 100% | 7% |
| Sigmoidoscopy | 0.3% | 1.4% | 16% | 6% |
| Annual FIT | 0.3% | 1.7% | 30% | 7% |
| Biennial FIT | 0.3% | 1.9% | 20% | 5% |
| 3% estimated 15-year CRC risk | ||||
| No screening | 0.9% | 3% | ||
| Colonoscopy | 0.3% | 2% | 100% | 11% |
| Sigmoidoscopy | 0.4% | 2.2% | 24% | 9% |
| Annual FIT | 0.3% | 2.6% | 35% | 10% |
| Biennial FIT | 0.4% | 2.9% | 25% | 8% |
| 4% estimated 15-year CRC risk | ||||
| No screening | 1.3% | 4% | ||
| Colonoscopy | 0.5% | 2.6% | 100% | 14% |
| Sigmoidoscopy | 0.6% | 2.9% | 31% | 12% |
| Annual FIT | 0.6% | 3.4% | 39% | 14% |
| Biennial FIT | 0.7% | 3.8% | 29% | 11% |
Note: Estimated cancer risk is based on the QCancer tool.
CRC = colorectal cancer; FIT = fecal immunochemical testing.
INCIDENCE
CRC screening also reduces the incidence of CRC through removal of premalignant adenomas, a benefit that varies between screening methods. Direct visualization reduces CRC incidence the most, with a 34% reduction from colonoscopy and 27% from sigmoidoscopy. With FIT, the reductions are much less: only 15% with annual FIT and 5% with biennial FIT. Although two more recent flexible sigmoidoscopy studies showed greater CRC reduction in men, absolute differences are too small to change recommendations. Risks of CRC incidence by QCancer risk and screening method are shown in Table 1.
HARM HIGHER WITH COLONOSCOPY
Harms of CRC screening are primarily from colonoscopy. Risks include colon perforation, bleeding, other gastrointestinal issues, and cardiovascular events from sedation. The BMJ/MAGIC Group estimated the probability of requiring colonoscopy with each screening method, as shown in Table 1.
Patient Counseling
Because of similar effects on CRC mortality, patient values are important when choosing a screening method. Conversations about patient preferences and values can be facilitated through the MAGICapp decision tool, available at http://magicproject.org/190220dist.
Editor's Note: Limited evidence in CRC screening has led to a variety of recommendations. These guidelines, produced by the BMJ/MAGIC Group, add two interesting proposals. First, use a risk calculator to eliminate screening in patients least likely to benefit. The QCancer calculator was designed for and verified in British patients and has not been studied in the United States. Second, consider the use of colonoscopy as a surrogate for screening harms because nearly all harms of screening are caused by the procedure. The AAFP continues to endorse the 2016 U.S. Preventive Services Task Force recommendations to screen patients between 50 and 75 years of age with FIT, flexible sigmoidoscopy, or colonoscopy and weigh screening risks and benefits for patients 76 to 85 years of age.—Michael J. Arnold, MD, contributing editor
Guideline source: BMJ/MAGIC Group
Evidence rating system used? Yes
Systematic literature search described? Yes
Guideline developed by participants without relevant financial ties to industry? Yes
Recommendations based on patient-oriented outcomes? Yes
Published source: BMJ. October 2, 2019;367:l5515
Available at: https://www.bmj.com/content/367/bmj.l5515

