Lemborexant (Dayvigo) is labeled for the treatment of insomnia to improve sleep onset and maintenance in adults. It is the second dual orexin receptor antagonist and blocks wake-promotion by blocking the binding of neuropeptides, orexin A, and orexin B. Lemborexant is a Schedule IV controlled substance.1
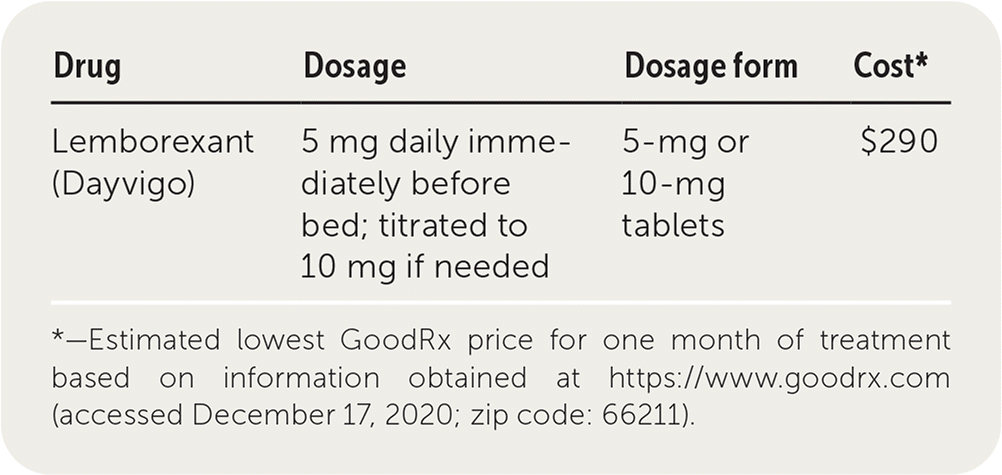
| Drug | Dosage | Dosage form | Cost* |
|---|---|---|---|
| Lemborexant (Dayvigo) | 5 mg daily immediately before bed; titrated to 10 mg if needed | 5-mg or 10-mg tablets | $290 |
*—Estimated lowest GoodRx price for one month of treatment based on information obtained at https://www.goodrx.com (accessed December 17, 2020; zip code: 66211).
Safety
Lemborexant has the potential for abuse, although the risk is similar to that of other insomnia medications such as zolpidem (Ambien) and suvorexant (Belsomra). It is a central nervous system depressant, and next-day impairment is possible at therapeutic doses. Rare but serious adverse effects include sleep paralysis, hallucinations, cataplexy-like symptoms, and complex sleep behaviors (e.g., walking, driving, or cooking while not fully awake). Lemborexant should be avoided in patients with sleep apnea, chronic obstructive pulmonary disease, or depression. It should not be used in patients taking other central nervous system depressants, cytochrome P450 3A4 (CYP3A4) enzyme inducers (e.g., antiepileptics, St. John's wort, barbiturates), or CYP3A4 enzyme inhibitors (e.g., antiarrhythmics, azole antifungals, macrolides, protease inhibitors). Lemborexant is contraindicated in patients with narcolepsy. It has not been studied in pregnant or breastfeeding patients, although no adverse effects were seen in animal studies.1
Tolerability
Lemborexant is generally well-tolerated, with discontinuation rates of less than 1% with short-term use (two weeks) and less than 10% with long-term use (12 months).2,3 The most common adverse effects include day time somnolence (10%), headache (6%), and abnormal dreams (2%). In clinical trials of patients without anxiety, depression, or a history of substance overuse, lemborexant did not affect next-day functioning or memory, except when taken with alcohol. Driving ability may be impaired in patients taking the 10-mg dose.1 Lemborexant and zolpidem have a similar safety profile in older adults.4 Lemborexant does not need to be tapered and can be discontinued abruptly because rebound insomnia and withdrawal symptoms do not occur.1
Effectiveness
Lemborexant has been compared with placebo and zolpidem for sleep onset and maintenance in two randomized, double-blind, placebo-controlled studies that included approximately 800 adults.2,3 It has been shown to improve objective and subjective sleep measures.2–4 Effects are seen within one week and are maintained for at least 12 months; they include faster sleep onset and decreased nighttime awakenings. After 12 months of nightly treatment, patients fell asleep an average of 26 minutes faster with either dose of lemborexant from a baseline delay of about one hour. Lemborexant delays the time to first awakening by about 43 minutes.2 In adults older than 55 years, lemborexant improves time to sleep onset by about 20 minutes vs. 7.5 minutes with zolpidem extended-release (P < .001). Lemborexant shows a modest improvement over zolpidem in waking after sleep onset, providing an additional seven to nine minutes of sleep before first awakening.4 Lemborexant has not been compared with suvorexant or with other hypnotics and has not been studied in shift workers or patients with jet lag.
Price
Lemborexant costs about $290 for a 30-day supply of 5-mg or 10-mg tablets. In comparison, suvorexant costs about $380 for a one-month supply. These medications are substantially more expensive than zolpidem, which costs about $20 for a one-month supply.
Simplicity
The recommended starting dosage of lemborexant is 5 mg taken once daily immediately before bed. The dosage may be increased to 10 mg if needed; however, patients older than 65 years should be limited to 5 mg once daily. Lemborexant should be taken only if the patient has at least seven hours to devote to sleep at night. The effectiveness of lemborexant is decreased if taken with food, particularly for sleep onset. Lemborexant has been shown to be safe and effective when taken nightly for up to one year. 1
Bottom Line
Lemborexant improves sleep onset and sleep maintenance for patients with insomnia. Because of its high cost, lemborexant should be reserved for patients in whom other treatments have failed or cannot be tolerated. Although daily hypnotic use should be minimized, lemborexant has been shown to be safe for long-term use. Compared with other insomnia medications, lemborexant has shown a small decrease in the risk of next-day impairment and a reduced risk of physical dependence. Sleep hygiene and cognitive behavior therapy should still be used as first-line therapy.

