Semaglutide (Wegovy) is a glucagon-like peptide-1 (GLP-1) receptor agonist. It is labeled for use as an adjunct to diet and exercise for chronic weight management in adults classified as obese (body mass index [BMI] of 30 kg per m2 or greater) or overweight (BMI of 27 kg per m2 or greater) in the presence of one weight-related comorbidity. Semaglutide is also available in a lower-dose injectable formulation (Ozempic) and an oral formulation (Rybelsus) for the treatment of type 2 diabetes mellitus.1
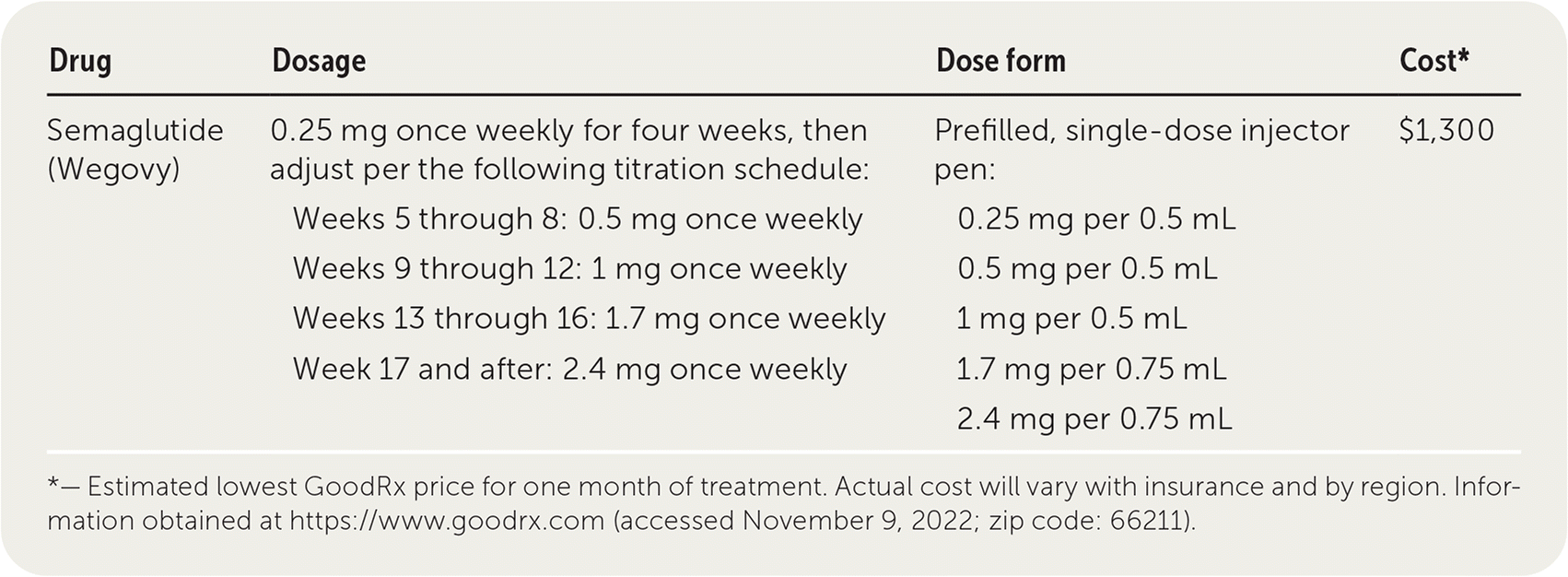
| Drug | Dosage | Dose form | Cost* |
|---|---|---|---|
| Semaglutide (Wegovy) | 0.25 mg once weekly for four weeks, then adjust per the following titration schedule: Weeks 5 through 8: 0.5 mg once weekly Weeks 9 through 12: 1 mg once weekly Weeks 13 through 16: 1.7 mg once weekly Week 17 and after: 2.4 mg once weekly | Prefilled, single-dose injector pen: 0.25 mg per 0.5 mL 0.5 mg per 0.5 mL 1 mg per 0.5 mL 1.7 mg per 0.75 mL 2.4 mg per 0.75 mL | $1,300 |
*— Estimated lowest GoodRx price for one month of treatment. Actual cost will vary with insurance and by region. Information obtained at https://www.goodrx.com (accessed November 9, 2022; zip code: 66211).
Safety
The most common adverse effect of semaglutide is an increase in heart rate, with an increase of 10 to 19 beats per minute in 41% of patients (34% with placebo) and 20 beats per minute in 26% of patients (vs. 16% with placebo). Serious adverse effects are rare but occur at a higher rate than with placebo; these include acute pancreatitis, acute gallbladder disease, retinopathy, acute kidney injury, and hypersensitivity reactions. Acute pancreatitis has been observed in patients taking GLP-1 receptor agonists. Semaglutide should not be used in patients with a history of pancreatitis because it has not been studied in this population.1 Although significant weight loss can increase the risk of cholelithiasis, the incidence in clinical trials was higher in patients taking semaglutide than in those taking placebo (1.6% vs. 0.7%; number needed to harm = 111).1 Clinically significant hypoglycemia has not been observed in patients without a history of diabetes. Concomitant use of semaglutide with insulin or insulin secretagogues may result in hypoglycemia, and a dose reduction in the concomitant medication may be necessary.
Because semaglutide-induced thyroid C-cell tumors have been reported in rodent studies, semaglutide should not be used in patients with a personal or family history of medullary thyroid cancer or multiple endocrine neoplasia syndrome type 2.1
Semaglutide may cause fetal harm and should be discontinued at least two months before a planned pregnancy. Semaglutide is not approved for use in patients younger than 18 years. The effect of semaglutide on breastfed infants is unknown.1
Tolerability
Gastrointestinal adverse effects, including nausea (44%), diarrhea (30%), vomiting (24%), and constipation (24%), are common at the start of therapy and when the dose is increased.1 About 6.8% of patients discontinue treatment due to adverse effects.1
Effectiveness
The effectiveness of semaglutide to induce weight loss has been evaluated in three studies that included 3,079 adults with obesity and 300 who were overweight. Participants had at least one weight-related comorbid condition such as dyslipidemia or hypertension, and this included 807 patients who had type 2 diabetes.2–4 In these studies, patients taking a dosage of 2.4 mg weekly experienced an average body weight reduction of −13.5%, compared with a −3.8% average reduction in the placebo groups. Most patients (78.6% vs. 36.4% with placebo) lost at least 5% of their body weight; 61.2% (vs. 16.4% with placebo) lost at least 10%; and 42.1% (vs. 7.4% with placebo) lost at least 15%. Sustained weight loss has been demonstrated with up to 68 weeks of treatment with semaglutide. Based on a single study of treatment discontinuation after short-term (five months) therapy, patients are likely to regain some weight.5 Semaglutide has been compared with the weight-loss medication liraglutide (Saxenda) and was found to be superior, resulting in significantly more weight loss after 68 weeks (mean weight reduction = −15.8% vs. −6.4%).6
Compared with placebo, semaglutide has been shown to result in greater improvements in cardiometabolic risk factors such as waist circumference, blood pressure, lipid levels, and A1C levels. However, the differences are minimal and the clinical relevance is unknown. No current research has evaluated its effect on mortality or cardiovascular, diabetes-related, or other outcomes.
Price
A one-month supply of semaglutide at the weekly target dosage of 2.4 mg costs approximately $1,300. This cost is similar to liraglutide, 3 mg daily, which costs about $1,050 per month. This is significantly more expensive than some other medications used for weight loss, including bupropion/naltrexone ($525 per month), orlistat ($300 per month), phentermine/topiramate (Qsymia; $200 per month), and phentermine ($15 per month). Many third-party plans do not cover weight-loss medications in the absence of another indicated diagnosis, making affordability a significant barrier to patients.
Simplicity
Semaglutide is supplied as a prefilled, single-dose injector pen. It is initiated at 0.25 mg weekly via subcutaneous injection and increased every four weeks up to a dosage of 2.4 mg weekly as tolerated. If a dose is not tolerated during the escalation period, patients should consider delaying the next dose increase by four weeks. The manufacturer recommends discontinuing treatment if a patient is unable to tolerate the 2.4-mg dose. Semaglutide pens should be stored in the refrigerator, but they may be kept at room temperature for up to 28 days.1
Bottom Line
Semaglutide may result in more weight loss than other currently available treatments, but many patients will experience gastrointestinal effects and may regain a portion of the weight lost if the medication is discontinued. Some patients may not want to use an injectable medication. Semaglutide is significantly more expensive than other available weight-loss medications and may not be covered by health insurance.

