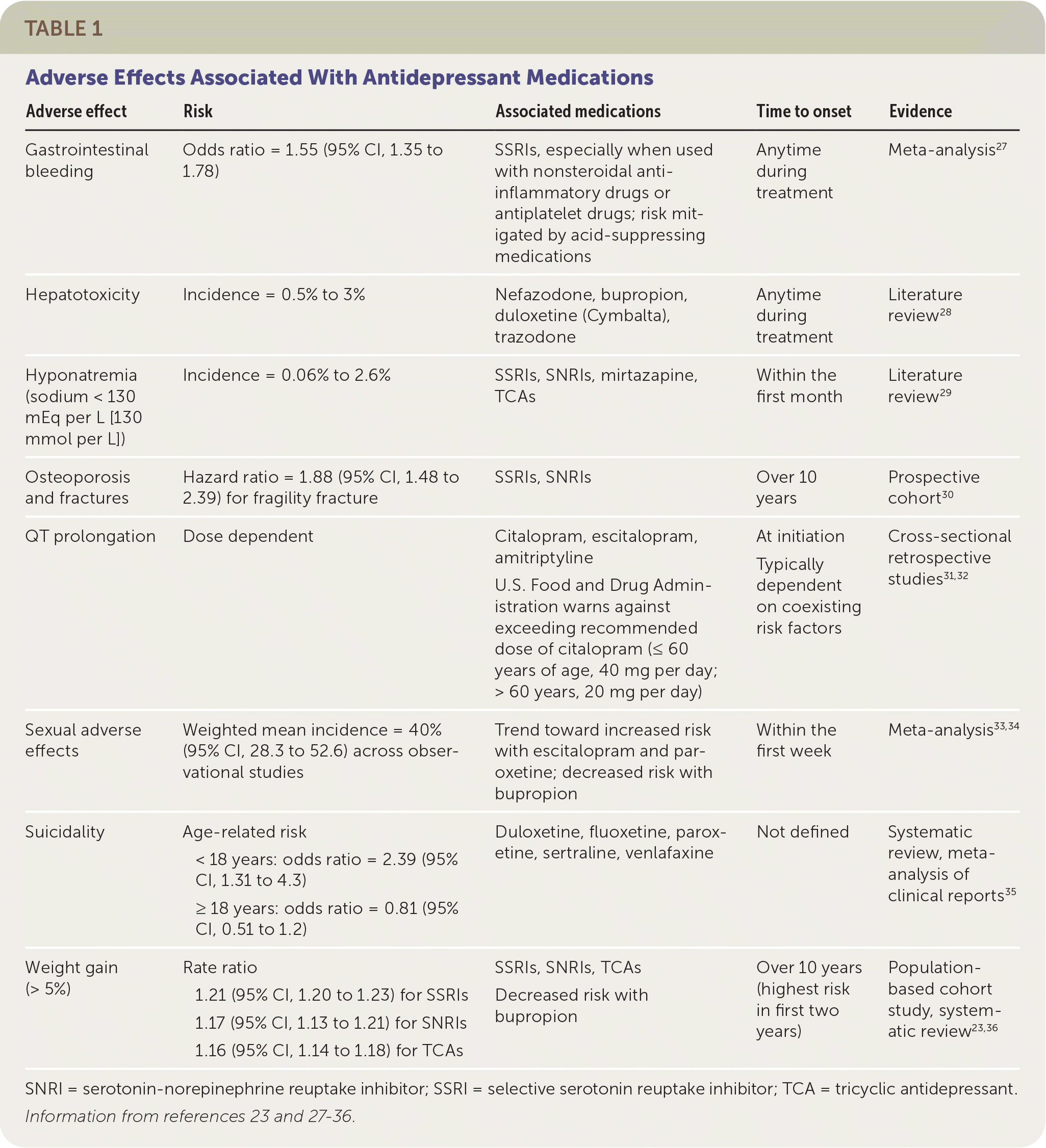
| Adverse effect | Risk | Associated medications | Time to onset | Evidence |
|---|---|---|---|---|
| Gastrointestinal bleeding | Odds ratio = 1.55 (95% CI, 1.35 to 1.78) | SSRIs, especially when used with nonsteroidal anti-inflammatory drugs or antiplatelet drugs; risk mitigated by acid-suppressing medications | Anytime during treatment | Meta-analysis27 |
| Hepatotoxicity | Incidence = 0.5% to 3% | Nefazodone, bupropion, duloxetine (Cymbalta), trazodone | Anytime during treatment | Literature review 28 |
| Hyponatremia (sodium < 130 mEq per L [130 mmol per L]) | Incidence = 0.06% to 2.6% | SSRIs, SNRIs, mirtazapine, TCAs | Within the first month | Literature review 29 |
| Osteoporosis and fractures | Hazard ratio = 1.88 (95% CI, 1.48 to 2.39) for fragility fracture | SSRIs, SNRIs | Over 10 years | Prospective cohort30 |
| QT prolongation | Dose dependent | Citalopram, escitalopram, amitriptyline U.S. Food and Drug Administration warns against exceeding recommended dose of citalopram (≤ 60 years of age, 40 mg per day; > 60 years, 20 mg per day) | At initiation Typically dependent on coexisting risk factors | Cross-sectional retrospective studies31,32 |
| Sexual adverse effects | Weighted mean incidence = 40% (95% CI, 28.3 to 52.6) across observational studies | Trend toward increased risk with escitalopram and paroxetine; decreased risk with bupropion | Within the first week | Meta-analysis33,34 |
| Suicidality | Age-related risk < 18 years: odds ratio = 2.39 (95% CI, 1.31 to 4.3) ≥ 18 years: odds ratio = 0.81 (95% CI, 0.51 to 1.2) | Duloxetine, fluoxetine, paroxetine, sertraline, venlafaxine | Not defined | Systematic review, meta-analysis of clinical reports35 |
| Weight gain (> 5%) | Rate ratio 1.21 (95% CI, 1.20 to 1.23) for SSRIs 1.17 (95% CI, 1.13 to 1.21) for SNRIs 1.16 (95% CI, 1.14 to 1.18) for TCAs | SSRIs, SNRIs, TCAs Decreased risk with bupropion | Over 10 years (highest risk in first two years) | Population-based cohort study, systematic review23,36 |