When monitoring growth and development in the premature infant, physicians should make adjustments for the estimated due date. With minor exceptions, administration of immunizations is based on the chronologic age. Administration of hepatitis B vaccine should be delayed until the infant weighs 2,000 g (4 lb, 5 oz). Administration of influenza vaccine should be considered in infants with chronic medical problems, and the pneumococcal vaccine may be beneficial at age two in children with chronic problems, especially pulmonary disease. Premature infants should also be monitored to assure appropriate nutrition. Breast-fed infants should probably receive vitamin supplements during the first year. Supplemental iron should be initiated at two weeks to two months after birth and continued for 12 to 15 months. Office care includes screening for problems that occur more frequently in premature infants, especially vision and hearing problems. Because many of these infants require care from multiple medical disciplines, coordination of care is another important role for the family physician. The goals of this care are to promote normal growth and development and minimize morbidity and mortality.
Infants who are born before 37 weeks of gestational age are defined as premature (Table 1). These infants usually weigh less than 2,500 g (5 lb, 8 oz) and constitute about 10 percent of all births.1 Infant mortality rises from five times normal at 37 weeks of gestational age to 45 times normal at 32 weeks of gestational age.2 Most of the problems associated with prematurity occur in infants with birth weights of 1,500 g (3 lb, 5 oz) or less, usually in those born at less than 32 weeks of gestational age.
TABLE 1 Terms Related to Prematurity
| Premature infant | |
| An infant born before 37 weeks of estimated gestational age | |
| Low birth weight | |
| Birth weight <2,500 g (5 lb, 8 oz) | |
| Very low birth weight | |
| Birth weight <1,500 g (3 lb, 5 oz) | |
| Extremely low birth weight | |
| Birth weight <1,000 g (2 lb, 3 oz) | |
| Chronologic or birth age | |
| Time since birth | |
| Gestational age | |
| Estimated time since conception; postconceptional age | |
| Corrected age | |
| Age corrected for prematurity | |
Survival has increased significantly in extremely premature infants. For example, at Washington University, St. Louis, the survival rate of infants weighing less than 800 g (1 lb, 12 oz) at birth increased from 20 percent in 1977 to 49 percent in 1990.3 Because of the increased survival rate, and because many neonatal intensive care units now allow early discharge, family physicians are increasingly likely to provide care to small premature infants after discharge from the hospital.
NICU and Discharge
The family physician should receive information about stabilization, transport and the patient's course in the neonatal intensive care unit (NICU). Discharge planning in the NICU is a multidisciplinary effort that begins with the infant's birth and admission to the unit. Important aspects include the parents' involvement in their infant's care, plans for special needs such as oxygen and apnea monitoring, and teaching the parents special skills such as cardiopulmonary resuscitation.
Social criteria for discharge include the following: (1) confirmation that the parents can provide the basic physical needs of the infant, (2) assurance that the parents are knowledgeable about all of their infant's needs and know how to identify problems that may arise following discharge and (3) a specified plan for continuing health care and parenting support. The stress parents experience while their infant is hospitalized may interfere with their ability to learn; therefore, parents' education often needs to be reinforced after the infant's discharge. Also, parents should have sufficient help at home to support them in the care of their infant.
Most NICUs have no minimum weight requirement for discharge. Medical guidelines for discharge are as follows: (1) body temperature is maintained while the infant is in an open crib, usually at 34 weeks of gestational age or at 2,000 g (4 lb, 6 oz) of weight; (2) the infant feeds by mouth well enough to have a weight gain of 20 to 30 g per day; (3) the infant is not receiving medications that require hospital management, and (4) no recent major changes in medications or oxygen administration have occurred.
Travel
Parents should use an infant car seat right from the first trip, when they take their child home from the NICU. The infant's head or body should not slouch and no straps should be placed around the infant's head or abdomen. Rolled towels or diapers may be used for postural support. The infant should not be placed in the front seat of the vehicle if an air bag on the passenger side could be activated. The infant should be visible to an adult passenger, either directly or through the rearview mirror. Infants with documented desaturation, apnea or bradycardia when in a semi-upright position should travel in a supine or prone position using an alternative safety device.4 If an apnea monitor is prescribed, it should be used while the infant is in the car seat. The baby should not be left alone in the car for even a short time.
Guidelines for air travel based on pulmonary function are summarized in Table 2.
Home and Office Visits
The infant should be seen in the family physician's office within a few days of discharge from the NICU. The physician should review the pregnancy, neonatal course and risk factors for complications. The infant's hospital care should be reviewed to identify problems, and appropriate follow-up consultation should be obtained by working with staff at the NICU. The infant's current status should be noted, including medications, diet and a typical 24-hour day. The infant should be brought to the physician's office every one or two weeks until appropriate weight gain and adaptation to home are documented.
Feeding
Caloric intake, fluid intake, and vitamin and mineral supplementation should be monitored during weekly visits. Nutritional requirements in the NICU are usually 120 kcal per kg per day. This amount usually drops to 100 kcal per kg per day by the time of discharge.
Infants being given a formula with a caloric content of over 24 kcal per ounce are more prone to hyperosmolar dehydration. They should be examined for signs of dehydration, and electrolyte levels should be checked if significant vomiting or diarrhea develops. Most infants do not tolerate formulas containing more than 30 kcal per ounce. (Breast milk and standard formula have about 20 kcal per ounce.) A rule of thumb is to switch to a regular formula when the infant reaches the term date.
Breast milk protects against infection, and breast-fed infants have better developmental scores at 18 months of age than formula-fed infants.5 Mothers who plan to breast feed should use a hospital-quality electric breast pump after the infant's birth to stimulate their breasts. The pumped milk may be fed to the infant. When the infant is able to suckle, the mother should start breast feeding to further encourage milk production. By the time of discharge from the hospital, most premature infants require feedings at least every three hours. Human milk fortifier (a supplement that is added to pumped breast milk) should be used as advised by the NICU.
If there is a question about the mother's supply of breast milk, she should nurse every one and one half to two hours during the day in the first 24 to 48 hours after the infant's discharge to ensure adequate milk production. After this initial period, the infant is normally nursed every two to three hours, or eight to 10 feedings per day.6(p22) Six to eight wet diapers per 24 hours indicate an adequate fluid intake.7 Fluid should not be withheld for longer than four hours.
If the infant refuses to nurse, the mother should try again in 30 minutes to one hour. The mother may encourage the infant to suckle by expressing drops of milk, drawing out the nipples or repositioning the infant. A supplemental nutritional system, which provides fluids through a tube while the infant is breast feeding, is useful for infants who require more calories than the amount they are receiving during regular breast feeding. A lactation consultant can help with many breast-feeding problems.
Solid food should be introduced at four to six months past the infant's due date. The infant's swallowing mechanism will not acceptably handle solid food until two and one half to three and one half months past the infant's due date. Cow's milk should ideally not be started until the infant's age is 12 months past the due date. Small-for-gestational-age infants or sick infants experiencing catch-up growth should be kept on formula or breast milk until their size is within the normal range for gestational age. Sick infants, such as those with bronchopulmonary dysplasia, often require special dietary intervention. In this instance, coordination between the dietary staff and other care providers is necessary.
Vitamins and Minerals
Vitamins D, E and K, and folic acid are especially important for low-birth-weight infants. Except for vitamin D deficiency, vitamin deficiencies are unlikely to occur after discharge from a neonatal intensive care center. The signs and symptoms of vitamin D deficiency are summarized in Table 3.
TABLE 3 Signs and Symptoms of Copper, Zinc and Vitamin D Deficiencies in Infants
| Deficiency | Requirements in preterm infants | Requirements in term infants | Test to confirm deficiency | Signs and symptoms |
|---|---|---|---|---|
| Copper | 100 μg per kg per day | Up to 6 months: 0.4 to 0.6 mg daily | Serum copper, serum ceruloplasmin | Microcytic hypochromic anemia, neutropenia, depigmentation of skin and hair, skin lesions resembling seborrheic dermatitis, anorexia, diarrhea, psychomotor retardation, apneic spells, failure to thrive |
| 6 to 12 months: 0.6 to 0.7 mg daily | ||||
| 1 to 2 years: 0.7 to 1.0 mg daily | ||||
| Zinc | 750 μg per kg per day | 5 mg daily | Plasma zinc | Acrodermatitis enteropathica (dermatitis around body openings and on extremities), diarrhea, alopecia, failure to thrive, increased susceptibility to infection |
| Vitamin D | 400 IU daily | 400 IU daily | Serum vitamin D | Enlarged costochondral junctions (rachitic rosary), bowlegs and knock knees, failure to thrive, hypocalcemia, hypophosphatemia |
Although vitamin deficiencies are rare, all breast-fed infants should probably receive vitamin supplementation during the first year of life.8 One quart of formula or a standard dose of infant vitamins that provides 400 IU of vitamin D per day is advised for both premature and term infants. If the infant is bottle-fed with a standard formula, supplemental vitamins are advised until the infant is ingesting 32 oz of formula a day. If a special formula is used, its vitamin content should be checked to determine if supplementation is required.
Supplemental iron is advised, either as iron-fortified formula or as a liquid, given in a dosage of 2 to 4 mg per kg per day in breast-fed infants or infants receiving low-iron formulas. Iron supplementation should be started two weeks to two months after birth and continued for 12 to 15 months.9 An iron supplement in suspension form, such as Fer-in-Sol, provides 15 mg of iron per 0.6 mL.
The infant should receive 0.25 mg of fluoride supplementation daily if the water in the household is not fluoridated. Many dental problems, including enamel hypoplasia, dental caries and delayed dental development, are reported to be more common in preterm infants than in term infants.10
Although rare, deficiencies of zinc and copper have been reported in premature infants at three to six months of age.11 Zinc deficiency occurs in breast-fed infants whose mothers have a rare metabolic deficiency that prevents the secretion of zinc into their breast milk.12 Zinc and copper requirements and the signs and symptoms of zinc and copper deficiency are listed in Table 3.
Growth
During the first two years of life, growth is plotted using age corrected for prematurity. Growth charts for the “average” premature infant have been designed for this purpose. Special neonatal growth charts are also available for the sick or small-for-gestational-age infant.13,14 After the infant reaches two years of age, a standard growth chart for chronologic age may be used.
Many premature infants have catch-up growth. Catch-up growth is usually first noted in the infant's head circumference, followed by the infant's weight and length. This usually occurs during the first two to three years of life and is maximum at 36 to 40 weeks after conception.6(p54) Little catch-up growth occurs after three years of age. Premature infants with intrauterine growth retardation and no catchup growth have a higher risk of developmental delay and other medical problems than premature infants with a normal growth rate.15 Even in adolescence, children who were born prematurely may be smaller than children not born prematurely.16 Menarche also occurs later in girls who were born prematurely.17 One study showed that women who were born prematurely are more likely to deliver premature infants themselves.18 Another study found a trend for this to happen, but it was not statistically significant.19
Development
The infant's development during the first two years should be plotted from the infant's estimated due date rather than the infant's birth date. The Denver Prescreening Developmental Questionnaire, the Denver Developmental Screening Test and the Gesell Screening Inventory are all accepted tests. Using a standardized developmental test is more important than the choice of test.
Developmental screening does not replace the neurologic examination. Because of this, detailed standardized examinations, such as the Neonatal Neurodevelopmental Examination, have been developed. The latter has been used primarily by developmental pediatricians and pediatric neurologists. It assesses postural reflexes and muscle tone, cranial nerve and motor function, sensory responses and behavior.20 If an abnormal finding is noted, such as neck extensor hypertonia, consultation with a developmental specialist should be considered.
In an attempt to prevent disabilities, a number of intervention programs have been developed and evaluated. These include interventions in the neonatal nursery, at home and at special centers. A review21 of 19 intervention programs for low-birth-weight infants, all evaluated since 1971, found that intervention programs provide support and improve parent–child interaction. Such programs, however, have had only modest success in altering neurodevelopmental outcomes.21 Most NICUs have developmental follow-up programs, and the family physician should encourage the parents to participate in these programs.
Sleep
The premature infant sleeps more total hours than a term infant. Unfortunately for new parents, the premature infant also wakes up more often than the term infant; the average sleep period for a premature infant is shorter than that for a term infant.22 The premature infant may also take several days or weeks to make the transition from the more brightly lit and noisier NICU to the home environment. Soothing music and dimming the lights gradually over several days or weeks may be helpful.
The infant should sleep on his or her back because it is now well established that the prone position is associated with an increased risk of sudden infant death syndrome (SIDS).23 Soft mattresses and other surfaces that could trap exhaled air are also associated with SIDS and should be avoided.
Physical Examination
Vital Signs
Hypertension is more common in premature infants than in term infants. Risk factors include bronchopulmonary dysplasia and renal artery thrombosis from umbilical artery catheterization. Although no firm guidelines exist on the frequency of blood pressure monitoring in premature infants, it is probably reasonable to check the blood pressure by palpation or Doppler in the physician's office at least twice in the first year.24 Normal values for blood pressure in preterm infants are given in Figure 1.
FIGURE 1.
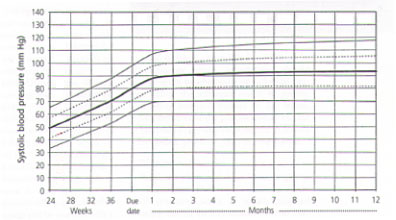
Normal systolic blood pressure in infants, according to age (± 2 standard deviations).
The respiratory rate should be corrected for the infant's due date. In term infants, respirations while awake average 50 ± 19 per minute at the due date and drop to 26.5 ± 8 per minute by 12 months past the due date. Signs of increased effort of breathing, including sternal retractions, use of accessory muscles and abdominal breathing, are as important as the respiratory rate.
The infant's heart rate depends on chronologic age. The heart rate averages 141 beats per minute at birth, peaking to 171 beats per minute at two months of age and gradually declining to 142 beats per minute at one year of age.25
Other Physical Findings
If head flattening occurs, it typically resolves by four years of age, regardless of the type of mattress used in the neonatal period. Oral intubation at birth may cause a high palatal vault, palatal grooving and dental abnormalities.
Premature infants are more likely than term infants to be left-handed or mixed-handed.26
Vision and Hearing
Strabismus is more common in premature infants than in term infants. Because strabismus can be a sign of intraocular pathology, ophthalmologic consultation is generally indicated in infants with strabismus. In many very-low-birth-weight infants, strabismus at six weeks of age resolves by the time the infant reaches nine months of age. Strabismus that is present at nine months of age is likely to persist.
Retinopathy of prematurity occurs in some infants born at 32 weeks of gestation or less, and evaluation for this condition will already have taken place in the NICU. The American Academy of Pediatrics, the American Association for Pediatric Ophthalmology and Strabismus, and the American Academy of Ophthalmology recommend obtaining an initial screening examination at four to six weeks' chronologic age, with follow-up examinations performed as required on the basis of the initial findings.27
Most NICUs obtain brainstem auditory evoked potential testing before the infant's discharge. Hyponatremia, metabolic alkalosis and prolonged mechanical ventilation with high oxygen concentrations are risk factors for hearing loss. The use of an aminoglycoside or furosemide is an additional risk factor.28
The World Health Organization defines hearing loss as an average loss of greater than 25 dB at frequencies of 500, 1,000 and 2,000 Hz, classified according to the less impaired side. Using this definition, about 5 percent of premature infants born before 32 weeks of gestational age have hearing loss by five years of age.29 Parents should be asked if they notice signs of hearing impairment in their infant. The infant's response to loud noise can be checked in the physician's office, and receptive and expressive language can be assessed with a developmental screening tool. Audiologic consultation should be obtained if the parents notice signs of hearing loss or if findings on the screening procedure are abnormal.
Immunizations
The timing of immunizations in the physician's office should be based on the infant's chronologic age, not the gestational age. The only exception is hepatitis B vaccination. The American Academy of Pediatrics Committee on Infectious Diseases has issued a statement indicating that it may be advisable to delay administration of hepatitis B vaccine until the infant weighs 2,000 g (4 lb, 6 oz).30
The full dose of all immunizations should be given. As with term infants, premature infants should be given the acellullar pertussis vaccine when it is available. The acellular pertussis vaccine is preferred over the whole-cell pertussis vaccine in infants with neurologic problems.
The medical record should be reviewed to determine if any immunizations were given in the NICU. Influenza vaccine should be given to infants over six months of age with chronic medical problems, especially lung disease. In all premature infants, consideration should also be given to administering influenza vaccine before the influenza season to parents and other frequent visitors in the home. Administration of the pneumococcal vaccine at two years of age may be beneficial in infants with chronic problems such as lung disease.
RSV Immune Globulin
Respiratory syncytial virus immune globulin intravenous (RSV-IGIV) prevents severe RSV infection when it is administered monthly during the RSV season.31 In two clinical trials, infants who received RSV-IGIV had a 41 percent reduction and a 65 percent reduction in the rate of hospitalization compared with the rate of hospitalization in infants who did not receive the agent. RSV-IGIV should be considered for use in infants under 24 months of age with bronchopulmonary dysplasia who required oxygen therapy in the preceding six months and in infants of a gestational age of 32 weeks or less. Once an RSV infection has developed, RSV-IGIV will not help.32 Because antibodies from RSV-IGIV block immunity to measles vaccine, measles-containing vaccines should not administered for nine months after the last dose of RSV-IGIV.31 No changes are necessary for other routine immunizations.

