
Am Fam Physician. 2001;63(6):1131-1135
A more recent article on endometrial biopsy is available.
“Office Procedures” forms on endometrial biopsy are provided on page 1139.
See patient information handout on endometrial biopsy.
Endometrial biopsy is an office procedure that serves as a helpful tool in diagnosing various uterine abnormalities. The technique is fairly easy to learn and may be performed without assistance. The biopsy is obtained through the use of an endometrial suction catheter that is inserted through the cervix into the uterine cavity. Twirling the catheter while moving it in and out of the uterine cavity enhances uptake of uterine tissue, which is aspirated into the catheter and removed. Endometrial biopsy is useful in the work-up of abnormal uterine bleeding, cancer screening, endometrial dating and infertility evaluation. Contraindications to the procedure include pregnancy, acute pelvic inflammatory disease, and acute cervical or vaginal infections. Postoperative infection is rare but may be further prevented through the use of prophylactic antibiotic therapy. Intraoperative and postoperative cramping are frequent side effects.
Endometrial biopsy is a safe and accepted method for the evaluation of abnormal or postmenopausal bleeding. The procedure is often performed to exclude the presence of endometrial cancer or its precursors (Tables 1 and 2). Office endometrial suction catheters are easy to use, and several have been reported to have diagnostic accuracy that is equal or superior to the dilatation and curettage (D&C) procedure. Suction is generated by withdrawing an internal piston from within the catheter, and the tissue sample is obtained by twirling the catheter while moving it up and down within the uterine cavity.
| Abnormal uterine bleeding |
| Postmenopausal bleeding |
| Cancer screening (e.g., hereditary nonpolyposis colorectal cancer) |
| Detection of precancerous hyperplasia and atypia |
| Endometrial dating |
| Follow-up of previously diagnosed endometrial hyperplasia |
| Evaluation of uterine response to hormone therapy |
| Evaluation of patient with one year of amenorrhea |
| Evaluation of infertility |
| Abnormal Papanicolaou smear with atypical cells favoring endometrial origin |
| Contraindications |
| Pregnancy |
| Acute pelvic inflammatory disease |
| Clotting disorders (coagulopathy) |
| Acute cervical or vaginal infections |
| Cervical cancer |
| Conditions possibly prohibiting endometrial biopsy |
| Morbid obesity |
| Severe pelvic relaxation with uterine descensus |
| Severe cervical stenosis |
Endometrial biopsy is a blind procedure and should be considered part of the evaluation that could include imaging studies, such as hysteroscopy or transvaginal ultrasonography. While a negative study is reassuring, further evaluation is warranted if a patient demonstrates continued abnormal bleeding.
Methods and Materials
EQUIPMENT
Nonsterile Tray for Examination for Uterine Position
Nonsterile gloves
Lubricating jelly
Absorbent pad to place beneath the patient on the examination table
Formalin container (for endometrial sample) with the patient's name and the date recorded on the label
20 percent benzocaine (Hurricaine) spray with the extended application nozzle
Sterile Tray for the Procedure
Place the following items on a sterile drape covering the Mayo stand with the following items placed on top:
Sterile gloves
Sterile vaginal speculum
Uterine sound
Sterile metal basin containing sterile cotton balls soaked in povidone-iodine solution
Endometrial suction catheter
Cervical tenaculum
Ring forceps (for wiping the cervix with the cotton balls)
Sterile 4 × 4 gauze (to wipe off gloves or equipment)
Sterile scissors (if the physician chooses to cut off the catheter tip to deliver the endometrial sample into the formalin container)
Keep sterile cervical dilators available, but do not open the sterile packaging unless the dilators are needed.
Once the physician is sterile-gloved and has placed the speculum, the nurse can spray the benzocaine spray onto the cervix for 5 seconds, avoiding contamination of the sterile speculum with the extended spray nozzle.
Procedure Description
The patient is placed in the lithotomy position and bimanual examination is performed (with nonsterile gloves) to determine the uterine size and position, and whether marked uterocervical angulation exists. Still wearing the nonsterile gloves, the physician can pick up the sterile speculum from the sterile tray and place it in the patient's vagina. Avoid contaminating the sterile instruments on the tray. Once the cervix is centered in the speculum, the cervix can be anesthetized by spraying 20 percent benzocaine spray for 5 seconds and then cleansing it with povidone-iodine solution.
Alternately, the physician can apply sterile gloves, and insert the sterile speculum into the patient's vagina. The physician should minimize contact of the sterile gloves with the nonsterile vulvar tissues. The cervix is centered in the speculum and cleansed with povidone-iodine solution. The gloves can be washed with povidone-iodine solution if contaminated. The nurse can then spray the cervix with the 20 percent benzocaine spray for 5 seconds, avoiding contamination of the sterile speculum with the extended spray nozzle.
The cervix is gently probed with the uterine sound. The cervix often is too mobile to allow for passage of the sound but can be stabilized with the tenaculum. The tenaculum is placed on the anterior lip of the cervix, grabbing enough tissue that the cervix will not lacerate when traction is applied. The author prefers placement of the tenaculum in most cases, for increased safety, and grasps the anterior lip of the cervix with the tenaculum teeth in the horizontal plane.
Pull outward on the tenaculum gently, straightening the uterocervical angle to reduce the chance of posterior perforation. Attempt to insert the uterine sound to the fundus. Occasionally, steady, moderate pressure is required to insert the sound through the closed internal cervical os.
If the uterine sound will not pass through the internal os, consider placement of small Pratt uterine dilators. The smallest size is inserted, followed by insertion of successively larger dilators until the sound passes easily to the fundus. The distance from the fundus to the external cervical os can be measured by the gradations on the uterine sound and generally will be 6 to 8 cm.
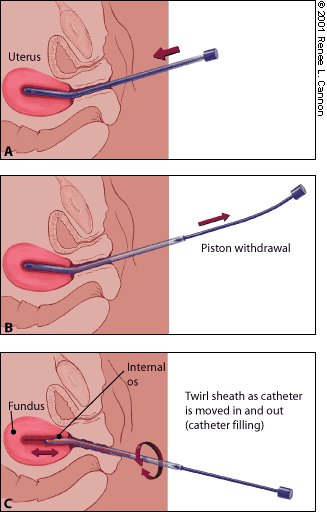
The endometrial biopsy catheter tip is inserted into the cervix, avoiding contamination from the nearby tissues. The catheter tip is then inserted into the uterine fundus or until resistance is felt. Once the catheter is in the uterine cavity, the internal piston on the catheter is fully withdrawn, creating suction at the catheter tip. The catheter tip is moved with an in-and-out motion, but the tip does not exit the endometrial cavity through the cervix, which maintains the vacuum effect. Use a 360-degree twisting motion to move the catheter between the uterine fundus and the internal cervical os (Figure 1). Make at least four up and down excursions to ensure that adequate tissue is in the catheter.
Once the catheter fills with tissue, it is withdrawn, and the sample is placed in the formalin container. To remove the sample from the endometrial catheter, the piston can be gently reinserted, forcing the tissue out of the catheter tip. Some physicians prefer to make a second pass into the uterus with the catheter to optimize tissue sampling. If a second pass is to be made, the catheter should not be contaminated when being emptied of the first specimen.
The tenaculum is gently removed. Pressure can be applied with cotton swabs if the tenaculum sites bleed following removal of the tenaculum. Excess blood and povidone-iodine solution are wiped from the vagina, and the vaginal speculum is removed.
Follow-Up
Normal endometrial tissue may be described as proliferative (estrogen effect or preovulatory) endometrium or secretory (progesterone effect or postovulatory) endometrium. Hormone therapy can be offered to patients with abnormal vaginal bleeding who have normal endometrial tissue on biopsy. If the biopsy is normal but the patient continues to experience excessive vaginal bleeding, further diagnostic work-up should occur.
Atrophic endometrium generally yields scant or insufficient tissue for diagnosis. Hormonal therapy may be considered for patients with atrophic endometrium. Persistent vaginal bleeding should warrant further diagnostic work-up.
Cystic or simple hyperplasia progresses to cancer in less than 5 percent of patients. Most individuals with simple hyperplasia without any atypia can be managed with hormonal manipulation (medroxyprogesterone [Provera], 10 mg daily for five days to three months) or with close follow-up. Most authors recommend a follow-up endometrial biopsy after three to 12 months, regardless of the management strategy.
Atypical complex hyperplasia is a premalignant lesion that progresses to cancer in 30 to 45 percent of women. Some physicians will treat complex hyperplasia with or without atypia with hormonal therapy (medroxyprogesterone, 10 to 20 mg daily for up to three months). Most physicians recommend a D&C procedure to exclude the presence of endometrial carcinoma and consider hysterectomy for complex or high-grade hyperplasia.
Biopsy specimens that suggest the presence of endometrial carcinoma (75 percent are adenocarcinoma) should prompt consideration of referral to a gynecologic oncologist for definitive surgical therapy.
Procedure Pitfalls/Complications
The Catheter Won't Go Up into the Uterus Easily in Perimenopausal Patients. The internal cervical os may be very tight in perimenopausal and menopausal patients. Because of the discomfort that can be created by instrumental cervical dilation, an alternative in older patients is to insert an osmotic laminaria (seaweed) 3-mm dilator in the patient that morning. Osmotic dilators cause gentle, slow opening of the cervix. The osmotic dilator is removed in the afternoon, and then the endometrial biopsy can be easily performed.
Patients Report Cramping Associated with the Procedure. Intraoperative and postoperative cramping frequently accompany instrumentation of the uterine cavity. Preprocedure oral nonsteroidal anti-inflammatory medications, such as ibuprofen (Motrin), can significantly reduce the prostaglandin-induced cramping. Spraying the cervix with a topical anesthetic, such as 20 percent benzocaine, can also help with discomfort.
The Procedure Should Not Be Performed in Pregnant Patients. Endometrial biopsy should not be performed in the presence of a normal or ectopic pregnancy. All patients with the potential for pregnancy should be considered for pregnancy testing prior to the performance of the procedure.
Infection Occurs Following the Procedure. Bacteremia, sepsis and acute bacterial endocarditis have been reported following endometrial biopsy. Because postprocedure bacteremia has been noted, some authors recommend considering antibiotics in post-menopausal women at risk for endocarditis. The risk for infection appears to be small, but some physicians recommend tetracycline, 500 mg twice daily, for four days following the procedure.
The Pathologist Reports That the Specimens Have Insufficient Sample for Diagnosis. Some physicians are less vigorous in obtaining specimens, and a single pass of the catheter may not yield adequate tissue. A second pass can be made with the suction catheter if it is not contaminated when it is emptied after the first pass. The second pass almost always prevents reporting an insufficient sample.
The Tenaculum Causes Discomfort When Applied to the Cervix. Topical anesthesia can reduce the discomfort from the tenaculum. Placement of the tenaculum can make the procedure safer for the patient. The tenaculum stabilizes the cervix and allows the physician to straighten the uterocervical angle. The tenaculum can reduce the chances of posterior perforation when the plastic catheter is inserted through the cervix and then through the thin-walled lower uterine segment.
Physician Training
Endometrial biopsy is a fairly easy technique to learn. Physicians are often comfortable performing the procedure unassisted after two to five precepted procedures. Physicians who perform other gynecologic procedures find that endometrial biopsy is a natural addition to their practice. The American Academy of Family Physicians offers a comprehensive training course in endometrial biopsy for physicians wanting intensive training.
This article is adapted with permission from Zuber TJ. Office procedures. Baltimore: Lippincott Williams & Wilkins, 1999.