Dermatophytes are fungi that require keratin for growth. These fungi can cause superficial infections of the skin, hair, and nails. Dermatophytes are spread by direct contact from other people (anthropophilic organisms), animals (zoophilic organisms), and soil (geophilic organisms), as well as indirectly from fomites. Dermatophyte infections can be readily diagnosed based on the history, physical examination, and potassium hydroxide (KOH) microscopy. Diagnosis occasionally requires Wood's lamp examination and fungal culture or histologic examination. Topical therapy is used for most dermatophyte infections. Cure rates are higher and treatment courses are shorter with topical fungicidal allylamines than with fungistatic azoles. Oral therapy is preferred for tinea capitis, tinea barbae, and onychomycosis. Orally administered griseofulvin remains the standard treatment for tinea capitis. Topical treatment of onychomycosis with ciclopirox nail lacquer has a low cure rate. For onychomycosis, “pulse” oral therapy with the newer imidazoles (itraconazole or flucona-zole) or allylamines (terbinafine) is considerably less expensive than continuous treatment but has a somewhat lower mycologic cure rate. The diagnosis of onychomycosis should be confirmed by KOH microscopy, culture, or histologic examination before therapy is initiated, because of the expense, duration, and potential adverse effects of treatment.
The dryness of the skin's outer layer discourages colonization by microorganisms, and the shedding of epidermal cells keeps many microbes from establishing residence.1 However, the skin's mechanisms of protection may fail because of trauma, irritation, or maceration. Furthermore, occlusion of the skin with nonporous materials can interfere with the skin's barrier function by increasing local temperature and hydration.2 With inhibition or failure of the skin's protective mechanisms, cutaneous infection may occur.
Microsporum, Trichophyton, and Epider-mophyton species are the most common pathogens in skin infections. Less frequently, superficial skin infections are caused by nondermatophyte fungi (e.g., Malassezia furfur in tinea [pityriasis] versicolor) and Candida species. This article reviews the diagnosis and treatment of common dermatophyte infections.
Dermatophytoses
Because dermatophytes require keratin for growth, they are restricted to hair, nails, and superficial skin. Thus, these fungi do not infect mucosal surfaces. Dermatophytoses are referred to as “tinea” infections. They are also named for the body site involved.
Some dermatophytes are spread directly from one person to another (anthropophilic organisms). Others live in and are transmitted to humans from soil (geophilic organisms), and still others spread to humans from animal hosts (zoophilic organisms). Transmission of dermatophytes also can occur indirectly from fomites (e.g., upholstery, hairbrushes, hats).
Anthropophilic organisms are responsible for most fungal skin infections. Transmission can occur by direct contact or from exposure to desquamated cells. Direct inoculation through breaks in the skin occurs more often in persons with depressed cell-mediated immunity. Once fungi enter the skin, they germinate and invade the superficial skin layers.
In patients with dermatophytoses, physical examination may reveal a characteristic pattern of inflammation, termed an “active” border (Figure 1). The inflammatory response is usually characterized by a greater degree of redness and scaling at the edge of the lesion or, occasionally, blister formation. Central clearing of the lesion may be present and distinguishes dermatophytoses from other papulosquamous eruptions such as psoriasis or lichen planus, in which the inflammatory response tends to be uniform over the lesion (Figure 2).
FIGURE 2.
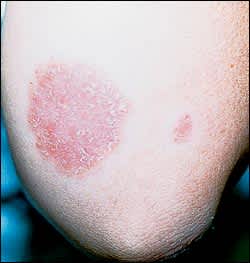
Annular lesion on the elbow, with a silvery scale and no central clearing. Potassium hydroxide microscopy of the lesion was negative. Although the lesion resembles tinea corporis, the presence of similar lesions on the extensor surfaces of the knee and a positive family history confirmed the diagnosis of psoriasis.
The location of the lesions also can help identify the pathogen. A dermatophytosis can most likely be ruled out if a patient has mucosal involvement with an adjacent red, scaly skin rash. In this situation, the more probable diagnosis is a candidal infection such as perlèche (if single or multiple fissures are present in the corners of the mouth) or vulvovaginitis or balanitis (if lesions are present in the genital mucosa).
Potassium hydroxide (KOH) microscopy aids in visualizing hyphae and confirming the diagnosis of dermatophyte infection. Other diagnostic modalities include Wood's lamp examination, fungal culture, and skin or nail biopsy (Table 1).2,3
Tinea Capitis
Tinea capitis, the most common dermatophytosis in children, is an infection of the scalp and hair shafts.4 Transmission is fostered by poor hygiene and overcrowding, and can occur through contaminated hats, brushes, pillowcases, and other inanimate objects. After being shed, affected hairs can harbor viable organisms for more than one year.
Tinea capitis is characterized by irregular or well-demarcated alopecia and scaling. When swollen hairs fracture a few millimeters from the scalp, “black dot” alopecia is produced. Tinea scalp infection also may result in a cell-mediated immune response termed a “kerion,” which is a boggy, sterile, inflammatory scalp mass. Cervical and occipital lymphadenopathy may be prominent.
Before 1950, most tinea capitis cases in North America were caused by fluorescent Microsporum species (bright blue-green). Today, about 90 to 95 percent of tinea scalp infections in adults and children are caused by Trichophyton tonsurans, which does not fluoresce.4,5 Therefore, Wood's lamp examination has become a less useful diagnostic test for tinea capitis.
Tinea capitis is generally identified by the presence of branching hyphae and spores on KOH microscopy (Table 1). If hyphae and spores are not visualized, Wood's lamp examination can be performed. If KOH microscopy and Wood's lamp examinations are negative, fungal culture may be considered when tinea capitis is strongly suspected.
TABLE 1 Diagnostic Methods for Dermatophyte Infections
| Potassium hydroxide (KOH) microscopy | |
|---|---|
| Value: aids in visualizing hyphae and confirming the diagnosis of dermatophyte infection | |
| Procedure: obtain scale from the active border of a lesion, pull out several loose hairs from the affected area or, in the case of nails, obtain subungual debris. A moist cotton swab rubbed vigorously over the active border of a lesion works as well as a scalpel blade and is safer. Transfer the scale, hair, or debris to a glass slide, and add a few drops of 10% to 20% KOH. For nail material or hair, gently warm the slide. The wet-mount preparation is then examined under a microscope (×400) with back-and-forth rotation of the focus knobs. This technique aids the visualization of hyphae (branching, rod-shaped filaments of uniform width with lines of separation [septa]). In tinea capitis, the hair shaft may be uniformly coated with minute dermatophyte spores. | |
| Wood's lamp examination (ultraviolet light) | |
| Value: generally of limited usefulness, because most dermatophytes currently seen in the United States do not fluoresce; may have value in the following situations: | |
| For diagnosing a brown, scaly rash in the scrotum or axilla: erythrasma, caused by the bacteriumCorynebacterium minutissimum, fluoresces a brilliant coral red, whereas tinea cruris or cutaneous candidal infections do not fluoresce. | |
| For diagnosing tinea (pityriasis) versicolor, which fluoresces pale yellow to white | |
| For diagnosing tinea capitis caused by two zoophilic Microsporum species that fluoresce blue-green (a minor percentage of tinea capitis cases in North America) | |
| Fungal culture | |
| Value: slow and expensive, but useful to confirm the diagnosis of onychomycosis when long-term oral therapy is being considered | |
| Procedure*: Skin, nail, or hair scrapings are sent in a sterile container for inoculation on Sabouraud's dextrose agar by a hospital or reference laboratory. The culture usually takes 7 to 14 days to be declared positive; it must be held 21 days to be declared negative. | |
| Skin or nail biopsy | |
| Value: may guide treatment decisions when the diagnosis is difficult to establish, a dermatophyte infection has not responded to previous treatment, or KOH microscopy is negative in a patient with dystrophic nails | |
*—Dermatophyte Test Medium is a commercially available medium supplied in a form that is ready for direct inoculation in the office. The yellow medium has a phenol red indicator that turns pink in the presence of the alkaline metabolic products of dermatophytes, usually within 6 to 7 days. The medium must be discarded after 2 weeks, because saprophytes produce a similar change. The Clinical Laboratory Improvement Act classifies this as a moderately complex test. Because of the cost of materials, performance, and qualified laboratory personnel, this test is rarely included in family physicians' office laboratories.
Alternatively, clinical features can point to the diagnosis. In one study,6 tinea capitis was confirmed by culture in 92 percent of children who had at least three of the following clinical features: scalp scaling, scalp pruritus, occipital adenopathy, and diffuse, patchy, or discrete alopecia.
When scaling and inflammation are prominent, other diagnoses to consider include seborrheic dermatitis (no hair loss), atopic dermatitis (lesions in flexural folds of the neck, arms, or legs), and psoriasis (nail changes and silvery scales on the knees or elbows). When alopecia is prominent, diagnoses to rule out include alopecia areata (complete, rather than patchy, hair loss), traction alopecia (history of tight hair braiding), and trichotillomania (hairs of differing lengths and a history of obsessive hair manipulation).
Topical treatment is not effective for tinea capitis. Systemic antifungal therapy is required to penetrate the hair follicles. Griseofulvin (Grisactin, Gris-PEG) is the only agent that the U.S. Food and Drug Administration (FDA) has labeled for the treatment of tinea capitis. Although griseofulvin remains the gold standard,7 it is a less than ideal agent for several reasons8,9: resistant organisms require dosage increases to effect a cure; treatment must be continued for six to 12 weeks; relapse rates are high because of rapid clearance of the drug from the skin with the cessation of therapy; and the liquid form for young children is a bitter-tasting solution.
Compared with griseofulvin, ketoconazole (Nizoral) is no more effective and has the potential for adverse hepatic effects and drug interactions.10 In one study involving a small number of children, treatment with itraconazole (Sporanox), in a dosage of 3 to 5 mg per kg per day for four weeks, resulted in clinical and mycologic cure rates of 90 to 100 percent.11 [Evidence level B, nonrandomized clinical trial] Fluconazole (Diflucan) and terbinafine (Lamisil) are promising agents; randomized, comparative studies with griseofulvin should clarify their role in the treatment of tinea capitis.12 One randomized trial13 in patients with tinea capitis caused by Trichophyton species showed that treatment with terbinafine, fluconazole, or itraconazole for two weeks was as effective as six weeks of griseofulvin therapy.
Adjunctive topical therapy with selenium sulfide (e.g., Exsel), ketoconazole, or povidone iodine (Betadine) lotion or shampoo (applied for five minutes twice weekly) is useful to decrease shedding of viable fungi and spores12,14,15; over-the-counter 1 percent selenium sulfide shampoo works as well as the prescription 2.5 percent strength.15 [Reference15: Evidence level A, randomized controlled trial (RCT)]
Tinea Corporis
Tinea corporis, or ringworm, typically appears as single or multiple, annular, scaly lesions with central clearing, a slightly elevated, reddened edge, and sharp margination (abrupt transition from abnormal to normal skin) on the trunk, extremities, or face (Figure 1). The border of the lesion may contain pustules or follicular papules. Itching is variable.
The diagnosis of tinea corporis is based on clinical appearance and KOH examination of skin scrapings from the active edge. The differential diagnosis includes nummular eczema, pityriasis rosea, Lyme disease, tinea versicolor, contact dermatitis, granuloma annulare, and psoriasis (Figure 2).
Previous topical corticosteroid use can alter the appearance of the lesions, so that raised edges with central clearing are not present. Corticosteroid use may also be a factor in the development of Majocchi's granuloma, a deep follicular tinea infection that usually involves the legs and is more common in women.3
Treatment of tinea corporis usually consists of measures to decrease excessive skin moisture and the use of topical antifungal creams (Table 2).16–19 Rarely, widespread infections may require systemic therapy.
TABLE 2 Topical Therapy for Dermatophyte Infections*
| Agent | Formulation* | Frequency of application | |
|---|---|---|---|
| Allylamines | |||
| Naftifine (Naftin) | 1% cream | Once daily | |
| 1% gel | Once or twice daily | ||
| Terbinafine (Lamisil) | 1% cream or solution | Once or twice daily | |
| Benzylamine | |||
| Butenafine (Mentax) | 1% cream | Once or twice daily | |
| Imidazoles | |||
| Clotrimazole (Lotrimin) | 1% cream, solution, or lotion | Twice daily | |
| Econazole (Spectazole) | 1% cream | Once daily | |
| Ketoconazole (Nizoral) | 1% cream | Once daily | |
| 1% shampoo | Twice weekly | ||
| Miconazole (Micatin) | 2% cream, spray, lotion, or powder | Twice daily | |
| Oxiconazole (Oxistat) | 1% cream or lotion | Once or twice daily | |
| Sulconazole (Exelderm) | 1% cream or lotion | Once or twice daily | |
| Miscellaneous | |||
| Ciclopirox (Loprox) | 1% cream or lotion | Twice daily | |
| Tolnaftate (Tinactin) | 1% cream, solution, or powder | Twice daily | |
*—Topical therapy is appropriate for tinea corporis, tinea pedis, tinea cruris, tinea manuum, and tinea faciei.
Tinea Barbae
Tinea barbae involves the skin and coarse hairs of the beard and mustache area. This dermatophyte infection occurs in adult men and hirsute women. Because the usual cause is a zoophilic organism, farm workers are most often affected. Tinea barbae may cause scaling, follicular pustules, and erythema (Figure 3).
The differential diagnosis includes bacterial folliculitis, perioral dermatitis, pseudofolliculitis barbae, contact dermatitis, and herpes simplex. One clue to the diagnosis is that hair removal is painless in tinea barbae but painful in bacterial infections.
Like tinea capitis, tinea barbae is treated with oral antifungal therapy. Treatment is continued for two to three weeks after resolution of the skin lesions.
Tinea Faciei
Tinea faciei tends to occur in the non-bearded area of the face. The patient may complain of itching and burning, which become worse after sunlight exposure. Some round or annular red patches are present. Often, however, red areas may be indistinct, especially on darkly pigmented skin, and lesions may have little or no scaling or raised edges. Because of the subtle appearance, this dermatophytosis is sometimes known as “tinea incognito.”20
The differential diagnosis includes seborrheic dermatitis, rosacea, discoid lupus erythematosus, and contact dermatitis. A high index of suspicion, along with a KOH microscopy of scrapings from the leading edge of the skin change, may help in establishing the diagnosis. Treatment is similar to that for tinea corporis.
Tinea Manuum
Tinea manuum is a fungal infection of one or, occasionally, both hands (Figure 4). It often occurs in patients with tinea pedis. The palmar surface is diffusely dry and hyperkeratotic. When the fingernails are involved, vesicles and scant scaling may be present, and the condition resembles dyshidrotic eczema. The differential diagnosis includes contact dermatitis, psoriasis, and callus formation.
Topical antifungal therapy and the application of emollients containing lactic acid (e.g., Lac-Hydrin Cream) are effective.21 Relapses may be frequent if onychomycosis or tinea pedis is not resolved.
Tinea Cruris
Tinea cruris, frequently called “jock itch,” is a dermatophyte infection of the groin. This dermatophytosis is more common in men than in women and is frequently associated with tinea pedis. Tinea cruris occurs when ambient temperature and humidity are high. Occlusion from wet or tight-fitting clothing provides an optimal environment for infection.
Tinea cruris affects the proximal medial thighs and may extend to the buttocks and abdomen. The scrotum tends to be spared. Patients with this dermatophytosis frequently complain of burning and pruritus. Pustules and vesicles at the active edge of the infected area, along with maceration, are present on a background of red, scaling lesions with raised borders. The feet should be evaluated as a source of the infection. Conditions that need to be distinguished from tinea cruris are listed in Table 3.20,21
Adjunctive treatment can include a low-dose corticosteroid (e.g., 2.5 percent hydrocortisone ointment [Cortaid]) for the first few days. Rarely, systemic antifungal therapy is needed for refractory tinea cruris. Patient education on avoiding prolonged exposure to moisture and keeping the affected area dry is important.
TABLE 3 Differential Diagnosis of Tinea Cruris
| Condition | Distinguishing features |
|---|---|
| Candidal intertrigo | Uniformly red, with no central clearing; satellite lesions |
| Erythrasma | Uniformly brown and scaly, with no active edge; fluoresces a brilliant coral red |
| Mechanical intertrigo | Sharp edge, no central clearing or scale |
| Psoriasis | Silvery scale and sharp margination; pitted nails; knee, elbow, and scalp lesions |
| Seborrheic dermatitis | Greasy scales; scalp (dandruff) and sternal involvement |
Tinea Pedis
Tinea pedis, or athlete's foot, has three common presentations. The interdigital form of tinea pedis is most common. It is characterized by fissuring, maceration, and scaling in the interdigital spaces of the fourth and fifth toes. Patients with this infection complain of itching or burning. A second form, usually caused by Trichophyton rubrum, has a moccasin-like distribution pattern in which the plantar skin becomes chronically scaly and thickened, with hyperkeratosis and erythema of the soles, heels, and sides of the feet. The vesiculobullous form of tinea pedis (Figure 5) is characterized by the development of vesicles, pustules, and sometimes bullae in an inflammatory pattern, usually on the soles. The differential diagnosis includes contact dermatitis, eczema, and pustular psoriasis.
Streptococcal cellulitis is a potential complication of all three forms of tinea pedis. Streptococcal infection of normal skin is unlikely. However, the presence of fungal maceration and fissuring permits streptococci to colonize the web spaces between the toes in patients with tinea pedis. The clinical features of symptomatic athlete's foot are a result of the interaction of fungi and bacteria.
Treatment of tinea pedis involves application of an antifungal cream to the web spaces and other infected areas. Infrequently, systemic therapy is used for refractory infections. In several studies, twice-daily application of the allylamine terbinafine resulted in a higher cure rate than twice-daily application of the imidazole clotrimazole (Lotrimin; 97 percent versus 84 percent), and at a quicker rate (one week for terbinafine versus four weeks for clotrimazole).16,17 [Reference16: Evidence level A, RCT] A pharmacoeconomic analysis18 of tinea treatments found topical terbinafine to be more cost-effective than imidazole or ciclopirox cream (Loprox).
When marked inflammation and vesicle formation occur and signs of early cellulitis are present, the addition of a systemic or topical antibiotic with streptococcal coverage is warranted.
Reinfection is common, especially if onychomycosis is present. Nail infections should be treated. In addition, footwear should be disinfected, and patients with tinea pedis should avoid walking barefoot in public areas such as locker rooms. Other measures to reduce recurrence include controlling hyperhidrosis with powders and wearing absorbent socks and nonocclusive shoes.
Tinea Unguium
Tinea unguium, a dermatophyte infection of the nail, is a subset of onychomycosis, which also may be caused by yeast and non-dermatophyte molds.19 Risk factors for this infection include aging, diabetes, poorly fitting shoes, and the presence of tinea pedis.
Onychomycosis accounts for about 40 to 50 percent of nail dystrophies.22,23 The differential diagnosis includes trauma, lichen planus, psoriasis, nail-bed tumor, peripheral vascular disease, atopic dermatitis, contact dermatitis, and yellow nail syndrome.
Because onychomycosis requires expensive, prolonged therapy (three to four months for fingernail infections and four to six months for toenail infections), the diagnosis should be confirmed before treatment is initiated24,25 (Table 4). Periodic acid-Schiff staining with histologic examination of the clipped, distal free edge of the nail and attached subungual debris is the most sensitive diagnostic method and is painless for patients.25
Tinea unguium, especially of the toenails, is difficult to eradicate. Topical agents have low efficacy. Mycologic cure rates for ciclopirox (Penlac) nail lacquer, applied daily for up to 48 weeks, have ranged from 29 to 47 percent.26 [Evidence level A, meta-analysis] Oral treatment with griseofulvin must be continued for 12 to 24 months, and ketoconazole carries a risk of hepatotoxicity. Fluconazole has not been studied extensively in the treatment of onychomycosis and is not labeled by the FDA for this indication.
TABLE 4 Confirming the Diagnosis of Onychomycosis
| Method | Technique | Sensitivity (%)24,25 |
|---|---|---|
| Potassium hydroxide (KOH) microscopy | Scrape the most proximal subungual area; examine on a KOH-treated, warmed glass slide (see Table 1). | 50 to 60 |
| Fungal culture | Scrape the most proximal subungual area; send scrapings in a sterile container to a hospital or reference laboratory, or spread scrapings on Dermatophyte Test Medium (see Table 1). | 20 to 70 |
| Periodic acid- Schiff staining | Clip the distal edge of the nail, along with attached subungual debris; place the most proximal sample in formalin for histologic examination in a hospital or reference laboratory. | 90 to 95 |
Mycologic and clinical cure rates are similar for 12 weeks of treatment with itraconazole in a dosage of 200 mg per day and terbinafine in a dosage of 250 mg per day.27 Itraconazole costs more for the same regimen. Continuous terbinafine therapy has a better mycologic cure rate than intermittent or “pulse” terbinafine therapy, in which 500 mg of terbinafine is given once daily for seven days of each of four months (94 percent versus 80 percent); however, continuous treatment is more expensive ($700 versus $400). Intermittent itraconazole therapy, in a dosage of 400 mg per day for seven days of each of four months, and intermittent terbinafine therapy are similarly effective.

