
Am Fam Physician. 2006;74(3):420-426
Patient information: See related handout on whooping cough, written by the author of this article.
Author disclosure: Nothing to disclose.
Bordetella pertussis is a highly contagious bacterium known to cause pertussis (whooping cough) and is transmitted via airborne droplets. Although childhood vaccination has dramatically reduced reported pertussis cases, the incidence of the disease has increased over the past 20 years, most notably in previously immunized adolescents and adults. Pertussis should be suspected in patients of all ages with cough who meet the clinical criteria for the disease. Diagnostic tests currently approved by the U.S. Food and Drug Administration for pertussis infection have low sensitivity. Regardless of test results, physicians should treat clinically suspected pertussis with antimicrobials and report cases to their state health department. A 14-day erythromycin regimen has been the treatment of choice; however, shorter-course macrolide antibiotics (e.g., azithromycin, clarithromycin) may be as effective with fewer adverse effects and better adherence to therapy. The recently recommended tetanus toxoid, reduced diphtheria toxoid, and acellular pertussis (Tdap) vaccine for adolescents and adults may decrease the incidence of pertussis in infants—the group at the greatest risk of pertussis complications.
Bordetella pertussis, a highly communicable gram-negative coccobacillus, causes pertussis (whooping cough). B. pertussis is an exclusively human pathogen that is transmitted via airborne droplets. The organism produces antigens that can cause local cell damage and may mediate systemic symptoms. B. pertussis is considered a difficult bacterium to grow in the laboratory.1
| Clinical recommendation | Evidence rating | Comments |
|---|---|---|
| Patients with suspected early pertussis should be tested with nasopharyngeal culture and polymerase chain reaction assay. | C | Consensus-based guideline8 |
| Antihistamines, steroids, beta agonists, and immunoglobulins are not routinely recommended for pertussis treatment. | A | Systematic review20 |
| Erythromycin is a recommended therapy to effectively eradicate Bordetella pertussis and reduce transmission rates. | A | Systematic review21 |
| Azithromycin (Zithromax) and clarithromycin (Biaxin) are recommended therapies for eradicating B. pertussis that are as effective as erythromycin but with better adherence to therapy. | A | Systematic review21 ; consistent findings from randomized-controlled trials25–27 |
| Close household contacts of patients with pertussis should be treated with antibiotics to prevent disease. | B | Consensus guidelines8,22,28 ; limited studies show that secondary cases were prevented29,30 ; a systematic review showed insufficient evidence to determine benefit21 |
| Adolescents should receive Tdap vaccination instead of a Td booster to reduce the incidence of pertussis. | C | Expert opinion34 ; disease-oriented evidence35–38 ; no outcome validation studies exist on preventing infantile pertussis |
| Adults should receive a one-time Tdap booster instead of a Td booster to reduce the incidence of pertussis. | C | Expert opinion34 ; disease-oriented evidence35–38 ; no outcome validation studies exist on preventing infantile pertussis |
Before the introduction of pertussis vaccinations, the average yearly rate of reported pertussis in the United States was 157 per 100,000 persons, with cyclic peaks occurring every two to five years,2,3 although underreporting may mean that the actual rates were much higher.1 After vaccinations were introduced in the 1940s, incidence of pertussis fell dramatically to less than one per 100,000 persons by 19704; however, rates have increased modestly since 1980,2,3 with significant increases in adolescent and adult populations.2–5
Vaccination Limitations
In the prevaccine era, more than 93 percent of reported pertussis cases occurred in children younger than 10 years.1 In 2003, most cases occurred in persons 10 years or older (Figure 1).5 As a result, previously vaccinated adolescents and adults whose immunity has decreased have become reservoirs for pertussis infection.1
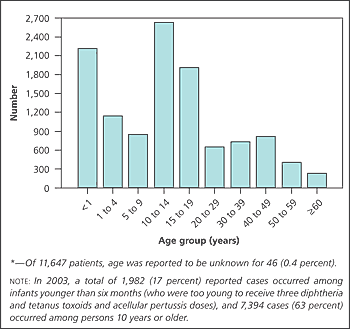
Immunity from the vaccine is not yet complete in the first year of life. Childhood pertussis vaccination has not created the herd immunity that might protect incompletely immunized infants. Despite vaccination, the incidence of pertussis infection in children younger than one year has increased to more than one half of all childhood pertussis cases (Figure 15).3,5 Pertussis is the only vaccine-preventable disease associated with increasing deaths in the United States, climbing from 4 deaths in 1996 to 17 in 20015 and occurring almost exclusively in infants younger than one year.2,4
Complications
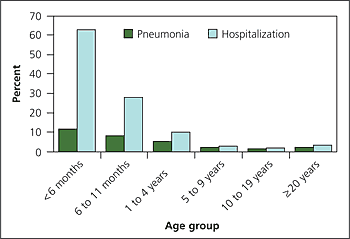
Pertussis can lead to hospitalization, pneumonia, dehydration, weight loss, sleep disturbance, seizures, and, rarely, encephalopathy or death.1 These complications vary depending on patient age (Figure 26). Most pertussis related hospitalizations occur in the first year of life.4 Young infants are at the greatest risk of secondary bacterial pneumonia, the most common cause of pertussis-related deaths.4,7 Acute dehydration and malnutrition occur in patients with cough that limits food and fluid intake. Cerebral hypoxia from severe paroxysms1 can cause seizures and encephalopathy. Refractory pulmonary hypertension can be a late sequela in infants with pertussis.1,7
Clinical Presentation
| Stage | Duration(weeks) | Symptoms | Comment |
|---|---|---|---|
| Catarrhal | One to two | Lacrimation, low-grade fever, malaise, mild conjunctival inflammation, rhinorrhea, late-phase nonproductive cough |
|
| Paroxysmal | One to six | Paroxysms (bursts of coughing during a single exhalation) followed by an inspiratory “whooping” sound, post-tussive cyanosis, and emesis |
|
| In infants younger than six months (especially those younger than four weeks): apnea, bradycardia, prolonged cough, poor feeding, no paroxysms | |||
| Convalescent | Two to 12 | Paroxysms gradually improve but recur with respiratory infections |
|
STAGES OF PERTUSSIS
The catarrhal stage consists of nonspecific cold-like symptoms. After one to two weeks, patients develop coughing (i.e., bursts of coughing during a single exhalation) followed by an inspiratory “whooping” sound.1,8 An audio recording of pertussis-associated coughing is available athttp://www.immunizationed.org. Paroxysms can be associated with post-tussive cyanosis and emesis. Infants younger than six months may present with severe cough of any duration, poor feeding, apnea, or bradycardia without coughing paroxysms. The convalescent stage includes slow resolution of paroxysms, although coughing may persist for several months.1,8
The cold-like symptoms common in the catarrhal stage often are initially misdiagnosed as viral upper respiratory tract infection. When coughing persists or paroxysms accompany these symptoms, the differential diagnosis includes infection with pathogens associated with community-acquired pneumonia (e.g., Chlamydia pneumoniae, Mycoplasma pneumoniae) and pertussis. Pertussis is most contagious in the catarrhal and early paroxysmal stages.
ATYPICAL PRESENTATION
Previously vaccinated adolescents and adults may have less severe paroxysmal symptoms.9,10 Children who are completely vaccinated have shorter courses of illness than incompletely vaccinated children.9,11 Girls older than three years may have more severe paroxysms than boys of the same age.11 The younger the child, the more severe paroxysms tend to be11; however, infants may not have paroxysms at all.1
An atypical presentation can cause a misdiagnosis during the early, most contagious stages of pertussis.12 If adolescents and adults (who often have minimal symptoms) are not treated, they may unknowingly expose susceptible infants to the disease.13 Despite atypical presentations, when carefully questioned, most adolescents and adults with pertussis report paroxysmal symptoms.14 Current public health initiatives focus on reducing the risk of infantile pertussis through education about early symptom recognition and vaccination of adolescents and adults.
Diagnostic testing
Tests used to confirm B. pertussis are listed in Table 2.15 Although each test has advantages and disadvantages, proper technique is important. A polyester swab of the nasopharynx is more effective than a swab of the throat or anterior nostril. The polyester swab should be inserted into the base of a nostril and left in the posterior pharynx for 10 seconds before withdrawing. Nasopharyngeal aspirates have higher bacterial recovery than swabs, and specimens can be split for multiple tests8; however, the equipment required for aspirates is not widely available.
| Test | Sensitivity (%) | Specificity (%) | PPV | NPV | Comments |
|---|---|---|---|---|---|
| Bordetella pertussis culture | 15 | 100 | 100 | 88 | Requires special culture media; takes seven to 12 days to receive results; up to 80 percent sensitive only in early disease; sensitivity is affected by antibiotics; CDC recommends using with polymerase chain reaction assay to confirm a pertussis diagnosis |
| Polymerase chain reaction assay | 94 | 97 | 84 | 99 | Can confirm diagnosis quickly (one to two days); expensive; not affected by antibiotics; no single test is universally accepted; not widely available; CDC recommends using with culture to confirm the diagnosis |
| Direct fluorescent antibody test | 52 | 98 | 83 | 92 | Requires specially trained personnel; can confirm diagnosis quickly; high false-positive rates; can be used when cultures are negative; not recommended by the CDC |
| Serology | Variable | Variable | — | — | No single test is universally accepted; not standardized nationally; not recommended by the CDC |
CULTURES
B. pertussis is difficult to grow in cultures. Direct agar inoculation or careful transport in special media before inoculation is required.8 Cultures can take seven to 12 days to confirm growth and are less sensitive after antimicrobial therapy is initiated.1 Because of its high false-negative rate, this technique is a poor confirmatory test when used alone late in the disease course.
POLYMERASE CHAIN REACTION ASSAY
A polymerase chain reaction (PCR) assay to detect B. pertussis is more sensitive than culture later in the disease course and is similar in specificity.15,16 A PCR assay can confirm pertussis infection quickly (within one or two days) and is not affected by antimicrobial therapy.17 Because false-positive results may occur with PCR assay, the Centers for Disease Control and Prevention (CDC) recommends testing patients with suspected pertussis using PCR assay and cultures.8
DIRECT FLUORESCENT ANTIBODY TEST
Direct fluorescent antibody (DFA) testing has been the traditional technique for detecting B. pertussis. Although DFA testing has high specificity and provides results quickly, its sensitivity is lower than PCR assay, and specially trained laboratory technicians are required to perform the test. The CDC does not recommend DFA testing.8
SEROLOGY
The role of serology for detecting pertussis has not been defined. Serologic tests are used most often in epidemiologic studies1,8 and can detect immune responses to various antigens and toxins produced by B. pertussis. The CDC does not recommend this test because it is not standardized nationally.8
Case Reporting
Physicians in the United States, Guam, and Puerto Rico are legally required to report pertussis cases to state health departments.18 The CDC classifies pertussis cases as clinical, confirmed, or probable (Table 38). Physicians should report pertussis when it is clinically suspected and should not await laboratory confirmation.18 The CDC recommends testing and treating patients with clinical or probable pertussis regardless of test results.8 Testing, treatment, and reporting should be considered in patients of all ages presenting with a cough lasting more than two weeks that develops a paroxysmal quality, inspiratory whooping, or post-tussive emesis; and in infants with severe cough, apnea, or bradycardia for any length of time.19
| Case | Definition | ||
|---|---|---|---|
| Clinical | Acute cough for 14 days plus one of the following: paroxysmal cough, post-tussive emesis, inspiratory “whooping,” and no other apparent cause | ||
| or | |||
| In an outbreak setting: acute cough for 14 days | |||
| Confirmed | Patient's illness meets criteria for “clinical case” plus one of the following: positive PCR assay, epidemiologic linkage to a laboratory- confirmed (PCR assay or culture) case | ||
| or | |||
| Acute cough illness of any duration and positive Bordetella pertussis culture | |||
| Probable | Patient's illness meets criteria for “clinical case” plus all of the following: negative PCR assay, negative B. pertussis culture, no epidemiologic linkage to a laboratory-confirmed (PCR assay or culture) case | ||
Treatment
The effectiveness of symptom-reducing treatments (e.g., antihistamines, steroids, beta agonists, immunoglobulins) is unclear, and these treatments have potentially serious adverse effects. A systematic review20 showed little evidence to justify their use for pertussis. Antibiotics have not been shown to reduce disease duration after the paroxysmal stage begins, but they can decrease transmission risk.21 Because pertussis is highly contagious, antibiotic prophylaxis is recommended to control outbreaks.
ANTIBIOTICS
The American Academy of Pediatrics (AAP) recommends a 14-day erythromycin regimen22 to treat pertussis, although a seven-day regimen may be as effective.23 Erythromycin can cause gastrointestinal side effects (e.g., nausea, emesis, diarrhea)24 and increases the risk of pyloric stenosis in infants younger than two months.24
Newer generation macrolides (e.g., azithromycin [Zithromax] and clarithromycin [Biaxin]) have similar bacterial eradication rates as erythromycin21,25,26 with less risk of side effects24–27 and better adherence.25,27 The CDC recommends erythromycin, azithromycin, or clarithromycin as preferred agents, although it only recommends azithromycin for neonates because limited data28 suggest it may be the safest choice in this group.28 Trimethoprim/sulfamethoxazole (Bactrim, Septra) has been shown to reduce pertussis transmission and is an alternative treatment for patients who are allergic to macrolides.21 Other antibiotics, such as ampicillin, have not been shown to reduce pertussis transmission or symptoms.21 Table 421–23,28 summarizes antibiotic therapies for pertussis.
PROPHYLAXIS
About 80 percent of susceptible persons become infected with pertussis after close contact with an infected household member.8 Studies1 of household contacts indicate that infection is common even without symptoms. If patients are not treated during the catarrhal stage, they are considered contagious until three weeks after the paroxysmal stage ends or until five days after starting antibiotics.8,22
The CDC and AAP advocate antibiotic prophylaxis to control pertussis outbreaks.8,22 This approach is controversial,21,29,30 however, and no systematic review has evaluated the effectiveness of prophylaxis for preventing new pertussis infections. Optimal doses and duration of antibiotic prophylaxis are uncertain, but the CDC recommends the same drugs and dosing as it does for antibiotic treatment (Table 421–23,28).8 In the United States, where pertussis vaccination rates are high, antibiotic prophylaxis is advised only in those who are in close contact with persons with pertussis, particularly incompletely immunized children or adults who are in close contact with high-risk children.8,22 Local and state health departments are responsible for managing outbreaks and have protocols that are recommended for physician use.
Prevention
Although pertussis vaccination has significantly reduced reported pertussis rates, its protectiveness is short-lived and incomplete. Immunity begins to decline four to 12 years after vaccination, causing adolescent and adult susceptibility.31 Therefore, neonates are susceptible to pertussis infection because they have not yet been immunized and they receive little passive immunity from their susceptible mothers.
The estimated effectiveness of original whole-cell pertussis vaccines was about 85 percent.32 Rare adverse reactions included hypotonic, hyporesponsive episodes; high fever; seizures; and anaphylaxis. Currently approved acellular vaccines produce fewer adverse reactions than whole-cell vaccines and have similar effectiveness.33 Two tetanus toxoid, reduced diphtheria toxoid, and acellular pertussis (Tdap) vaccines recently were approved by the U.S. Food and Drug Administration for use in adolescents and adults. Boostrix is approved for 10- to 18-year-olds, and Adacel is approved for 11- to 64-year-olds.34 These booster vaccines produce antibodies35 that may decline at the same rate following natural B. pertussis infection.36 Boostrix has been shown to be 62 to 92 percent effective against pertussis in adolescents and adults,37 although the duration of this protection is unknown. Routine vaccination of adolescents and adults may be cost-effective and improve overall health outcomes.38
The CDC's Advisory Committee on Immunization Practices recommends the Tdap vaccine for 11- to 12-year- olds rather than the tetanus-diphtheria (Td) booster currently given to adolescents.34 The committee also recommends the Tdap vaccine for 13- to 18-year-olds who did not receive an 11- to 12-year Td booster and for 11- to 18-year- olds who were vaccinated with Td.34 The committee recommends a single-dose Tdap booster rather than the Td booster for 19- to 65-year-olds.39 Future studies are needed to determine if this strategy will reduce pertussis-related morbidity and mortality.