
Am Fam Physician. 2006;74(12):2069-2074
Patient information: See related handout on vasectomy, written by the authors of this article.
Author disclosure: Nothing to disclose.
Vasectomy remains an important option for contraception. Research findings have clarified many questions regarding patient selection, optimal technique, postsurgical follow-up, and risk of long-term complications. Men who receive vasectomies tend to be non-Hispanic whites, well educated, married or cohabitating, relatively affluent, and have private health insurance. The strongest predictor for wanting a vasectomy reversal is age younger than 30 years at the time of the procedure. Evidence supports the use of the no-scalpel technique to access the vasa, because it is associated with the fewest complications. The technique with the lowest failure rate is cauterization of the vasa with or without fascial interposition. The ligation techniques should be used cautiously, if at all, and only in combination with fascial interposition or cautery. A single postvasectomy semen sample at 12 weeks that shows rare, nonmotile sperm or azoospermia is acceptable to confirm sterility. No data show that vasectomy increases the risk of prostate or testicular cancer.
Although vasectomy is the safest and most inexpensive option for permanent sterilization,1 female sterilization is used about three times as often (27 versus 9.2 percent of couples who use contraception; Table 1).2 In addition, there was a 3 percent overall decrease in the use of sterilization (male and female) and an increase in the use of less-reliable forms of contraception from 1995 to 2002.2,3 Family physicians should be aware of these trends and provide patients with up-to-date information about contraception options.
| Method | Prevalence (%)* |
|---|---|
| Oral contraceptive | 30.6 |
| Female sterilization | 27.0 |
| Male condom | 18.0 |
| Vasectomy | 9.2 |
| Three-month injectable | 5.3 |
| Withdrawal | 4.0 |
| Other† | 4.0 |
| Intrauterine device | 2.0 |
Patient Selection
Vasectomy reversal is relatively expensive and its success rate is highly variable.4 Given these considerations, physicians should counsel men about the permanence of vasectomy and the risk factors associated with reversal requests. Two studies showed that change in marital status was the most common reason for wanting a reversal.5,6 A case-control study showed that age younger than 30 years at the time of the vasectomy (odds ratio [OR] = 12.5) and having a spouse who worked outside of the home (OR = 2.1) were associated with later requests for reversal.7 The authors conceded that the latter association likely was multifactorial and culturally based. There was no association between requests for reversal and the patient’s religion, occupation, or number of marriages. Having no children at the time of the vasectomy was associated with a lower risk of a reversal request (OR = 0.1).7
Knowing the characteristics associated with men who choose vasectomy may assist physicians with patient education. A cross-sectional survey of men (n = 719) who received vasectomies at a nationally representative sample of medical practices showed that being married or cohabitating at the time of the procedure and earning an annual household income of $25,000 or more were most highly associated with the decision to have a vasectomy (91.4 and 92.9 percent of sterilized men, respectively).8 Men who undergo vasectomy also tend to be non-Hispanic whites, be well educated, and have private health insurance.8
Racial disparities were notable in black and Hispanic men. Although these groups represent 11.0 and 9.8 percent of the U.S. male population, respectively, the study showed that only 4.5 percent of vasectomies were for black men and 4.8 percent were for Hispanic men.8 Therefore, men who choose vasectomy are not a representative sample of the general U.S. male population. It is unknown if these findings represent cultural barriers to male sterilization or financial barriers from lower socioeconomic status.
The study also explored the most important reasons given for choosing a vasectomy over other forms of contraception.8 The most common reason for choosing vasectomy was that this method represented the surest way to prevent having more children (49.9 percent), followed by having a wife or partner who disliked other forms of contraception (12.3 percent) and the patient himself disliking other forms of contraception (10.0 percent). When asked why they chose vasectomy over female sterilization, most men responded that vasectomy was simpler and safer.8
Technique
Technique options can be divided into three components of vasectomy: accessing the vasa, disrupting the vasa, and closing the surgical site (scrotum).
ACCESSING THE VASA
Accessing the vasa can be achieved with a single or double incision to the scrotum using a scalpel, or with the no-scalpel technique using special instruments. No-scalpel vasectomy has been shown to reduce the risk of complications (i.e., bleeding and infection) compared with the incision technique (Table 2).9–11 Two randomized controlled trials showed a 1.7- to 6.8-fold reduction in bleeding and hematomas and a 1.6- to 7.5-fold reduction in infections with the no-scalpel technique compared with the incision technique.9,11
DISRUPTING THE VASA
Disruption of the vasa can be performed numerous ways (Figure 112–18). Simple suture ligation with excision likely is the most common method worldwide,12 although this method has been shown to be less effective than previously reported.19 In addition, there is increasing concern that ligating the vasa, regardless of technique, often causes tip necrosis and sloughing; this can lead to recanalization of a vasal segment.20 A large randomized trial that included patients in seven countries noted vasectomy failure (i.e., live sperm present 24 weeks after the procedure) in 12.7 percent of men who received suture ligation and excision alone from an experienced physician.14
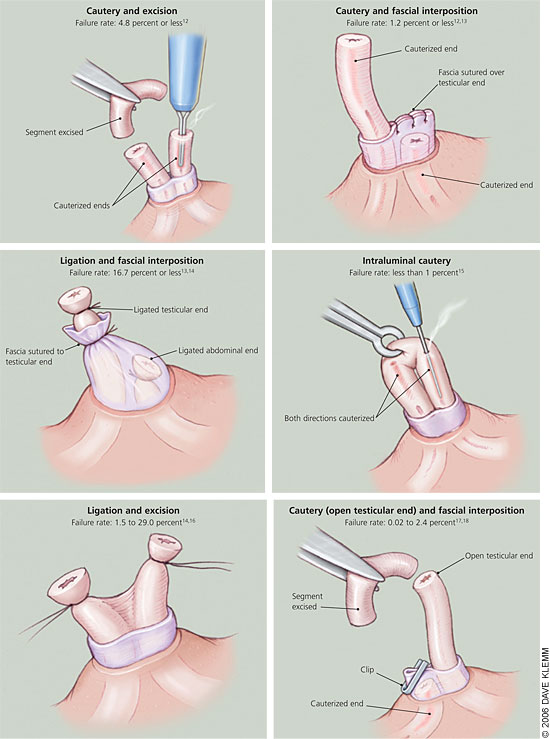
A retrospective Canadian study comparing cautery and fascial interposition with clip ligation and excision reported an 8.7 percent failure rate in patients who received clip ligation and excision.17 The seven-country study showed that adding fascial interposition to suture ligation and excision decreased the failure rate to 5.9 percent.14 These data confirm added protection from fascial interposition. The use of cautery, alone or with fascial interposition, appears to significantly decrease failure rates. The Canadian study showed that cautery with fascial interposition had a 0.3 percent failure rate.17 An observational study reported an early failure rate (i.e., live sperm 12 weeks after vasectomy) of 1 percent with cautery alone.13
Intraluminal cautery without separation of the vasa currently is the preferred method practiced at Marie Stopes International health centers (multinational family planning clinics where 100,000 vasectomies have been performed worldwide).15 These clinics report a less than 1 percent failure rate with this technique.15 Two studies have compared thermal and electric cautery.18,21 One study showed a nonsignificant increase in failure rates with electric cautery,18 and the other study showed histologic evidence of more reliable occlusion with thermal cautery.21 Although these studies provide some evidence that thermal cautery is more effective than electric, no firm conclusions about optimal route or type of cautery can be made.
Leaving the testicular end open after the vas disruption phase of vasectomy has the theoretical advantage of decreasing post-vasectomy vasal pressure; however, it also causes a sperm granuloma at the open end.22 Early studies reported a significant decrease in chronic pain with this technique, implying that most chronic pain resulted from congestive epididymitis rather than a granuloma.22,23 A large retrospective study of 3,761 men who received vasectomies showed no difference in postoperative scrotal pain between open- and closed-ended techniques.17 Ultimately, physicians should use study data along with their own experience and training to determine which vasal disruption technique is the best option for their patients.
SCROTAL CLOSURE
Scrotal closure can be accomplished with clips, clamps, sutures, or cyanoacrylate tissue adhesive. The incision also may be left open to heal by secondary intention. Data comparing these methods are limited; therefore, further data are needed before recommendations regarding closure technique can be made.
Follow-up Care
Follow-up after a vasectomy should include one or more semen samples to ensure that recanalization has not occurred.24 Unfortunately, many men do not comply with this recommendation. A case series reported that less than one half (42 percent) of men who received a vasectomy provided a postvasectomy semen sample. Of those who provided an initial sample, only 25 percent provided a subsequent sample.25
Two studies showed that the median time to loss of sperm motility was three weeks after vasectomy and the median time to azoospermia was 10 weeks.26,27 A finding of rare, nonmotile sperm after 12 weeks reliably predicts long-term sterility28; therefore, subsequent monthly samples after 12 weeks is recommended only for those with motile sperm.29 Patients who want earlier confirmation of sterility should be counseled that a second sample is more likely to be needed if the first sample is given before 12 weeks.
Long-term Complications
Some men may be concerned that vasectomy is linked to prostate cancer. However, a population-based, case-control study including men with newly diagnosed prostate cancer showed no association between prostate cancer and vasectomy (relative risk = 0.94),30 and a meta-analysis provided no evidence of an association.31 Studies also have shown that there is no measurable association between vasectomy and testicular cancer.32,33