Effective management of vaginal bleeding in late pregnancy requires recognition of potentially serious conditions, including placenta previa, placental abruption, and vasa previa. Placenta previa is commonly diagnosed on routine ultrasonography before 20 weeks' gestation, but in nearly 90 percent of patients it ultimately resolves. Women who have asymptomatic previa can continue normal activities, with repeat ultrasonographic evaluation at 28 weeks. Persistent previa in the third trimester mandates pelvic rest and hospitalization if significant bleeding occurs. Placental abruption is the most common cause of serious vaginal bleeding, occurring in 1 percent of pregnancies. Management of abruption may require rapid operative delivery to prevent neonatal morbidity and mortality. Vasa previa is rare but can result in fetal exsanguination with rupture of membranes. Significant vaginal bleeding from any cause is managed with rapid assessment of maternal and fetal status, fluid resuscitation, replacement of blood products when necessary, and an appropriately timed delivery.
Vaginal bleeding after midpregnancy is associated with maternal and fetal risks. Maternal morbidity may be caused by acute hemorrhage and operative delivery, and the fetus may be compromised by uteroplacental insufficiency and premature birth. Optimal management of late pregnancy bleeding depends on accurate identification of the cause and a timely intervention specific to its severity.
SORT: KEY RECOMMENDATIONS FOR PRACTICE
| Clinical recommendation | Evidence rating | References |
|---|---|---|
| A sterile speculum examination can be performed safely before ultrasonographic evaluation of placental localization, but a digital examination should be avoided until placenta previa is excluded by ultrasonography. | C | 1 |
| Women with bleeding in late pregnancy who are Rh negative should receive Rho(D) immune globulin (Rhogam) after performance of a Kleihauer-Betke test to determine the appropriate dose. | C | 2 |
| Placenta previa is a common incidental finding on second trimester ultrasonography and should be confirmed in the third trimester. | A | 6 |
| Corticosteroids should be administered to women who have bleeding from placenta previa at 24 to 34 weeks' gestation. | A | 18 |
| Outpatient management of placenta previa is appropriate in selected patients who do not have active bleeding and who can rapidly access a hospital with operative labor and delivery services. | A | 19, 20 |
| Magnetic resonance imaging of the pelvis may help confirm a diagnosis of invasive placenta and identify organ involvement associated with placenta percreta. | C | 28 |
| Treatment of preeclampsia with magnesium sulfate decreases the risk of placental abruption and improves maternal outcomes. | A | 38 |
A = consistent, good-quality patient-oriented evidence; B = inconsistent or limited-quality patient-oriented evidence; C = consensus, disease-oriented evidence, usual practice, expert opinion, or case series. For information about the SORT evidence rating system, see page 1135 or https://www.aafp.org/afpsort.xml.
Initial Assessment of Vaginal Bleeding
The history, a physical examination, ultrasonography for placental location, and a brief period of observation usually differentiate minor from serious causes of vaginal bleeding. Cervical dilatation during normal labor is commonly accompanied by a small amount of blood or blood-tinged mucus (bloody show), and many pregnant women experience spotting or minor bleeding after sexual intercourse or a digital vaginal examination. Cervicitis, cervical ectropion, cervical polyps, and cervical cancer are possible underlying causes. Evaluation with a sterile speculum may be performed safely before ultrasonographic evaluation of placental location; however, digital examination should not be performed unless ultrasonography excludes a placenta previa.1
Management of Antepartum Hemorrhage
The initial management of significant bleeding in late pregnancy is similar regardless of the etiology. Visual estimates of blood loss should be recorded but may be inaccurate or fail to account for concealed hemorrhage. Hypotension, tachycardia, and maternal symptoms of hemodynamic instability are ominous indicators, and women with these signs require immediate intravenous access, fluid resuscitation, and the availability of blood products. Baseline laboratory tests include hematocrit, platelet count, fibrinogen level, coagulation studies, blood type, and antibody screen. Women who are Rh negative should receive Rho(D) immune globulin (Rhogam); a Kleihauer-Betke test should be performed to determine the appropriate dose.2 Continuous fetal monitoring is recommended.3 Decelerations or loss of variability may resolve with adequate maternal resuscitation; however, a persistently nonreassuring fetal heart rate tracing may require urgent cesarean delivery before the etiology of the hemorrhage is established.
Placenta Previa
Placenta previa is a placental implantation that overlies or is within 2 cm (0.8 inches) of the internal cervical os.4 The placenta is described as a complete previa when it covers the os and as a marginal previa when the edge lies within 2 cm of the os. When the edge is 2 to 3.5 cm (1.4 inches) from the os, the placenta may be described as low lying5 (Figure 1). Transvaginal ultrasonography allows precise assessment of the distance between the internal os and the placental edge (Figure 2).
Figure 1.
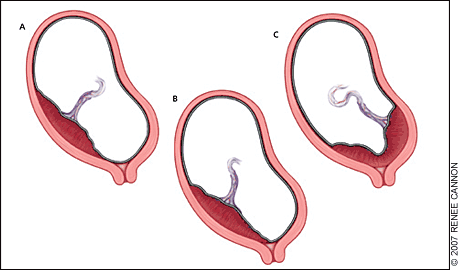
Types of placenta previa: (A) low lying, (B) marginal, and (C) complete.
Figure 2.
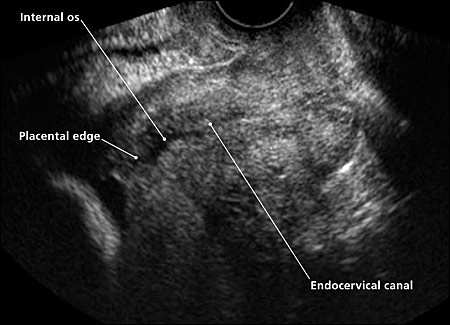
Transvaginal ultrasonogram of a complete posterior placenta previa. The placental edge covers the internal os.
Reprinted with permission from the University of New Mexico Department of Obstetrics and Gynecology.
The incidence of placenta previa increases with age, parity, tobacco use, and number of cesarean deliveries.6,7 Additional risk factors are listed in Table 1.6,8–11
Table 1 Risk Factors for Major Causes of Vaginal Bleeding in Late Pregnancy
| Placenta previa6 |
| Chronic hypertension |
| Multiparity |
| Multiple gestations |
| Older age |
| Previous cesarean delivery |
| Tobacco use |
| Uterine curettage |
| Placental abruption8–10 |
| Chronic hypertension |
| Multiparity |
| Preeclampsia |
| Previous abruption |
| Short umbilical cord |
| Sudden decompression of an overdistended uterus |
| Thrombophilias |
| Tobacco, cocaine, or methamphetamine use |
| Trauma: blunt abdominal or sudden deceleration |
| Unexplained elevated maternal alpha fetoprotein level |
| Uterine fibroids |
| Vasa previa11 |
| In vitro fertilization |
| Low-lying and second trimester placenta previa |
| Marginal cord insertion |
| Multiple gestation |
| Succenturiate-lobed and bilobed placentas |
CLINICAL PRESENTATION
Placenta previa is a common incidental finding on second trimester ultrasonography. It is evident on approximately 4 percent of ultrasound studies performed at 20 to 24 weeks' gestation12 but is present at term in only 0.4 percent of pregnancies.6 When placenta previa is suspected on transabdominal ultrasonography, transvaginal ultrasonography should be performed.13 Transvaginal ultrasonography is safe14 and is more accurate than transabdominal ultrasonography because the placental edge and the internal os, located deep within the bony pelvis, often are obscured by acoustic shadows from the symphysis or the fetus. Visualization of relevant structures is poor on 50 percent of transabdominal ultrasonograms, and additional transvaginal ultrasonography changes the diagnosis in 26 percent of cases.13 The migration of the placenta away from the lower uterine segment is caused by the growth of placental trophoblasts toward the fundus, with its richer blood supply, and the development or elongation of the lower uterine segment.
Symptomatic placenta previa usually manifests as vaginal bleeding in the late second or third trimester, often after sexual intercourse. The bleeding typically is painless unless labor or placental abruption occurs. This initial sentinel bleed usually is not sufficient to produce hemodynamic instability or to threaten the fetus in the absence of cervical instrumentation or cervical digital examination.
MANAGEMENT
Women with bleeding from placenta previa generally are admitted to the hospital for an initial assessment.15 Because most neonatal morbidity and mortality associated with placenta previa results from complications of prematurity, the main therapeutic strategy is to prolong pregnancy until fetal lung maturity is achieved16 (Figure 3). Tocolytic agents may be used safely to prolong gestation if vaginal bleeding occurs with preterm contractions.17 Corticosteroids should be administered to women who have bleeding from placenta previa at 24 to 34 weeks' estimated gestation.18
Figure 3. Management of Placenta Previa
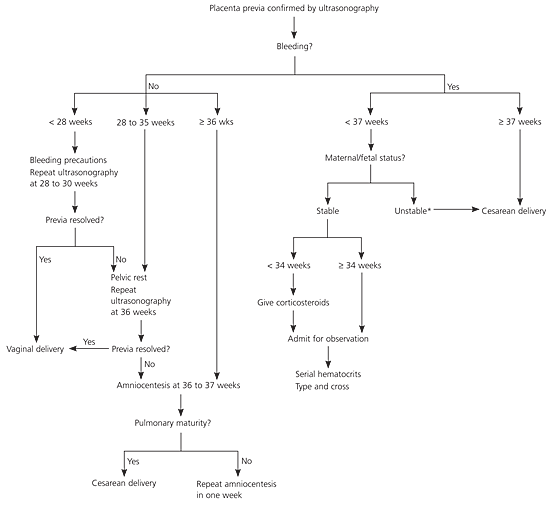
Algorithm for the management of placenta previa.
*— Severe bleeding, hemodynamic instability, or nonreassuring fetal heart tones without improvement after fluid resuscitation.
A Cochrane review found few randomized trials of interventions for placenta previa.19 Outpatient management is appropriate for selected patients who do not have active bleeding and who can rapidly access a hospital with operative labor and delivery services.20 Cervical cerclage has been proposed as a means of prolonging pregnancies complicated by placenta previa, because bleeding occurs in late pregnancy as the placenta is sheared from a lengthening lower uterine segment and dilating cervix.21 The Cochrane meta-analysis found that cerclage decreased the risk of premature birth before 34 weeks (relative risk = 0.45; 95% confidence interval, 0.23 to 0.87); however, it is recommended that additional studies of cerclage be performed before this clinical practice is introduced.19
The likelihood of a previa persisting until term increases if the previa is complete, if it is present at a later gestational age, or if there is a history of cesarean delivery (Table 2).22 The length by which the placenta overlaps the internal os at 18 to 23 weeks is highly predictive for the persistence of placenta previa.23,24 If the overlap is less than 1.5 cm (0.6 inches) at 18 to 23 weeks, placenta previa typically resolves23; if the overlap is 2.5 cm (1 inch) or greater at 20 to 23 weeks, persistence to term is likely.24 Women with asymptomatic previa in the second trimester can continue normal activities until follow-up ultrasonography is performed at approximately 28 weeks. Women with persistent placenta previa in the third trimester should report any bleeding and abstain from intercourse and use of tampons.
Table 2 Likelihood of Placenta Previa Persisting to Term by Type, History of Cesarean Delivery, and Gestational Age at Detection
| Type of previa | Previous cesarean delivery? | Percentage of previas persisting to delivery with ultrasonographic detection at: | ||
|---|---|---|---|---|
| 15 to 19 weeks | 24 to 27 weeks | 32 to 35 weeks | ||
| Complete | No | 20 | 56 | 90 |
| Complete | Yes | 41 | 84 | 89 |
| Partial | No | 6 | 12 | 39 |
| Partial | Yes | 7 | 40 | 63 |
| Overall | 12 | 49 | 73 | |
note: With complete previa, the placenta covers the entire cervical os; with partial previa, the inferior placental edge partially covers or reaches the margin of the os.
Adapted with permission from Dashe JS, McIntire DD, Ramus RM, Santos-Ramos R, Twickler DM. Persistence of placenta previa according to gestational age at ultrasound detection. Obstet Gynecol 2002;99(5 pt 1):695.
Because placenta previa may resolve close to term, it is recommended that no decision on mode of delivery be made until after ultrasonography at 36 weeks.25 Women whose placental edge is 2 cm or more from the internal os at term can expect to deliver vaginally unless heavy bleeding ensues.4 Women whose placenta is located 1 to 2 cm (0.4 to 0.8 inches) from the os may attempt vaginal delivery in a facility capable of moving rapidly to cesarean delivery if necessary.4 Women with a nonbleeding placenta previa may have amniocentesis at 36 to 37 weeks to document pulmonary maturity before a scheduled cesarean delivery.16 Management of placenta previa is summarized in Figure 3.
Women with a history of cesarean delivery who present with placenta previa or a placenta located at the site of the previous incision should be evaluated for potential placenta accreta with color-flow Doppler by an experienced sonographer26 (Figure 4). The risks of placenta accreta, increta, and percreta increase with the number of previous cesarean deliveries.27 Magnetic resonance imaging of the pelvis may help confirm the diagnosis of an invasive placenta and delineate organ involvement in women with a placenta percreta.28 A suspected placenta accreta necessitates preparation for possible cesarean hysterectomy, including appropriate surgical expertise and availability of blood.
Placental Abruption
Placental abruption is the separation of the placenta from the uterine wall before delivery. Abruption is the most common cause of serious vaginal bleeding, occurring in 1 percent of pregnancies. Neonatal death occurs in 10 to 30 percent of cases.8 Approximately 50 percent of placental abruptions occur before 36 weeks' gestation, resulting in adverse outcomes secondary to prematurity.29
The incidence of abruption increased between 1979 and 2001, possibly as a result of rising rates of hypertension and stimulant abuse and increased diagnosis by ultrasonography.9 Risk factors associated with abruption include tobacco or cocaine use, chronic hypertension, preeclampsia, thrombophilias, abdominal trauma, and abruption in a previous pregnancy.8 Sudden uterine decompression after rupture of membranes or delivery of a first twin may precipitate placental abruption.10 Additional risk factors are listed in Table 1.6,8–11
CLINICAL PRESENTATION
Placental abruption typically manifests as vaginal bleeding, uterine tenderness or back pain, and evidence of fetal distress. Preterm labor, growth restriction, and intrauterine fetal death also may occur.30 The fundus often is tender to palpation, and pain occurs between contractions. Bleeding may be completely or partially concealed or may be bright, dark, or intermixed with amniotic fluid. Disseminated intravascular coagulation may result from the release of thromboplastin into the maternal circulation with placental separation.31 This occurs in about 10 percent of abruptions and is more common with fetal death.31 A chronic form of abruption may manifest as recurrent vaginal bleeding with episodic pain and contractions.8
MANAGEMENT
Because the unpredictable nature of abruption does not allow for controlled trials, management remains empiric. A Cochrane review found no randomized controlled trials assessing interventions for placental abruption that met inclusion criteria.32 Initial management includes rapid stabilization of maternal cardiopulmonary status and assessment of fetal well-being. Delay can be fatal to the fetus; 30 percent of perinatal deaths in one case series occurred within two hours of admission.33 Definitive management should never be delayed for ultrasound confirmation because ultrasonography is not reliable in diagnosing abruption34 (Figure 5). Acute blood clots and the placenta are hyperechoic on ultranography and difficult to distinguish from one another.
Maternal stabilization requires serial evaluation of the hematocrit and coagulation studies to determine whether disseminated intravascular coagulation is present.8 Tocolysis generally is contraindicated except in mild abruption before 34 weeks' gestation, when it may be used to allow administration of corticosteroids.35 A nonreassuring fetal heart tracing necessitates rapid, usually cesarean, delivery.31,36 A decision-to-delivery interval of 20 minutes or less resulted in improved neonatal outcomes in a case-control study of severe abruption.36 Occasionally, abruption occurs during the second stage, and an operative vaginal delivery may be attempted. When fetal death occurs secondary to abruption, vaginal delivery should be the goal.37 Serial ultrasonography and antepartum surveillance in the third trimester are appropriate for women with chronic abruption because of the potential for uteroplacental insufficiency.30
PREVENTION
The incidence of placental abruption may be decreased by cessation of tobacco, cocaine, or amphetamine use, and appropriate care for hypertensive disorders of pregnancy. One trial demonstrated a reduction in the incidence of abruption with intrapartum treatment of preeclampsia using magnesium sulfate.38
Vasa Previa
Vasa previa is the velamentous insertion of the umbilical cord into the membranes in the lower uterine segment resulting in the presence of fetal vessels between the cervix and presenting part. Although it is uncommon (the incidence is 1 in 2,500 births), it is important for physicians to be familiar with vasa previa because rapid intervention is essential for fetal survival.11 Studies demonstrate a 33 to 100 percent rate of perinatal mortality secondary to vasa previa.11 Risk factors for vasa previa include in vitro fertilization, placenta previa, and bilobed and succenturiate-lobed placentas (Table 16,8–11 ).11
Vasa previa typically manifests as onset of hemorrhage at the time of amniotomy or spontaneous rupture of membranes. The hemorrhage is fetal blood, and exsanguination can occur rapidly because the average blood volume of a term fetus is approximately 250 mL.39 Rarely, vessels are palpated in the presenting membranes, prohibiting artificial rupture and vaginal delivery.
If fetal heart tones are reassuring, a blood sample from the vaginal vault may be obtained to check for fetal blood cells or fetal hemoglobin. The Apt test is most commonly used; it is based on the resistance of fetal hemoglobin to denaturation by alkaline agents and can be performed in the labor and delivery unit.40 Delivery should not be deferred for confirmation of fetal blood in women with severe hemorrhage or when fetal heart tones are nonreassuring.
There are no strategies for primary prevention of vasa previa; however, hemorrhage theoretically is preventable with antenatal screening for women at high risk and cesarean delivery at 37 to 38 weeks when vasa previa is present. Screening is carried out with transvaginal color-f low Doppler to identify the presence of vessels in the fetal membranes. Although it has been suggested for women at increased risk,41 there is no evidence that screening in a general population changes outcomes, and because the condition is rare (one diagnosis per 5,215 screenings), this approach is cost prohibitive.42
ALSO is a registered trademark of the American Academy of Family Physicians.

