A more recent article on managing menopausal symptoms is available.
Am Fam Physician. 2010;82(7):801-807
Related letter: "Do Vaginal Estrogens Increase Endometrial Proliferation Risk?"
Patient information: See related handout on treating menopausal symptoms, written by the authors this article.
Author disclosure: Nothing to disclose.
The results of recent large clinical trials have led physicians and patients to question the safety of menopausal hormone therapy. In the past, physicians prescribed hormone therapy in an attempt to improve overall health and prevent cardiac disease. Hormone therapy appears to increase the risk of breast cancer when used for more than three to five years; therefore, regulatory agencies now advise that physicians prescribe it only to treat menopausal symptoms such as hot flashes and vaginal atrophy, with the smallest effective dosage and for the shortest possible duration. Although estrogen is the most effective treatment for hot flashes, alternatives such as venlafaxine and gabapentin are effective for some patients. Herbal formulations such as dong quai, ginseng, kava, and dietary soy, among others, do not appear to benefit patients more than placebo. In contrast to systemic estrogen therapy, topical estrogen therapy for vulvovaginal atrophy is more appealing for certain patients because it does not require the addition of a progestogen for endometrial protection. Some have advocated selective estrogen reuptake modulators as alternatives to hormone therapy for the prevention of menopausal osteoporosis. The decision to use either therapy depends on clinical presentation and a thorough evaluation of the risks and benefits, because both have potential detrimental health effects and both are linked to an increased risk of venous thromboembolism.
Menopause is a normal, physiologic transition when a woman stops producing eggs and ovarian function diminishes, sometimes causing bothersome symptoms such as hot flashes or vaginal dryness. The media attention to recent clinical trials of treatments for menopausal symptoms has provided women with a large amount of information, some of it contradictory. This makes it challenging for patients to understand which treatment options are available, effective, and safe. In the past, physicians typically prescribed hormone therapy for menopause, whereas now they increasingly prescribe medications to treat specific symptoms. The U.S. Food and Drug Administration (FDA) issued a boxed warning for hormone preparations and, with the American College of Obstetricians and Gynecologists (ACOG), advises that hormone therapy be prescribed at the lowest effective dosages and for the shortest possible duration.1,2 Dozens of medications, dominated by hormone formulations, are available to treat menopausal symptoms. When counseling menopausal patients, physicians should understand the benefits and risks of hormone therapy and alternative treatments, and the key differences among the various delivery systems.
| Clinical recommendation | Evidence rating | References |
|---|---|---|
| Bioidentical and compounded formulations | ||
| The FDA and the American College of Obstetricians and Gynecologists haveissued warnings about the lack of data on safety and effectiveness forcompounded hormone preparations. | C | 20, 21 |
| Bone health | ||
| Estrogen therapy is an option to reduce the risk of postmenopausal osteoporoticfractures; although it is not FDA-approved for treatment of osteoporosis, it isan option when nonhormone therapies are not tolerated. | B | 3, 4, 8 |
| Cancer risk | ||
| Use of combined estrogen and progestogen therapy for more than three to five years increases the risk of breast cancer. | B | 3, 8 |
| Estrogen therapy alone does not appear to increase the risk of breast cancer. | B | 4 |
| Combined estrogen and progestogen therapy appears to reduce the risk of colorectal cancer. The change in risk, if any, is uncertain for estrogen therapy. | B | 3, 4 |
| Because unopposed estrogen therapy in women with a uterus increases the risk of endometrial carcinoma, progestogen therapy is recommended for women with an intact uterus who are taking estrogen. | C | 8 |
| Dosage and duration | ||
| Hormone therapy is an option for women with menopausal symptoms, using the lowest effective dosage for the shortest possible duration, with periodic reevaluation. | C | 1, 2 |
| Attempts to wean hormone therapy should be considered annually after three to five years of treatment. | C | 3, 10 |
| Heart disease | ||
| Hormone therapy is not recommended for cardiac protection in women of any age and does not treat existing heart disease. | A | 2, 8 |
| Early hormone therapy (at the initiation of menopause) is reasonable for relief of menopausal symptoms in women at low risk of cardiovascular disease. | B | 2, 3 |
| Beginning hormone therapy in a woman's 60s or 70s increases the risk of coronary heart disease; this should be reserved for symptomatic women who cannot tolerate nonhormone medications and who have had a thorough discussion of the risks and benefits with their physician. | A | 3, 8 |
| Stroke | ||
| Combined estrogen and progestogen therapy and estrogen therapy alone increase the risk of ischemic stroke, particularly during the first one to two years after initiation of therapy. | A | 3, 4 |
| There does not appear to be an increased risk of stroke in women who begin hormone therapy between 50 and 59 years of age, although information is inconsistent. | B | 3 |
| Vasomotor symptoms | ||
| Estrogen is the most effective treatment for menopausal vasomotor symptoms and is FDA-approved for this indication. | A | 14 |
| VTE | ||
| Estrogen therapy alone and combined estrogen and progestogen therapy increase the risk of VTE, particularly during the first one to two years of use, although the risk is less for women who are younger than 60 years or are taking estrogen alone. | A | 3, 4, 9 |
| Observational data, but not data from randomized trials, suggest that transdermal estrogen may confer less risk of VTE than oral estrogen. | B | 16 |
| Vulvovaginal symptoms | ||
| Local estrogen therapy is the most effective treatment for moderate to severe vulvar and vaginal atrophy, and is FDA-approved for this indication; it does not require the addition of a progestogen. | A | 8 |
Risks and Benefits of Hormone Therapy: Focus on Cardiac Health
In 2002, the Women's Health Initiative (WHI) clinical trial was published, causing patients and physicians to question the safety of menopausal hormone therapy.3 Before this study, many patients took hormones in an attempt to improve overall health, prevent future cardiac disease, and treat menopausal symptoms, in part because results of observational studies showed apparent protection from cardiac disease. The WHI study compared a combined oral regimen consisting of conjugated equine estrogen (Premarin) and medroxyprogesterone acetate (Provera) with placebo, and found that the combined regimen increased the risk of coronary artery disease, breast cancer, stroke, and venous thromboembolism (VTE). This large study of approximately 16,000 women also found a decreased risk of colorectal cancer, hip fractures, and total fractures.3
Critics of the 2002 WHI trial believe it is not appropriate to generalize results to all menopausal women, because the population studied may not represent women who typically use hormone therapy. Specifically, the average age of participants was 63 years, more than a decade older than the average age of the menopausal transition (51 years), when women are more likely to start hormone therapy.
Other studies have helped refine information about hormone therapy. The 2004 publication of a conjugated estrogen–only arm of the WHI trial in women without a uterus showed that those taking estrogen alone had no significant change in coronary heart disease or breast cancer.4 Similar to the WHI trial that used the combination of estrogen and progestogen, the estrogen-only WHI trial showed an increase in strokes and a small increase in the risk of VTE.3,4
A subsequent post-hoc reanalysis of data from both WHI trials found nonsignificant trends that starting hormone therapy at the beginning of menopause might not increase cardiac risk compared with a later start.5 Although others have found that starting estrogen therapy right after menopause decreases physiologic measures of coronary atherosclerosis and that estrogen alone reduces coronary artery calcification scores compared with placebo, those are disease-oriented outcomes that do not necessarily equate to improved patient-oriented outcomes.6,7 This information has renewed interest in the possible role of hormone therapy for cardiac protection in recently menopausal women. Conversely, women who begin therapy in their 60s or 70s have an increased risk of coronary heart disease; beginning therapy at this age should be reserved for symptomatic women who cannot tolerate nonhormone medications and who have had a thorough discussion of the risks and benefits with their physician.3,8 Counseling patients about the use of hormone therapy is challenging; until future research helps to better stratify risks and benefits, it is currently recommended that patients not use hormone therapy to prevent or treat cardiac disease.2,8
Noncardiac Risks and Benefits of Hormone Therapy
Many patients are interested in potential noncardiac health benefits and risks of using hormone therapy. Colorectal cancer is one of the most commonly diagnosed cancers in the United States. Combined estrogen and progestogen therapy appears to reduce the risk of colorectal cancer. The change in risk, if any, is uncertain for estrogen therapy alone.3,4 Potential noncardiac risks of hormone therapy include ischemic stroke and VTE, although evidence suggests that the risk of these conditions may depend on the age at which hormone therapy is initiated. For example, combined estrogen and progestogen therapy and estrogen therapy alone increase the risk of ischemic stroke, particularly during the first one to two years after initiation of therapy.3,4 However, there does not appear to be an increased risk of stroke in women who begin hormone therapy between 50 and 59 years of age, although information is inconsistent.3 Estrogen therapy alone and combined estrogen and progestogen therapy increase the risk of VTE, particularly during the first one to two years of use, although the risk is less for women who are younger than 60 years or are taking estrogen alone.3,4,9
Deciding When to Start and End Hormone Therapy
Physicians should prescribe hormone therapy only after discussing the indications for treatment, the possible contraindications, and the relative risks and benefits of therapy and alternatives. Patients should have a thorough history and physical examination, and should be up to date on mammography and, if indicated, bone densitometry testing. Typically, symptomatic patients will start hormone therapy in perimenopause or after cessation of menses. Starting hormone therapy years after menopause is rarely necessary and increases the risk of cardiovascular disease.2,3 A reasonable rule of thumb is to use hormone therapy only to treat menopausal symptoms, using the lowest effective dosage for the shortest possible duration, with reevaluation on a regular basis.1,2
One of the most challenging decisions for patients taking hormone therapy is when to stop. One analysis found that more than 90 percent of patients using hormone therapy had stopped use by five years after menopause, so physicians can reassure patients that indefinite use is uncommon.10 The medical literature provides inadequate data at this time on which to base clinical decisions about the safety of long-term hormone therapy, although the potential increased risk of breast cancer after three to five years is concerning.3,8 A reasonable approach is to attempt discontinuation annually.3,10 A survey of patients discontinuing hormone therapy found that those who quit by tapering had reduced symptoms compared with those who quit “cold turkey,” although those tapering also had a higher incidence of restarting hormone therapy at a later date.11
Vasomotor Symptoms
Treating vasomotor symptoms is the most common indication for hormone therapy. Most postmenopausal women report hot flashes, although more than 85 percent have resolution within four to five years.12 Of the available treatment options for hormone therapy, estrogens are FDA-approved for treating vasomotor symptoms and are the most effective, reducing hot flashes by more than 80 percent.13,14 Patient choice should guide the route of therapy, because oral estrogen, transdermal patches, and transdermal creams are similarly effective.15 There are no data from randomized trials, but observational studies have shown that transdermal patches, which avoid the first-pass liver effect, may have a lower risk of VTE compared with oral estrogen.8,16
All medications used to treat vasomotor symptoms should be started at the lowest effective dosage and increased, if necessary, after eight weeks. Although lower dosages may take up to eight weeks to reach maximal effectiveness, starting with a lower dosage lessens adverse effects such as breast tenderness and uterine bleeding, and reduces the overall dosage that the patient receives.17 A study of 425 women with moderate to severe hot flashes found that a dosage as low as 14 mcg per day of 17-beta estradiol transdermally was effective, with or without 7.5 mcg of levonorgestrel per day.18 Because unopposed estrogen can cause endometrial hyperplasia and cancer in women with a uterus, such patients should use a combination of estrogen and progestogen for endometrial protection.8
Despite the effectiveness of estrogen, some patients may ask about alternative therapies, “bioidentical” hormone therapy, and compounded hormone formulations. Numerous studies have shown that the following alternative therapies are not consistently better than placebo for treating vasomotor symptoms: acupuncture, dong quai, Chinese herbal formulations, evening primrose oil, ginseng, red clover extract, kava, dietary soy, and black cohosh.13 Phytoestrogens (isoflavones) such as red clover and soy have received much media attention but do not appear to be effective for alleviation of menopausal symptoms.19
Online marketing of bioidentical hormones and compounded hormone formulations has increased recently. The term bioidentical refers to hormones such as estrone, 17-beta estradiol, and estriol that are identical to human hormones. Compounding refers to the mixture of various hormone preparations. One such formulation, “triest,” contains estriol, a placental hormone with unproven effectiveness or safety for treating menopausal symptoms. These unregulated formulations likely have adverse effects, such as endometrial hyperplasia, similar to those of pharmaceutical medications containing estrogen. The FDA and ACOG have published warnings about the limited safety and effectiveness data on these formulations.20,21 Physicians may want to refer to the FDA-approved medications listed in Table 1 as a guide for patients who desire bioidentical hormone therapy.

| Medication | Available formulations | |
|---|---|---|
| Estrogen | ||
| Oral estradiol | ||
| Estrace oral tablet | 0.5, 1.0, 2.0 mg | |
| Gynodiol oral tablet | 0.5, 1.0, 1.5, 2.0 mg | |
| Transdermal estradiol | ||
| Alora patch | 0.025, 0.05, 0.075, 0.1 mg per day | |
| Climara patch | 0.025, 0.0375, 0.05, 0.06, 0.075, 0.1 mg per day | |
| Divigel pouch | 0.25, 0.5, 1.0 mg | |
| Elestrin pump | 0.52 mg estradiol | |
| Estraderm patch | 0.05, 0.1 mg per day | |
| Estrasorb gel pouch | 0.025 mg per day | |
| Estrogel gel | 0.75 mg per pump | |
| Evamist spray | 1.5 mg | |
| Menostar patch | 0.014 mg per day | |
| Vivelle patch | 0.025, 0.0375, 0.05, 0.075, 0.1 mg per day | |
| Vaginal estradiol | ||
| Estrace vaginal cream | 0.1 mg estradiol per g | |
| Estring vaginal ring | 0.0075 mg estradiol per day over three months | |
| Femring vaginal ring | 0.05 mg estradiol per day over three months | |
| Vagifem vaginal tablet | 0.010 mg twice weekly | |
| Progestogen | ||
| Crinone vaginal progesterone gel* | 4% or 8%, every one to two days, depending on indication | |
| Prometrium micronized oral progesterone capsule | 100 or 200 mg per day | |
Nonestrogen therapies may be preferable for certain patients, including those who want to avoid the risks of hormone therapy. The selective serotonin reuptake inhibitors venlafaxine (Effexor), paroxetine (Paxil), and fluoxetine (Prozac) reduce hot flashes compared with placebo, whereas sertraline (Zoloft) and clonidine (Catapres) appear to be ineffective.13 A study of venlafaxine for treating hot flashes in breast cancer survivors found that a dosage of 75 mg per day reduced hot flashes by 61 percent compared with placebo.22 Gabapentin (Neurontin), 900 mg per day in a divided dose, decreased hot flashes by 45 versus 29 percent for placebo, but may have substantial adverse effects.23
Vulvovaginal Symptoms
Vaginal estrogen formulations are available as creams, tablets, or an estrogen-releasing ring. Local vaginal therapy is highly effective for treating atrophy, with success rates of 80 to 100 percent.13 The type of formulation used depends on patient preference, because all are effective. Physicians should reassure patients that vaginal estrogen does not cause endometrial proliferation and that adding a progestogen for endometrial protection is not necessary.8 There are two types of vaginal estradiol ring products: Estring delivers a topical dosage of estradiol for the treatment of menopausal atrophy, whereas Femring delivers a systemic dosage of estrogen.
Potential adverse effects of vaginal estrogen alone include burning after application, candidal infections, breast or perineal pain, and vaginal bleeding. Creams, tablets, and the estradiol vaginal ring are equally effective for treating symptoms, but the cream appears to have more adverse effects (such as uterine bleeding, and breast and perineal pain) than the tablets. Further, conjugated equine estrogen cream may lead to more endometrial hyperstimulation than the estrogen ring.24 Vaginal administration of estradiol may cause an increase in serum estradiol levels in some patients, but data are lacking about the long-term risks of breast cancer, VTE, or endometrial proliferation for patients who use low-dose vaginal estrogen therapy. The decision to use vaginal estrogen formulations for women with a history of hormone-dependent cancers should be based on the woman's preference after consultation with her oncologist.
There are few alternatives to estrogen therapy for menopausal vulvovaginal symptoms. Oral phytoestrogens are not effective for vaginal symptoms,25 but evidence suggests that an over-the-counter vaginal moisturizer (Replens) used three times weekly may be as effective as vaginal estrogen for symptom relief.26 This is a reasonable starting point for patients with vulvovaginal symptoms, particularly those who wish to avoid hormone medications.
Osteoporosis
There are a number of options for the prevention or treatment of postmenopausal osteoporosis. Because of concerns about the safety of hormone therapy, attention has focused on selective estrogen receptor modulators as an alternative. Raloxifene (Evista) is FDA-approved for the treatment and prevention of postmenopausal osteoporosis, whereas many estrogen-only and combination estrogen and progestogen products are approved for the prevention of osteoporosis (but not treatment) in postmenopausal women.8 Large randomized trials have revealed that selective estrogen receptor modulators have several interesting properties, including a significant reduction in breast cancer risk.27,28 Raloxifene also increases the risk of VTE, fatal stroke, and hot flashes.28,29 Unlike unopposed estrogen therapy, raloxifene does not appear to increase the risk of endometrial cancer.29 Table 2 reviews the risks and benefits of estrogen and progestogen compared with raloxifene.3,4,28 The decision to use estrogen therapy or raloxifene is difficult because both medications have benefits and risks. Raloxifene is a reasonable option for postmenopausal women without hot flashes or known risk factors for VTE, or those who have risk factors for breast cancer.
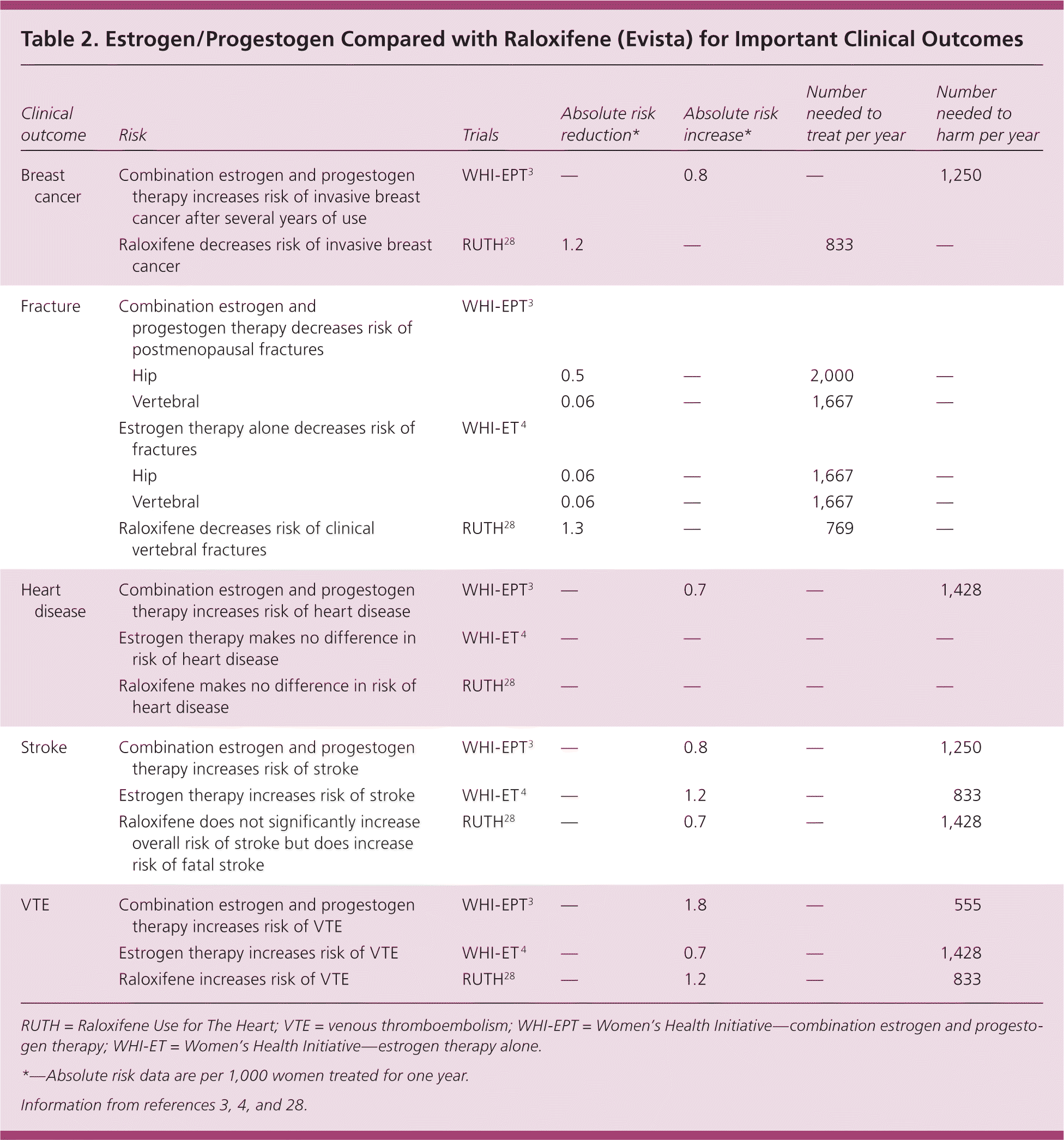
| Clinical outcome | Risk | Trials | Absolute risk reduction* | Absolute risk increase* | Number needed to treat per year | Number needed to harm per year | |
|---|---|---|---|---|---|---|---|
| Breast cancer | Combination estrogen and progestogen therapy increases risk of invasive breast cancer after several years of use | WHI-EPT3 | — | 0.8 | — | 1,250 | |
| Raloxifene decreases risk of invasive breast cancer | RUTH28 | 1.2 | — | 833 | — | ||
| Fracture | Combination estrogen and progestogen therapy decreases risk of postmenopausal fractures | WHI-EPT3 | |||||
| Hip | 0.5 | — | 2,000 | — | |||
| Vertebral | 0.06 | — | 1,667 | — | |||
| Estrogen therapy alone decreases risk of fractures | WHI-ET 4 | ||||||
| Hip | 0.06 | — | 1,667 | — | |||
| Vertebral | 0.06 | — | 1,667 | — | |||
| Raloxifene decreases risk of clinical vertebral fractures | RUTH28 | 1.3 | — | 769 | — | ||
| Heart disease | Combination estrogen and progestogen therapy increases risk of heart disease | WHI-EPT3 | — | 0.7 | — | 1,428 | |
| Estrogen therapy makes no difference in risk of heart disease | WHI-ET 4 | — | — | — | — | ||
| Raloxifene makes no difference in risk of heart disease | RUTH28 | — | — | — | — | ||
| Stroke | Combination estrogen and progestogen therapy increases risk of stroke | WHI-EPT3 | — | 0.8 | — | 1,250 | |
| Estrogen therapy increases risk of stroke | WHI-ET 4 | — | 1.2 | — | 833 | ||
| Raloxifene does not significantly increase overall risk of stroke but does increase risk of fatal stroke | RUTH28 | — | 0.7 | — | 1,428 | ||
| VTE | Combination estrogen and progestogen therapy increases risk of VTE | WHI-EPT3 | — | 1.8 | — | 555 | |
| Estrogen therapy increases risk of VTE | WHI-ET 4 | — | 0.7 | — | 1,428 | ||
| Raloxifene increases risk of VTE | RUTH28 | — | 1.2 | — | 833 | ||