Chronic diarrhea, defined as a decrease in stool consistency for more than four weeks, is a common but challenging clinical scenario. It can be divided into three basic categories: watery, fatty (malabsorption), and inflammatory. Watery diarrhea may be subdivided into osmotic, secretory, and functional types. Watery diarrhea includes irritable bowel syndrome, which is the most common cause of functional diarrhea. Another example of watery diarrhea is microscopic colitis, which is a secretory diarrhea affecting older persons. Laxative-induced diarrhea is often osmotic. Malabsorptive diarrhea is characterized by excess gas, steatorrhea, or weight loss; giardiasis is a classic infectious example. Celiac disease (gluten-sensitive enteropathy) is also malabsorptive, and typically results in weight loss and iron deficiency anemia. Inflammatory diarrhea, such as ulcerative colitis or Crohn disease, is characterized by blood and pus in the stool and an elevated fecal calprotectin level. Invasive bacteria and parasites also produce inflammation. Infections caused by Clostridium difficile subsequent to antibiotic use have become increasingly common and virulent. Not all chronic diarrhea is strictly watery, malabsorptive, or inflammatory, because some categories overlap. Still, the most practical diagnostic approach is to attempt to categorize the diarrhea by type before testing and treating. This narrows the list of diagnostic possibilities and reduces unnecessary testing. Empiric therapy is justified when a specific diagnosis is strongly suspected and follow-up is available.
Chronic diarrhea is defined as a decrease in stool consistency continuing for more than four weeks.1 Although reasonably common (3 to 5 percent of the population), it represents a considerable diagnostic challenge, with several hundred conditions in the differential diagnosis.1 This article focuses on a diagnostic approach that is practical for the primary care physician.
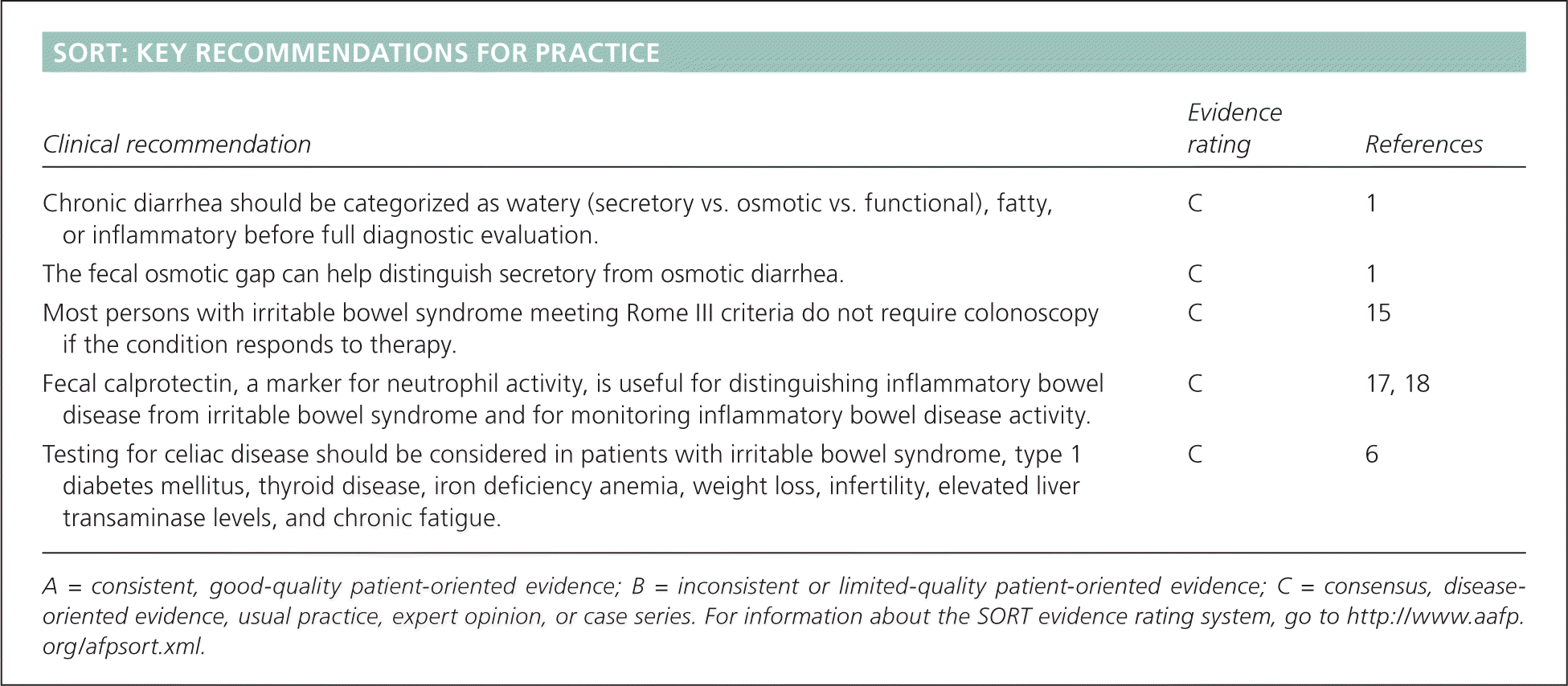
SORT: KEY RECOMMENDATIONS FOR PRACTICE
| Clinical recommendation | Evidence rating | References |
|---|---|---|
| Chronic diarrhea should be categorized as watery (secretory vs. osmotic vs. functional), fatty, or inflammatory before full diagnostic evaluation. | C | 1 |
| The fecal osmotic gap can help distinguish secretory from osmotic diarrhea. | C | 1 |
| Most persons with irritable bowel syndrome meeting Rome III criteria do not require colonoscopy if the condition responds to therapy. | C | 15 |
| Fecal calprotectin, a marker for neutrophil activity, is useful for distinguishing inflammatory bowel disease from irritable bowel syndrome and for monitoring inflammatory bowel disease activity. | C | 17, 18 |
| Testing for celiac disease should be considered in patients with irritable bowel syndrome, type 1 diabetes mellitus, thyroid disease, iron deficiency anemia, weight loss, infertility, elevated liver transaminase levels, and chronic fatigue. | C | 6 |
A = consistent, good-quality patient-oriented evidence; B = inconsistent or limited-quality patient-oriented evidence; C = consensus, disease-oriented evidence, usual practice, expert opinion, or case series. For information about the SORT evidence rating system, go to https://www.aafp.org/afpsort.xml.
Categorization
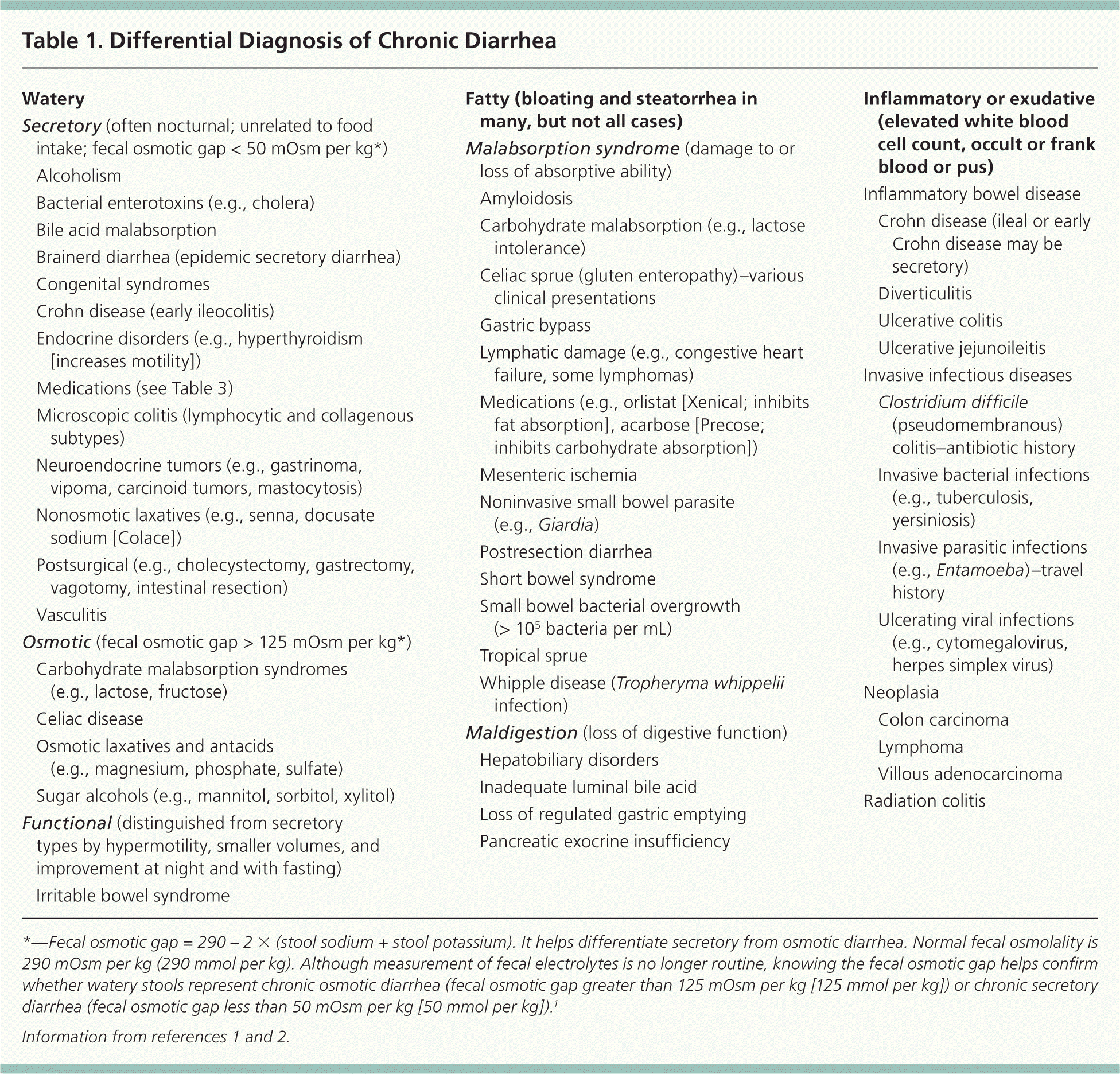
Chronic diarrhea may be divided into three basic categories: watery, fatty (malabsorption), and inflammatory (with blood and pus). However, not all chronic diarrhea is strictly watery, malabsorptive, or inflammatory, because some categories overlap. The differential diagnosis of chronic diarrhea is described in Table 1.1,2
Table 1. Differential Diagnosis of Chronic Diarrhea
| Watery | |
| Secretory (often nocturnal; unrelated to food intake; fecal osmotic gap < 50 mOsm per kg*) | |
| Alcoholism | |
| Bacterial enterotoxins (e.g., cholera) | |
| Bile acid malabsorption | |
| Brainerd diarrhea (epidemic secretory diarrhea) | |
| Congenital syndromes | |
| Crohn disease (early ileocolitis) | |
| Endocrine disorders (e.g., hyperthyroidism [increases motility]) | |
| Medications (see Table 3) | |
| Microscopic colitis (lymphocytic and collagenous subtypes) | |
| Neuroendocrine tumors (e.g., gastrinoma, vipoma, carcinoid tumors, mastocytosis) | |
| Nonosmotic laxatives (e.g., senna, docusate sodium [Colace]) | |
| Postsurgical (e.g., cholecystectomy, gastrectomy, vagotomy, intestinal resection) | |
| Vasculitis | |
| Osmotic (fecal osmotic gap > 125 mOsm per kg*) | |
| Carbohydrate malabsorption syndromes (e.g., lactose, fructose) | |
| Celiac disease | |
| Osmotic laxatives and antacids (e.g., magnesium, phosphate, sulfate) | |
| Sugar alcohols (e.g., mannitol, sorbitol, xylitol) | |
| Functional (distinguished from secretory types by hypermotility, smaller volumes, and improvement at night and with fasting) | |
| Irritable bowel syndrome | |
| Fatty (bloating and steatorrhea in many, but not all cases) | |
| Malabsorption syndrome (damage to or loss of absorptive ability) | |
| Amyloidosis | |
| Carbohydrate malabsorption (e.g., lactose intolerance) | |
| Celiac sprue (gluten enteropathy)–various clinical presentations | |
| Gastric bypass | |
| Lymphatic damage (e.g., congestive heart failure, some lymphomas) | |
| Medications (e.g., orlistat [Xenical; inhibits fat absorption], acarbose [Precose; inhibits carbohydrate absorption]) | |
| Mesenteric ischemia | |
| Noninvasive small bowel parasite (e.g., Giardia) | |
| Postresection diarrhea | |
| Short bowel syndrome | |
| Small bowel bacterial overgrowth (> 105 bacteria per mL) | |
| Tropical sprue | |
| Whipple disease (Tropheryma whippelii infection) | |
| Maldigestion (loss of digestive function) | |
| Hepatobiliary disorders | |
| Inadequate luminal bile acid | |
| Loss of regulated gastric emptying | |
| Pancreatic exocrine insufficiency | |
| Inflammatory or exudative (elevated white blood cell count, occult or frank blood or pus) | |
| Inflammatory bowel disease Crohn disease (ileal or early Crohn disease may be secretory) | |
| Diverticulitis | |
| Ulcerative colitis | |
| Ulcerative jejunoileitis | |
| Invasive infectious diseases | |
| Clostridium difficile (pseudomembranous) colitis–antibiotic history | |
| Invasive bacterial infections (e.g., tuberculosis, yersiniosis) | |
| Invasive parasitic infections (e.g., Entamoeba)–travel history | |
| Ulcerating viral infections (e.g., cytomegalovirus, herpes simplex virus) | |
| Neoplasia | |
| Colon carcinoma | |
| Lymphoma | |
| Villous adenocarcinoma | |
| Radiation colitis | |
*—Fecal osmotic gap = 290 – 2 × (stool sodium + stool potassium). It helps differentiate secretory from osmotic diarrhea. Normal fecal osmolality is 290 mOsm per kg (290 mmol per kg). Although measurement of fecal electrolytes is no longer routine, knowing the fecal osmotic gap helps confirm whether watery stools represent chronic osmotic diarrhea (fecal osmotic gap greater than 125 mOsm per kg [125 mmol per kg]) or chronic secretory diarrhea (fecal osmotic gap less than 50 mOsm per kg [50 mmol per kg]).1
Watery diarrhea may be subdivided into osmotic (water retention due to poorly absorbed substances), secretory (reduced water absorption), and functional (hypermotility) types.1 Osmotic laxatives, such as sorbitol, induce osmotic diarrhea. Secretory diarrhea can be distinguished from osmotic and functional diarrhea by virtue of higher stool volumes (greater than 1 L per day) that continue despite fasting and occur at night. Stimulant laxatives fall into this secretory category because they increase motility.3 Persons with functional disorders have smaller stool volumes (less than 350 mL per day) and no diarrhea at night.3
Medical History
A history is the critical first step in diagnosis. It is important to understand exactly what patients mean when they say they have diarrhea. A patient may not actually have diarrhea, but incontinence occasioned by fecal impaction.4 Stool volume, frequency, and consistency can help categorize the diarrhea, as previously mentioned. A travel history is essential. Travel to the tropics vastly expands the list of diagnostic possibilities, but in no way rules out common causes. Bloody diarrhea after a trip to Africa may still be ulcerative colitis rather than amebic dysentery.
Physical Examination
Physical examination provides additional clues to the cause of diarrhea. Recent weight loss or lymphadenopathy could result from chronic infection or malignancy. Eye findings, such as episcleritis or exophthalmia, suggest that the diarrhea is attributable to inflammatory bowel disease (IBD) and hyperthyroidism, respectively. Dermatitis herpetiformis, an itchy blistering rash, is found in 15 to 25 percent of patients with celiac disease.5 An abdominal examination for scars (surgical causes of diarrhea), bowel sounds (hypermotility), tenderness (infection and inflammation), and masses (neoplasia) should be followed by rectal examination, including fecal occult blood testing. Anal fistulae suggest Crohn disease. A quick office anoscopy may detect ulcerations or impacted stool. Such impactions are a common cause of pseudodiarrhea or paradoxical diarrhea, which is actually seepage around impacted stools.
Laboratory Testing
Basic blood work may include a complete blood count, albumin level, erythrocyte sedimentation rate, liver function testing, thyroid-stimulating hormone level, and electrolyte levels. Iron deficiency anemia may be an indicator of celiac disease and warrants screening.6 A minimal stool assessment should include a fecal leukocyte level and fecal occult blood test, with further testing as indicated. Fecal calprotectin, a neutrophil activity marker, is extremely useful in identifying IBD. Clostridium difficile stool toxin should be obtained for diarrhea after hospitalization or antibiotic use. A stool laxative screen (e.g., sodium, potassium, magnesium, phosphate, sulfate, phenolphthalein, bisacodyl [Dulcolax]) should be ordered if abuse is suspected.7 More specialized stool tests, including fecal fat analysis or a Sudan stain for fat, can be used to evaluate malabsorptive disorders.
A fecal pH test is quick and can be performed in the office if the patient is not taking antibiotics. At least 0.5 mL of fecal matter is collected, and a strip of nitrazine paper is dipped in the sample and compared with a color scale. A pH of less than 5.5 indicates an acidic sample and may suggest lactose intolerance. Fecal electrolyte levels can be used to distinguish secretory from osmotic diarrhea. Although fecal pH tests and fecal electrolyte levels are helpful, they are often omitted from the initial workup. Abnormal laboratory results help distinguish organic from functional disease.8
Travel risk factors might warrant a stool culture and sensitivity test, stool ova and parasite examination, and Giardia and Cryptosporidium stool antigen tests. Giardia and Cryptosporidium infections are easily missed on routine ova and parasite examination, although stool acid-fast staining identifies Cryptosporidium. Finally, sigmoidoscopy or colonoscopy is often required to establish a specific diagnosis. Microscopic colitis can be diagnosed only by colon biopsy.9 When it is necessary to distinguish between secretory and other categories of diarrhea, 24-hour stool collection can quantify stool production.
Diagnostic Approach
It is usually impractical to test and treat the many possible causes of chronic diarrhea. In most cases, it is more reasonable to categorize by type of diarrhea before testing and treating to narrow the list of diagnostic possibilities and reduce unnecessary testing.10 First, physicians should determine if the diarrhea can be categorized as watery, fatty, or inflammatory, and then pick specific tests to sort out the much smaller differential diagnosis within each category (Figure 11,11 ).
Figure 1. Diagnosis of Chronic Diarrhea
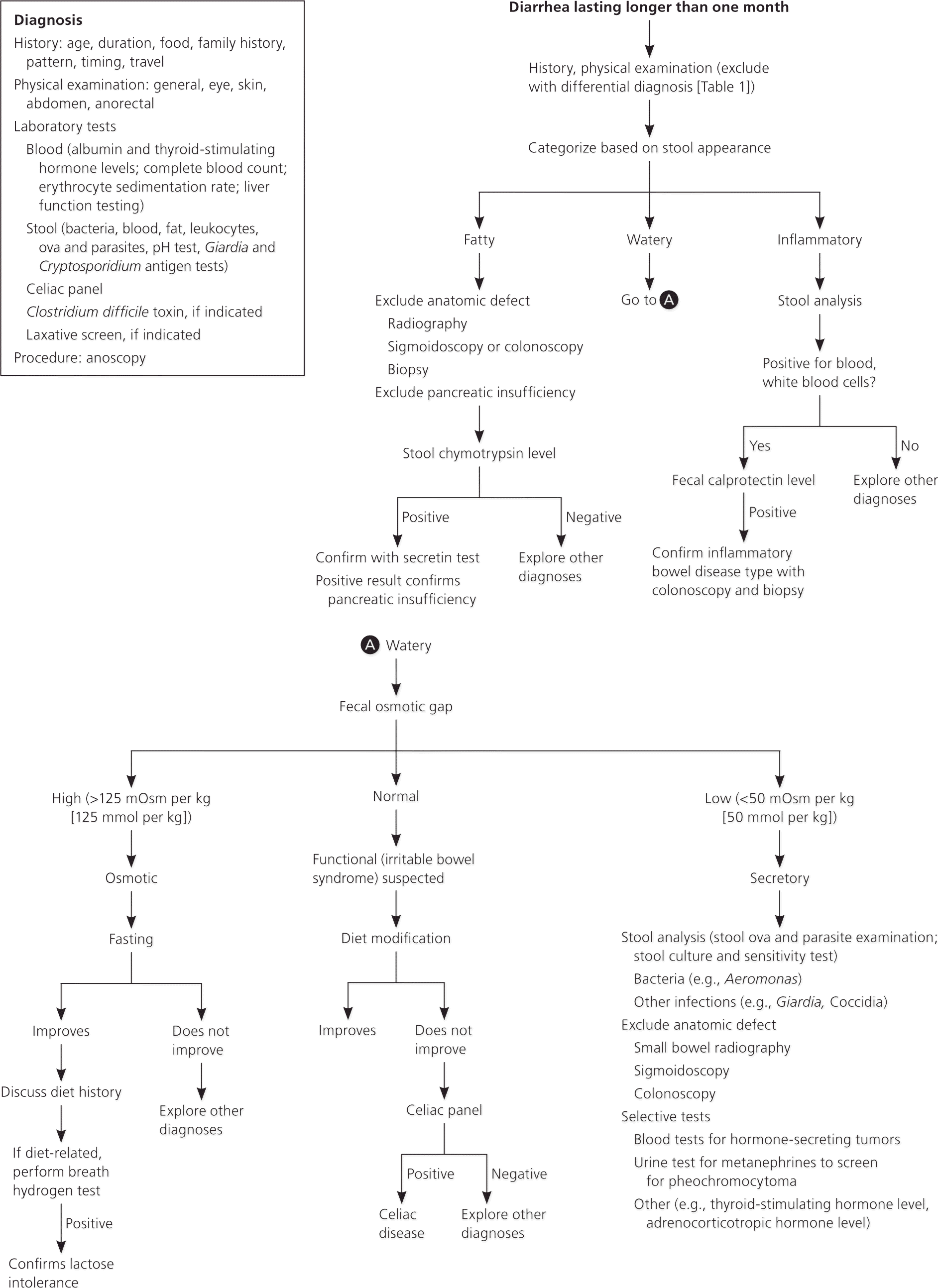
Algorithm for the diagnosis of chronic diarrhea.
Information from references 1 and 11.
Empiric therapy may be justified if a specific diagnosis is strongly suspected or resources are limited. Life-threatening conditions should be excluded. A trial of metronidazole (Flagyl) for malabsorption in a traveler would cure possible giardiasis. Likewise, an empiric trial of bile acid resins can help confirm bile acid malabsorption.1 Follow-up should be performed in patients on empiric therapy because the patient may not improve, or may initially improve only to relapse.
Major Causes
IRRITABLE BOWEL SYNDROME (FUNCTIONAL)
Irritable bowel syndrome (IBS) is the most common cause of functional diarrhea in the developed world. IBS is a symptom complex of crampy abdominal pain accompanied by altered bowel habits, either with diarrhea or constipation. Usually watery diarrhea occurs while awake, often following meals. Discomfort is alleviated by defecation, and stool mucus is noted in one-half of patients.12 Women are diagnosed twice as often as men. “Alarm” symptoms such as nocturnal diarrhea, progressive pain, weight loss, or blood in the stool suggest another diagnosis.
IBS is exacerbated by emotional stress or eating, but it may also be a response to recent infection. IBS symptoms subsequent to a bout of traveler's diarrhea (postinfectious IBS) may take months to resolve, despite a negative workup.13 Two sets of useful diagnostic criteria exist for IBS: Manning and Rome III (Table 2).12,14 Although IBS is usually considered a diagnosis of exclusion, it is unnecessary to perform an exhaustive evaluation in healthy young patients meeting Rome III criteria if they respond to fiber, exercise, and dietary modification. Screening for celiac disease and iron deficiency anemia is recommended, but routine colonoscopy is not cost-effective unless alarm symptoms are present.15 Screening for celiac disease is justifiable because patients with IBS may have four times the incidence of celiac disease compared with the general population.16 All laboratory test results are normal in patients with IBS.
Table 2. Diagnostic Criteria for Irritable Bowel Syndrome

| Manning criteria | |
| Onset of pain linked to more frequent bowel movements | |
| Looser stools associated with onset of pain | |
| Pain relieved by passage of stool | |
| Noticeable abdominal bloating | |
| Sensation of incomplete evacuation more than 25 percent of the time | |
| Diarrhea with mucus more than 25 percent of the time | |
| Rome III criteria | |
| Symptoms of recurrent abdominal pain or discomfort and a marked change in bowel habits for at least six months, with symptoms experienced on at least three days per month for at least three months. Two or more of the following must apply: | |
| Pain is relieved by a bowel movement | |
| Onset of pain is related to a change in frequency of stool | |
| Onset of pain is related to a change in appearance of stool | |
INFLAMMATORY BOWEL DISEASE (INFLAMMATORY)
IBD may manifest as ulcerative colitis or Crohn disease. Both often involve blood and pus in the stool, with onset between 15 and 40 years of age. Early cases are often confused with IBS, but symptoms inevitably progress.
Early ulcerative colitis, distal colitis, and proctitis result in episodes of rectal bleeding, diarrhea, pain, and tenesmus. Moderate (left-sided) and severe (extensive) colitis are associated with bloody diarrhea, weight loss, fever, and anemia.
Crohn disease typically causes an ileitis but later affects the entire gastrointestinal tract to become exudative. Early symptoms can be subtle, leading to a long delay in diagnosis. Abdominal pain, diarrhea, fever, perianal fistulae, and stools positive for blood are common, although bloody diarrhea is unusual.
The diagnosis of IBD is usually supported by colonoscopy. Laboratory testing should include a complete blood count, fecal leukocyte level, erythrocyte sedimentation rate, and fecal calprotectin level.17 An elevated fecal calprotectin level is emerging as a reliable way to establish the diagnosis of IBD as distinguished from IBS, and to monitor its activity once diagnosed.17 Fecal calprotectin level is a newer stool screen that reflects fecal leukocyte activity; the stool sample can be collected by the patient at home because calprotectin is stable in stool samples for up to seven days at room temperature. In adults, fecal calprotectin level is 93 percent sensitive and 96 percent specific for IBD, although specificity is less (76 percent) in children and teenagers.18 Routine screening with fecal calprotectin level could result in a 67 percent reduction in the use of colonoscopies for IBD diagnosis in adults, because only those with a positive result would need them (Figure 2).18
Figure 2. Workup for Suspected IBD
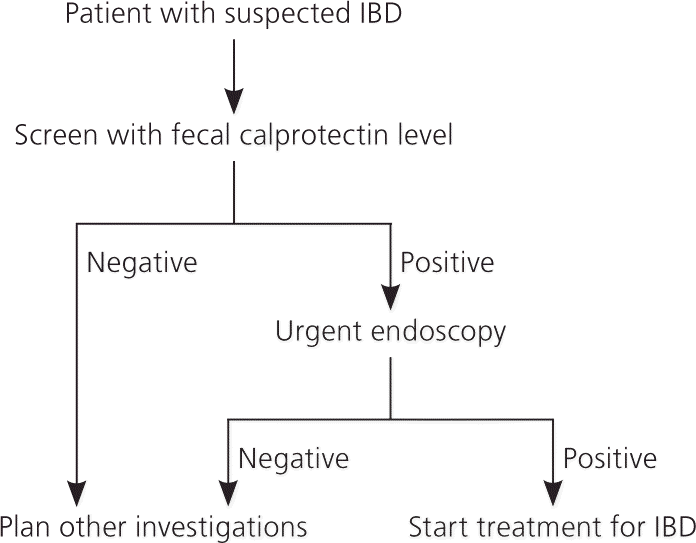
Workup for suspected inflammatory bowel disease (IBD) in adults.
Adapted with permission from van Rheenen PF, Van de Vijver E, Fidler V. Faecal calprotectin for screening of patients with suspected inflammatory bowel disease: diagnostic meta-analysis. BMJ . 2010;341:c3369.
MICROSCOPIC COLITIS (SECRETORY)
Microscopic colitis is characterized by intermittent, secretory diarrhea in older patients, although persons of all ages can be affected.19 It should be suspected in older patients with persistent, often nocturnal diarrhea that does not respond to fasting. It is thought to be fairly common, comprising 10 percent of chronic diarrhea cases.20 The cause is unknown, but there is some evidence that more than six months of nonsteroidal anti-inflammatory drug use increases the risk.21 Despite microscopic inflammation, there are no systemic symptoms, and blood and white blood cells are absent from the stool. Colonoscopy is normal, but biopsy from the transverse colon confirms the diagnosis. Two histologic patterns are found: lymphocytic colitis (lymphocytic infiltrate of the lamina propria) and collagenous colitis (in which the subepithelial collagen layer is also increased to more than 10 mm).19
MALABSORPTIVE DIARRHEA (FATTY)
Malabsorptive and maldigestive diarrhea result from impaired nutrient absorption and impaired digestive function, respectively. Celiac disease (gluten-sensitive enteropathy), intestinal bypass, mesenteric ischemia, small bowel bacterial overgrowth, Whipple disease, and giardiasis can all cause loss of absorptive capacity. Absent pancreatic enzymes or bile acids can cause maldigestion. Classic symptoms include abdominal distention with foul-smelling, large, floating, pale, fatty stools (steatorrhea) and weight loss.
Celiac disease or sprue is small intestine malabsorption provoked by gluten (wheat) ingestion in genetically susceptible patients, often of European descent. It is suspected that diagnosed celiac disease cases represent only a fraction of patients with this condition.22 Although predominantly a malabsorptive condition, celiac disease can also initially present as a watery diarrhea that may be confusted with IBS. Patients most commonly present with chronic diarrhea, fatigue, iron deficiency anemia, and weight loss, but this classic constellation is often absent.22 In the United States, more than 2 million persons have celiac disease, or about one in 133 persons, and this increases to as many as one in 22 persons if a first-degree relative is affected.23 A high index of suspicion is essential to diagnose celiac disease in most adults. A positive celiac panel, typically consisting of immunoglobulin A (IgA) antigliadin, antiendomysium, and antitissue transglutaminase antibodies, is usually followed by duodenal biopsy for confirmation.24 IgA antiendomysium and antitissue transglutaminase antibodies have much better accuracy than native gliadin antibodies (IgA antigliadin antibodies).22 The deamidated gliadin peptide antibody test is a more accurate second-generation IgA antigliadin antibody test but is not yet routinely included in panels.25 It should be noted that use of gluten-free diets before testing can give false-negative results, as can IgA deficiency or age younger than two years.22
Testing should be expanded to include all patients with unexplained chronic diarrhea, IBS, iron deficiency anemia, chronic fatigue, weight loss, infertility, and elevated liver transaminase levels. Symptomatic patients with type 1 diabetes mellitus and thyroid disease should be tested, because these conditions may predispose to celiac disease.6 Asymptomatic patients with a family history of celiac disease generally should not be tested, although physicians should remain open to discussing testing.6
CHRONIC INFECTIONS (PRIMARILY INFLAMMATORY)
Most microbial gastrointestinal infections cause acute self-limited diarrhea, but others persist, resulting in inflammation (invasive bacteria and parasites) or occasionally malabsorption (giardiasis). A history of travel and antibiotic use is important. Bacterial causes include Aeromonas, Campylobacter, C. difficile, Plesiomonas, and Yersinia. Parasitic diarrhea may be caused by Cryptosporidium, Cyclospora, Entamoeba, Giardia, Microsporida, and Strongyloides. Giardia, the most common of these, is best diagnosed with fecal antigen testing.1 Brainerd diarrhea is an epidemic form of long-lasting secretory diarrhea associated with consumption of unpasteurized milk or contaminated water. The suspected infectious agent has never been identified.26
C. difficile, the cause of pseudomembranous colitis, has emerged as a major infection in U.S. hospitals. This gram-positive anaerobic bacillus is easily spread through ingestion of spores, rapidly colonizing the colon following antibiotic therapy. A history of using fluoroquinolones, clindamycin, penicillins, or cephalosporins in the past three months is often associated.27 Approximately 3 percent of healthy adults may be asymptomatic carriers, but this increases to at least 40 percent in hospitalized patients.28
Proton pump inhibitors and IBD further increase the risk of C. difficile infection.29 Older, sicker patients are more likely to develop clinical disease. Because IBD and C. difficile infection produce similar symptoms, it is necessary to exclude the latter in ill patients.27
A new hypervirulent C. difficile strain (NAP1/BI/027) produces a binary toxin in addition to the usual A and B toxins.27 Outbreaks are characterized by severe watery diarrhea, often complicated by toxic megacolon. C. difficile infection is best diagnosed by diarrheic stool cytotoxin assay. Colonoscopy is unnecessary with a positive stool toxin.
DRUG-INDUCED (VARIOUS)
Although C. difficile infection is induced by antibiotics, drugs can directly cause diarrhea. Laxatives, antacids, proton pump inhibitors, and antineoplastic agents are medications that can lead to diarrhea; other examples of drugs associated with diarrhea are listed in Table 3.2 Laxative or antacid abuse is a common cause. Diarrhea may be osmotic (from magnesium, phosphates, sulfates, or sorbitol) or secretory following use of stimulant laxatives. Symptoms resolve when the offending agent is eliminated.
Table 3. Drugs Associated with Diarrhea
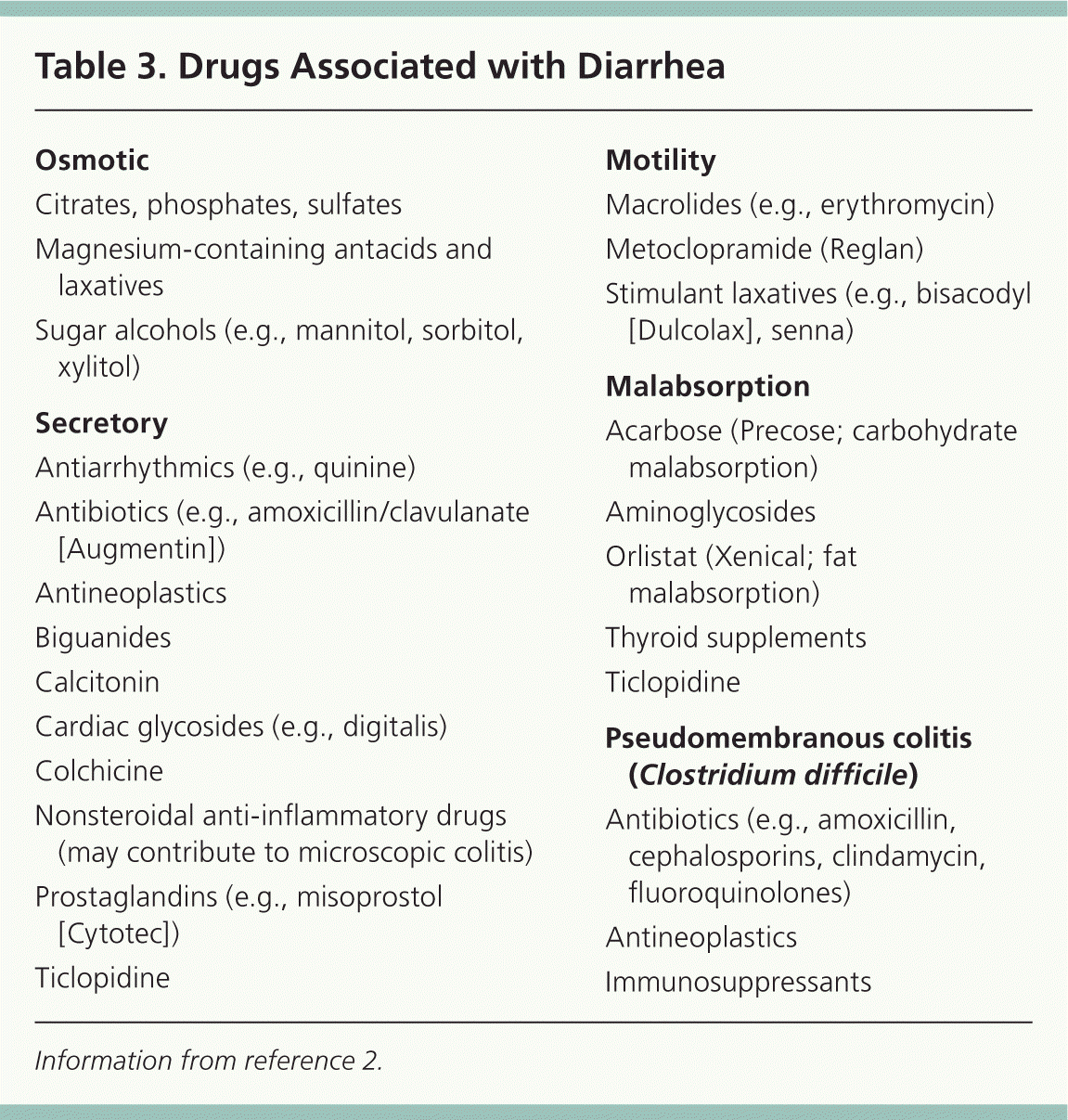
| Osmotic |
| Citrates, phosphates, sulfates |
| Magnesium-containing antacids and laxatives |
| Sugar alcohols (e.g., mannitol, sorbitol, xylitol) |
| Secretory |
| Antiarrhythmics (e.g., quinine) |
| Antibiotics (e.g., amoxicillin/clavulanate [Augmentin]) |
| Antineoplastics |
| Biguanides |
| Calcitonin |
| Cardiac glycosides (e.g., digitalis) |
| Colchicine |
| Nonsteroidal anti-inflammatory drugs (may contribute to microscopic colitis) |
| Prostaglandins (e.g., misoprostol [Cytotec]) |
| Ticlopidine |
| Motility |
| Macrolides (e.g., erythromycin) |
| Metoclopramide (Reglan) |
| Stimulant laxatives (e.g., bisacodyl [Dulcolax], senna) |
| Malabsorption |
| Acarbose (Precose; carbohydrate malabsorption) |
| Aminoglycosides |
| Orlistat (Xenical; fat malabsorption) |
| Thyroid supplements |
| Ticlopidine |
| Pseudomembranous colitis (Clostridium difficile) |
| Antibiotics (e.g., amoxicillin, cephalosporins, clindamycin, fluoroquinolones) |
| Antineoplastics |
| Immunosuppressants |
Information from reference 2.
ENDOCRINE AND OTHER TYPES OF DIARRHEA (SECRETORY/HYPERMOTILITY)
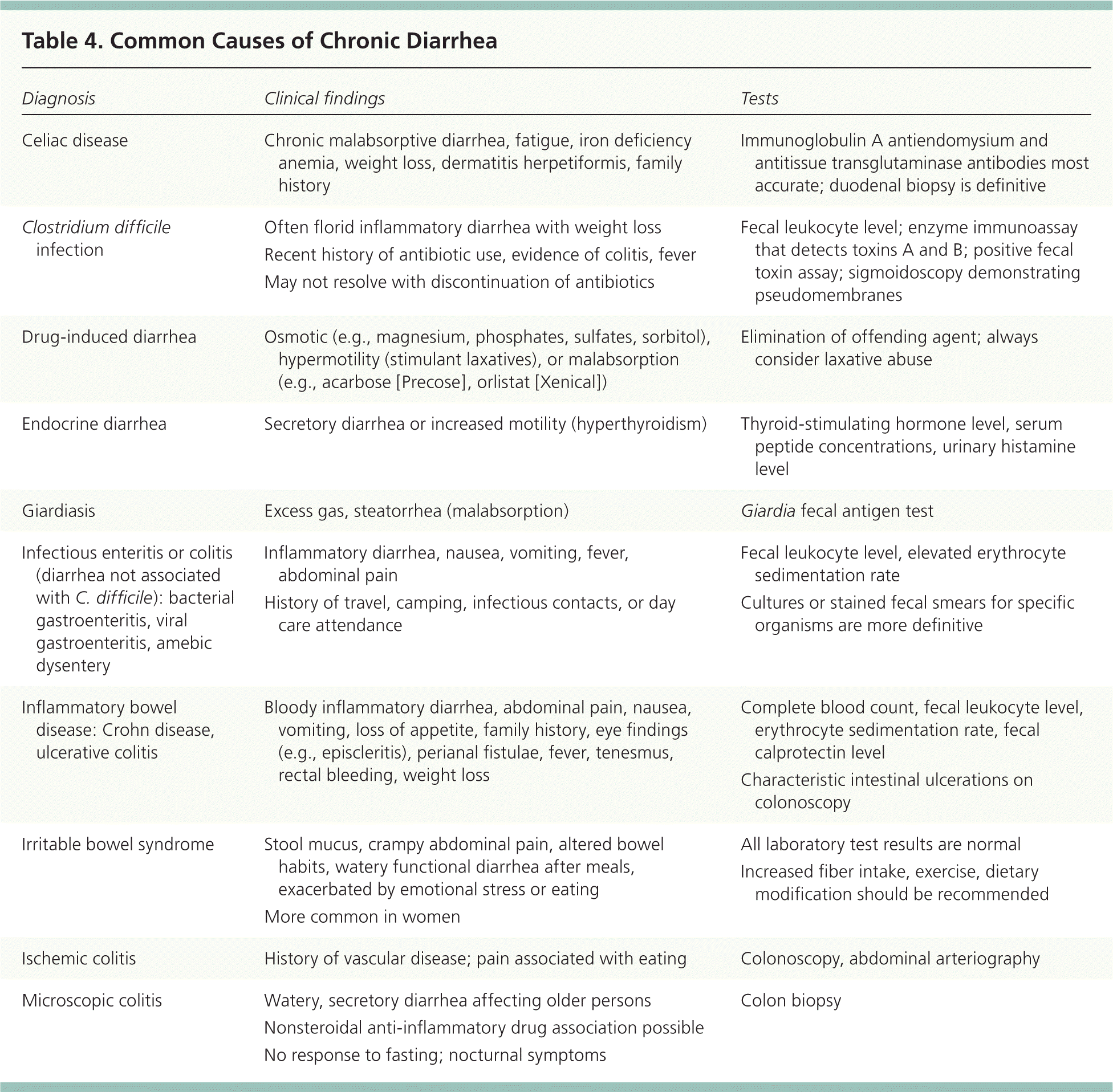
Endocrine causes of chronic secretory diarrhea include Addison disease, carcinoid tumors, vipoma, gastrinoma (Zollinger-Ellison syndrome), and mastocytosis. Hyperthyroidism increases motility. Serum peptide concentrations (e.g., gastrin, calcitonin, vasoactive intestinal peptide) and urinary histamine level should be obtained only when these conditions are suspected.1 Neoplastic diarrhea is associated with colon carcinoma, villous adenocarcinoma, and lymphoma. Common causes of chronic diarrhea are listed in Table 4.
Table 4. Common Causes of Chronic Diarrhea
| Diagnosis | Clinical findings | Tests |
|---|---|---|
| Celiac disease | Chronic malabsorptive diarrhea, fatigue, iron deficiency anemia, weight loss, dermatitis herpetiformis, family history |
|
| Clostridium difficile infection | Often florid inflammatory diarrhea with weight loss |
|
| Recent history of antibiotic use, evidence of colitis, fever | ||
| May not resolve with discontinuation of antibiotics | ||
| Drug-induced diarrhea | Osmotic (e.g., magnesium, phosphates, sulfates, sorbitol), hypermotility (stimulant laxatives), or malabsorption (e.g., acarbose [Precose], orlistat [Xenical]) |
|
| Endocrine diarrhea | Secretory diarrhea or increased motility (hyperthyroidism) |
|
| Giardiasis | Excess gas, steatorrhea (malabsorption) |
|
| Infectious enteritis or colitis (diarrhea not associated with C. difficile): bacterial gastroenteritis, viral gastroenteritis, amebic dysentery | Inflammatory diarrhea, nausea, vomiting, fever, abdominal pain |
|
| History of travel, camping, infectious contacts, or day care attendance | ||
| Inflammatory bowel disease: Crohn disease, ulcerative colitis | Bloody inflammatory diarrhea, abdominal pain, nausea, vomiting, loss of appetite, family history, eye findings (e.g., episcleritis), perianal fistulae, fever, tenesmus, rectal bleeding, weight loss |
|
| Irritable bowel syndrome | Stool mucus, crampy abdominal pain, altered bowel habits, watery functional diarrhea after meals, exacerbated by emotional stress or eating |
|
| More common in women | ||
| Ischemic colitis | History of vascular disease; pain associated with eating |
|
| Microscopic colitis | Watery, secretory diarrhea affecting older persons |
|
| Nonsteroidal anti-inflammatory drug association possible | ||
| No response to fasting; nocturnal symptoms |

