
Am Fam Physician. 2012;86(2):161-168
A more recent article on basal cell and cutaneous squamous cell carcinomas is available.
Patient information: See related handout on nonmelanoma skin cancer, written by the author of this article.
Author disclosure: No relevant financial affiliations to disclose.
Family physicians are regularly faced with identifying, treating, and counseling patients with skin cancers. Nonmelanoma skin cancer, which encompasses basal cell and squamous cell carcinoma, is the most common cancer in the United States. Ultraviolet B exposure is a significant factor in the development of basal cell and squamous cell carcinoma. The use of tanning beds is associated with a 1.5-fold increase in the risk of basal cell carcinoma and a 2.5-fold increase in the risk of squamous cell carcinoma. Routine screening for skin cancer is controversial. The U.S. Preventive Services Task Force cites insufficient evidence to recommend for or against routine whole-body skin examination to screen for skin cancer. Basal cell carcinoma most commonly appears as a pearly white, dome-shaped papule with prominent telangiectatic surface vessels. Squamous cell carcinoma most commonly appears as a firm, smooth, or hyperkeratotic papule or plaque, often with central ulceration. Initial tissue sampling for diagnosis involves a shave technique if the lesion is raised, or a 2- to 4-mm punch biopsy of the most abnormal-appearing area of skin. Mohs micrographic surgery has the lowest recurrence rate among treatments, but is best considered for large, high-risk tumors. Smaller, lower-risk tumors may be treated with surgical excision, electrodesiccation and curettage, or cryotherapy. Topical imiquimod and fluorouracil are also potential, but less supported, treatments. Although there are no clear guidelines for follow-up after an index nonmelanoma skin cancer, monitoring for recurrence is prudent because the risk of subsequent skin cancer is 35 percent at three years and 50 percent at five years.
Nonmelanoma skin cancer, which includes basal cell carcinoma and squamous cell carcinoma, is the most common cancer in the United States. Approximately 80 percent of nonmelanoma skin cancers are basal cell carcinoma and 20 percent are squamous cell carcinoma. Although the National Cancer Institute does not formally track the incidence and prevalence of nonmelanoma skin cancers, multiple longitudinal studies indicate that the incidence has risen sharply over the past two decades.1
| Clinical recommendation | Evidence rating | References | Comments |
|---|---|---|---|
| Treatment of basal cell carcinoma with Mohs micrographic surgery has the lowest recurrence rate, although it is best considered for tumors greater than 2 cm in size, for more invasive histologic subtypes (micronodular, infiltrative, and morpheaform), or for tumors at sites with higher risk of recurrence. | A | 1, 22 | Systematic review of recurrence rates of basal cell carcinoma with different therapies22 ; review with multiple sources1 |
| Because it is more difficult to control recurrent basal cell carcinoma, incomplete excision of the primary tumor, with pathology demonstrating tumor at the surgical margin, should be followed by immediate reexcision or Mohs micrographic surgery. | C | 24 | — |
| Cryotherapy is an appropriate treatment for nodular and superficial basal cell carcinoma, but is not indicated for tumors more than 3 mm deep. | C | 26 | — |
| Use of topical imiquimod (Aldara) or fluorouracil for the treatment of basal cell carcinoma should be limited to patients with small tumors in low-risk locations who are unwilling or unable to undergo treatment with better-established therapies. | B | 27 | Systematic review evaluating clearance rates and adverse effects of imiquimod and fluorouracil treatment for nonmelanoma skin cancer |
Basal cell carcinoma is the most common type of cancer, with incidence estimates ranging from 124 to 849 per 100,000 persons per year, depending on geographic location.2 The age-adjusted incidence of squamous cell carcinoma is estimated at 100 to 150 per 100,000 persons per year, with a 2:1 male-to-female ratio. Risk is markedly increased with advancing age and closer proximity to the equator.3
Risk Factors and Prevention
The Canadian Program in Evidence-Based Care identifies persons with any of the following factors as having very high risk of skin cancer (i.e., 10 or more times the risk of the general population)4:
Current immunosuppressive therapy after organ transplantation
Personal history of skin cancer
Two or more first-degree relatives with melanoma
Total of 100 nevi or at least five atypical (dysplastic) nevi
More than 250 treatments with psoralen plus ultraviolet A (UVA) therapy for psoriasis
Radiation therapy for cancer as a child
Although both basal cell carcinoma and squamous cell carcinoma traditionally have been attributed to cumulative sun exposure, important differences distinguish the two (Table 1).2,5 Exposure to UVB is the most significant factor in the development of squamous cell carcinoma. Organ transplant recipients have a 65-fold increased risk of squamous cell carcinoma compared with the general population.6

| Characteristics | Basal cell carcinoma | Squamous cell carcinoma |
|---|---|---|
| Ultraviolet exposure | Weaker association, exposure in childhood and adolescence more important | Stronger association, cumulative exposure more important |
| Tumor location | Most tumors (85 percent) occur on the head and neck region, with 25 to 30 percent occurring on the nose alone; does not correlate well with areas of maximal sun exposure; approximately one-third occur on areas that receive little or no ultraviolet exposure5 | More common on the back of the hands and forearms; tumors on the head and neck are most common on areas that receive maximal sun exposure |
| Patient age | Up to 20 percent of tumors occur in patients younger than 50 years2 | Uncommon in patients younger than 50 years |
| Other patient characteristics | Few, if any, identifiable phenotypic markers associated with high risk | Fair skin, blue eyes, red or light colored hair, inability to tan |
The U.S. Preventive Services Task Force has found insufficient evidence to recommend for or against routine screening for skin cancer using whole-body skin examination, but recommends that physicians remain alert for skin lesions with malignant features when performing physical examinations for other reasons.7
The American Cancer Society recommends a cancer-related checkup, including skin examination, every three years in patients 20 to 40 years of age and yearly in patients older than 40 years. It also recommends that patients perform a skin self-examination monthly.8
Use of any tanning device may increase the risk of squamous cell carcinoma by 2.5-fold and the risk of basal cell carcinoma by 1.5-fold, even after adjusting for history of sun exposure.9 Relative risk estimates for squamous cell and basal cell carcinoma steadily increase with younger age at first exposure to a tanning device.9 Although major guidelines for adults do not currently address the use of tanning devices, the Centers for Disease Control and Prevention recommends against the use of tanning beds and lamps in its guidelines for preventing skin cancer in school children.10
Pathophysiology
BASAL CELL CARCINOMA
Basal cell carcinoma arises from basal keratinocytes of the epidermis, hair follicles, and eccrine sweat ducts. Histologically, the cells are basophilic with large nuclei. Because basal cell carcinoma requires surrounding stroma for support during growth, it is virtually incapable of metastasizing via blood or lymphatics.
Basal cell carcinoma has five major histologic patterns: nodular (21 percent), superficial (17 percent), micronodular (15 percent), infiltrative (7 percent), and morpheaform (1 percent). A mixed pattern, containing two or more major histologic patterns, occurs in nearly 40 percent of cases.11
SQUAMOUS CELL CARCINOMA
Actinic keratosis is the principal precursor to squamous cell carcinoma. Actinic keratosis and squamous cell carcinoma represent the same disease process at different stages of evolution, with neoplastic transformation of epidermal keratinocytes generally triggered by UV radiation. As the cells proliferate, they may extend into the dermis, at which point they are termed squamous cell carcinoma.12 Other major risk factors for cutaneous squamous cell carcinoma include chronically diseased or injured skin (e.g., ulcers, sinus tracts), exposure to iodizing or UV radiation, immunosuppression, and xeroderma pigmentosa.1
Cutaneous squamous cell carcinoma spreads by local infiltration and expansion, and may follow tissue planes and conduits, such as nerves and vessels.13 Distant metastasis by hematogenous dissemination occurs in approximately 5 percent of cases.1 Large tumors (greater than 2 cm in diameter) are three times more likely to metastasize than smaller tumors. Squamous cell carcinoma on the lip, ear, or chronically diseased or injured skin, and in patients who are immunosuppressed are also associated with a higher rate of metastasis, exceeding 40 percent.1
Estimates of the 10-year rate of malignant transformation of actinic keratosis range from 6 to 10 percent,14,15 with approximately 60 percent of squamous cell carcinoma arising from actinic keratosis. However, when considering treatment of actinic keratosis, it is important to consider that up to 25 percent of cases spontaneously resolve over 12 months, particularly if patients reduce their UV exposure.16 Thicker tumors, especially those on the scalp, are more likely to progress to squamous cell carcinoma.
Clinical Presentation
BASAL CELL CARCINOMA
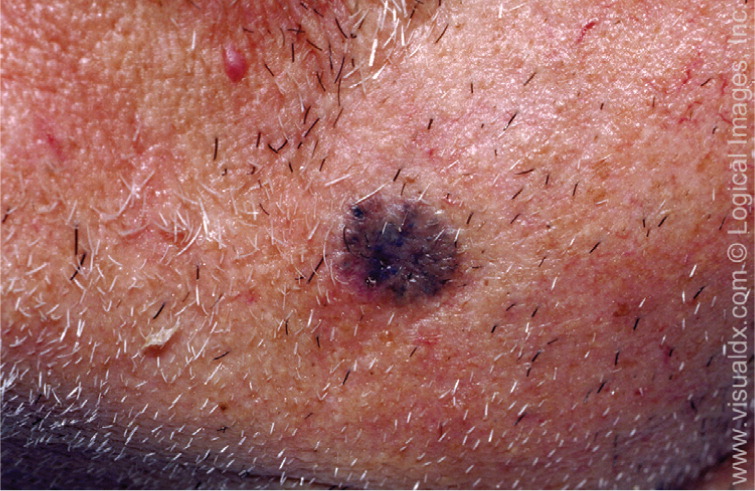
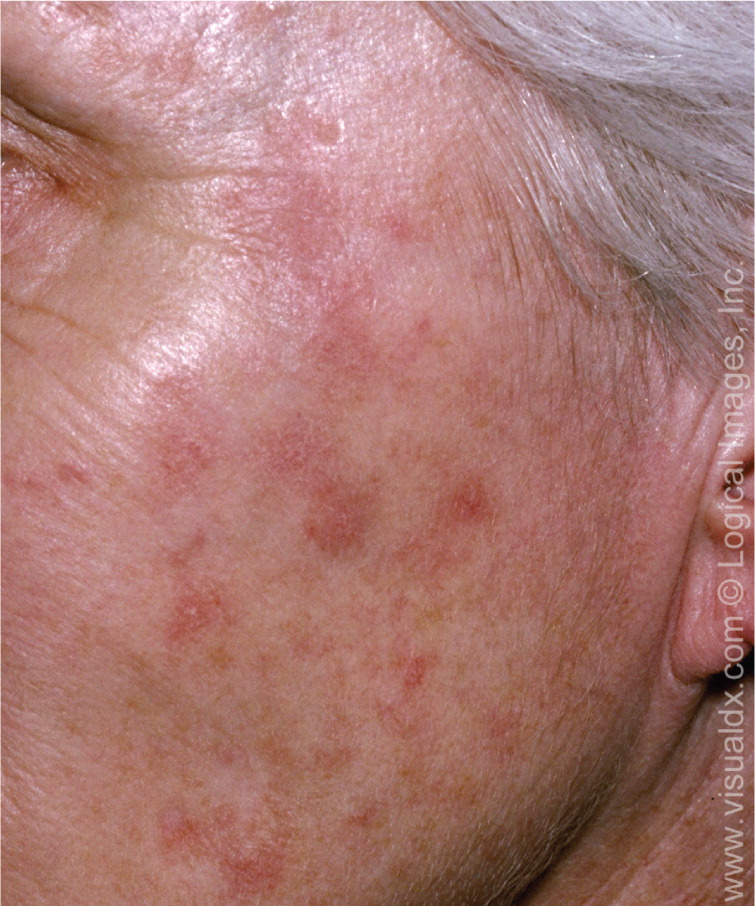
The clinical presentation of nodular basal cell carcinoma is variable (Figures 1 and 2), but it most commonly appears as a pearly pink or white, dome-shaped papule with prominent telangiectatic surface vessels that develop as the lesion enlarges. The tumors are poorly cohesive and have a characteristic texture when removed with curettage. Pigmented basal cell carcinoma (Figure 3) typically has a nodular histologic pattern and contains melanin, which may give the lesion a blue, brown, or black color.17 It is often mistaken for melanoma.
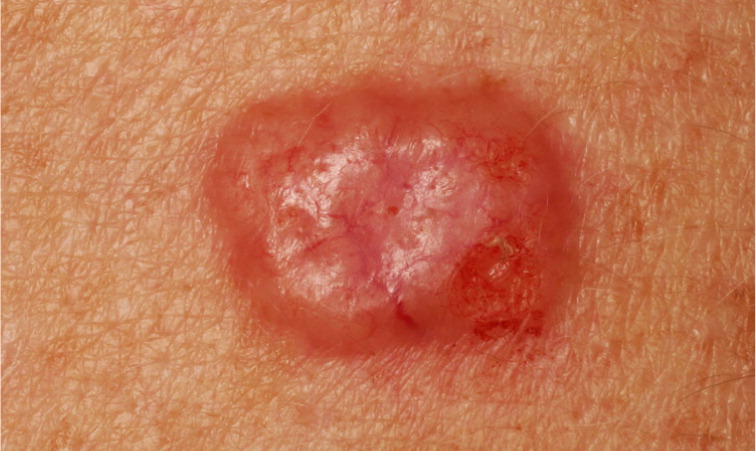
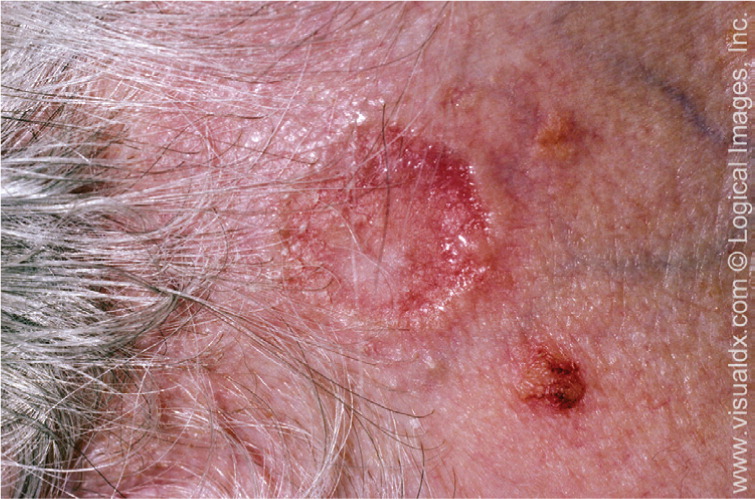
Superficial basal cell carcinoma (Figure 4) most commonly occurs on the trunk and extremities and appears as a scaly plaque resembling eczema or psoriasis, but often retaining the characteristic raised, pearly white borders of the nodular subtype. Superficial basal cell carcinoma is the least invasive of the subtypes.
Micronodular and infiltrative basal cell carcinoma are histologic variants of the nodular pattern, and may be difficult to distinguish clinically from the nodular subtype. However, their malignant potential is greater than that of nodular basal cell carcinoma.
Morpheaform basal cell carcinoma does not exhibit the usual pearly appearance of the nodular subtype. Rather, it resembles localized scleroderma with a firm, yellowish, ill-defined mass that may extend 5 mm or more beyond its clinically apparent borders.18
SQUAMOUS CELL CARCINOMA
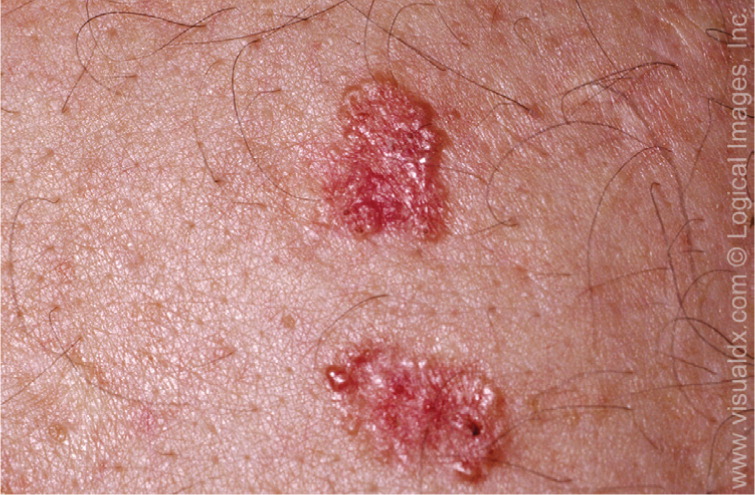
Actinic keratosis (Figure 5) presents as a sharp, scaly plaque on an erythematous base, typically measuring 2 to 6 mm in diameter. The lesion is more easily recognized by palpation than by visual inspection.
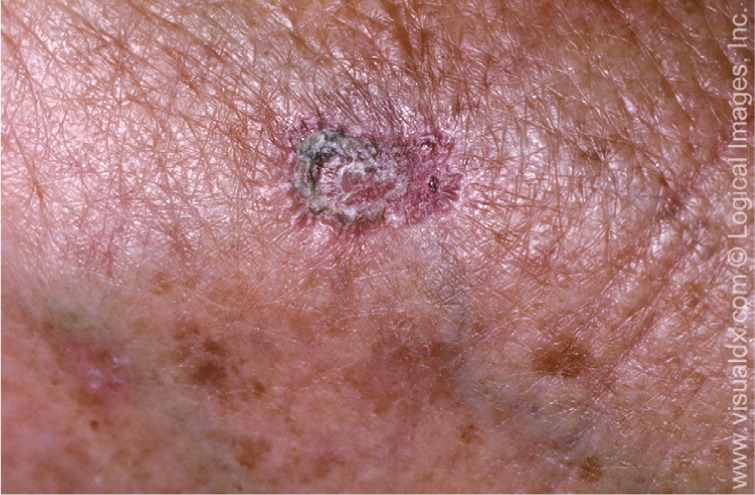
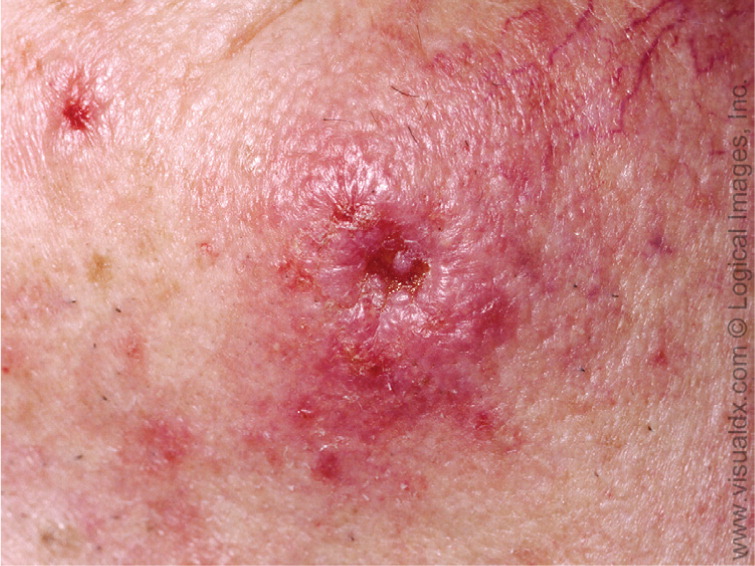
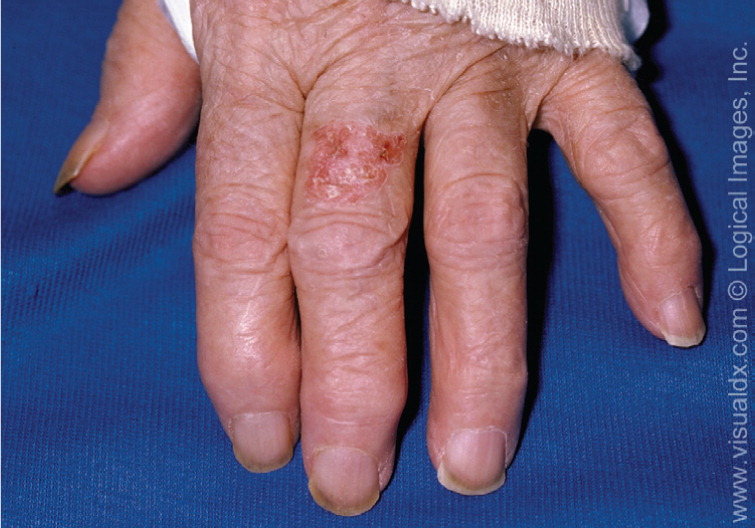
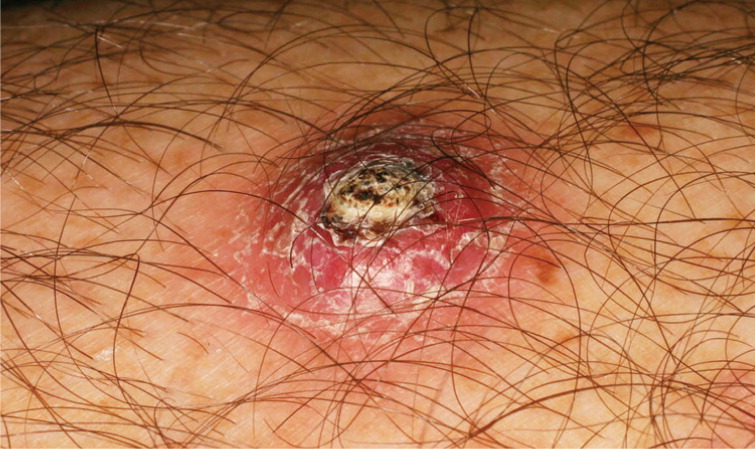
Verrucous carcinoma is a less common but more invasive variant of squamous cell carcinoma that may resemble a large wart. Bowen disease (squamous cell carcinoma in situ) presents as a slowly growing, scaly, red plaque that typically appears on sun-exposed skin19 (Figure 8). A cutaneous horn may begin as an actinic keratosis and degenerate into squamous cell carcinoma.
Diagnosis
Initial tissue sampling for diagnosis of suspected nonmelanoma skin cancer is typically performed using a shave technique if the lesion is raised, or using a 2- to 4-mm punch biopsy of the most abnormal-appearing skin. Complete excision may be an appropriate initial diagnostic procedure for smaller tumors. Pigmented tumors or those eliciting any clinical suspicion of melanoma should always be evaluated using a full-thickness technique. Exfoliative cytology may be considered for evaluation of suspected basal cell carcinoma in patients in whom even a 2-mm punch biopsy would be inappropriate.20
Treatment

| Treatment | Advantages | Disadvantages | Most appropriate use |
|---|---|---|---|
| Superficial ablative techniques | |||
| Electrodesiccation and curettage, cryotherapy |
|
|
|
| Full thickness | |||
| Excision |
|
|
|
| Mohs micrographic surgery |
|
|
|
| Radiotherapy |
|
|
|
BASAL CELL CARCINOMA
A large body of literature addresses recurrence of basal cell carcinoma after various therapies, but lack of uniformity in reporting methods limits the usefulness of these data in developing evidence-based guidelines. Risk of recurrence after treatment is influenced by multiple factors besides treatment modality, including the location, size, and histologic subtype of the tumor and the age, sex, and immune status of the patient.22 Table 3 lists risk factors for recurrence of nonmelanoma skin cancer.23

| Factor | Low risk | High risk |
|---|---|---|
| Basal cell and squamous cell carcinoma | ||
| Location* and size† | Low-risk location and < 20 mm in size | Low-risk location and ≥ 20 mm in size |
| Moderate-risk location and < 10 mm in size | Moderate-risk location and ≥ 10 mm in size | |
| High-risk location and < 6 mm in size‡ | High-risk location and ≥ 6 mm in size‡ | |
| Borders | Well-defined | Poorly defined |
| Primary vs. recurrent | Primary | Recurrent |
| Other | — | Immunosuppression |
| Basal cell carcinoma | ||
| Subtype | Nodular,§ superficial | Aggressive growth pattern∥ |
| Other | — | Site of prior radiotherapy, perineural involvement |
| Squamous cell carcinoma | ||
| Subtype | — | Adenoid (acantholytic), adenosquamous (showing mucin production), or desmoplastic |
| Degree of differentiation | Well differentiated | Moderately or poorly differentiated |
| Depth: Thickness¶ or Clark level | < 2 mm or I, II, III | ≥ 2 mm or IV, V |
| Other | — | Site of prior radiotherapy or chronic inflammatory process, perineural or vascular involvement, neurologic symptoms, rapidly growing tumor |
Treatment of basal cell carcinoma with Mohs micrographic surgery has the lowest recurrence rate. However, because of cost and limited availability, it is best considered for larger tumors (greater than 2 cm), for more invasive histologic subtypes (micronodular, infiltrative, and morpheaform), or for tumors at sites with higher risk of recurrence.1,22 Smaller nodular tumors that are located outside of the H region of the face, which includes the nose, eyelids, chin, jaw, and ear, can be appropriately treated with standard surgical excision. The recurrence rate for tumors treated with Mohs micrographic surgery is approximately 1 percent at five years, whereas standard surgical excision has an approximately 5 percent recurrence rate at five years.22
Because it is difficult to control recurrent basal cell carcinoma, incomplete excision of the primary tumor, with pathology demonstrating tumor at the surgical margin, should be followed by immediate reexcision or Mohs micrographic surgery.24
Recurrence rates after electrodesiccation and curettage of primary tumors occurring in low-, intermediate-, and high-risk sites are 8.6, 12.9, and 17.5 percent, respectively.25
Cryotherapy is an appropriate treatment for nodular and superficial basal cell carcinoma. Biopsy should be performed before the procedure to determine tumor depth, because cryotherapy is not indicated for tumors that are more than 3 mm deep.26 Reported five-year recurrence rates for basal cell carcinoma treated with cryotherapy range from 3.5 to 16.5 percent, depending on tumor size and location.22
A 2009 systematic review of topical imiquimod (Aldara) or fluorouracil in the treatment of nonmelanoma skin cancer found a wide range of clearance rates, depending on drug regimen and tumor type. Evidence supports the use of these therapies as monotherapy for superficial basal cell carcinoma; however, the strength of recommendation is weak. Use of topical imiquimod or fluorouracil should be limited to patients with small tumors in low-risk locations who are unwilling or unable to undergo better-established therapies.27
SQUAMOUS CELL CARCINOMA
Surgical excision, including Mohs micrographic surgery, is recommended as first-line treatment for most squamous cell carcinoma.28 Electrodesiccation and curettage or cryotherapy may be considered for smaller, low-risk lesions. Because squamous cell carcinoma is typically sensitive to radiotherapy, this treatment option is recommended for tumors in high-risk, surgically difficult areas and offers favorable cosmetic results early on. Cosmesis can worsen with time, however, and radiotherapy usually is not considered for patients younger than 55 years.21
A 2010 Cochrane review found little evidence when comparing the effectiveness of different treatments for primary, nonmetastatic squamous cell carcinoma. One trial found that postsurgical chemotherapy with or without radiation treatment had no benefit over surgical excision alone.29
Bowen disease may also be treated with surgical excision, electrodesiccation and curettage, cryotherapy, or topical fluorouracil.30 If actinic keratosis is treated, cryotherapy is the most appropriate choice.31 Topical fluorouracil may be used for more extensive cases, although the intense inflammation associated with the therapy may limit patient tolerance.32
Follow-up and Monitoring
Among the 1,805 patients diagnosed with basal cell or squamous cell carcinoma in a longitudinal study, the risk of subsequent skin cancer was 35 percent at three years and 50 percent at five years. This represented a greater than 10-fold increase in incidence compared with the general population. Risk factors for new tumors included male sex, age older than 60 years, greater number of prior skin cancers, severe actinic skin damage, and increased ease of burning with sun exposure. Smoking history also affected the rate of recurrent squamous cell carcinoma, with current smokers at greater risk (rate ratio = 2.01) than former smokers (rate ratio = 1.62). Smoking history did not affect the rate of recurrent basal cell carcinoma. Recurrent skin cancers tended to be the same cell type as initial tumors.33
Because of inconsistent reporting and monitoring, there are no clear guidelines for follow-up of basal cell carcinoma. In patients with squamous cell carcinoma, up to 95 percent of metastases and local recurrences are detected within five years of initial treatment, with 70 to 90 percent occurring within the first two years. Thus, consistent follow-up for five years after treatment of squamous cell carcinoma is prudent.34
Data Sources: A Medline search included randomized controlled trials, clinical trials, meta-analyses, and systematic reviews. Also searched were the Cochrane Database of Systematic Reviews, Agency for Healthcare Research and Quality Evidence Reports, National Guideline Clearinghouse, U.S. Preventive Services Task Force, and Centers for Disease Control and Prevention. Keywords were basal cell carcinoma, squamous cell carcinoma, nonmelanoma skin cancer, diagnosis, treatment, prevention, and screening. Limits included English language, humans, and adults 19 years and older. Search dates: August 2010 and June 2011.
