
A more recent article on over-the-counter medications in pregnancy is available.
This is a corrected version of the article that appeared in print.
Am Fam Physician. 2014;90(8):548-555
Author disclosure: No relevant financial affiliations.
Many pregnant women take over-the-counter (OTC) medications despite the absence of randomized controlled trials to guide their use during pregnancy. Most data come from case-control and cohort studies. In 1979, the U.S. Food and Drug Administration began reviewing all prescription and OTC medications to develop risk categories for use in pregnancy. Most OTC medications taken during pregnancy are for allergy, respiratory, gastrointestinal, or skin conditions, as well as for general analgesia. Acetaminophen, which is used by about 65% of pregnant women, is generally considered safe during any trimester. Cold medications are also commonly used and are considered safe for short-term use outside of the first trimester. Many gastrointestinal medications are now available OTC. Histamine H2 blockers and proton pump inhibitors have not demonstrated significant fetal effects. Nonsteroidal anti-inflammatory drugs are generally not recommended in pregnancy, especially during organogenesis and in the third trimester. There is even fewer data regarding use of individual herbal supplements. Ginger is considered safe and effective for treating nausea in pregnancy. Topical creams are considered safe based on small studies and previous practice. All OTC medication use should be discussed with patients, and the effects of the symptoms should be balanced with the risks and benefits of each medication. Because of the expanding OTC market, formalized studies are warranted for patients to make a safe and informed decision about OTC medication use during pregnancy.
More than 90% of pregnant women take a prescription or over-the-counter (OTC) medication.1 Although there are no randomized controlled trials to guide the use of OTC medications during pregnancy, women often use them for skin, allergy, respiratory, and gastrointestinal conditions in addition to general analgesia. All physicians caring for reproductive-aged women should be familiar with the indications, risks, and benefits of OTC medications in pregnancy. Given limited data on the variety of OTC medications available, physicians need to counsel pregnant women about potential risks, and it is beneficial to discuss all OTC medications the patient is taking at the preconception visit and all other routine visits. Table 1 lists online resources for more information about OTC medication use during pregnancy.
| Clinical recommendation | Evidence rating | References |
|---|---|---|
| First- and second-generation antihistamines do not appear to increase fetal risk in any trimester. | B | 5–9 |
| Acetaminophen as a single agent does not increase fetal risk in any trimester and is considered safe for use in pregnancy. | B | 28, 31, 32, 35 |
| Use of nonsteroidal anti-inflammatory drugs during pregnancy has potential risks. The risk-benefit ratio is best determined with physician consultation. | C | 29, 36–39 |
| Histamine H2 blockers and proton pump inhibitors can be used during any trimester of pregnancy without risk of anomalies. | B | 55–58 |

| Centers for Disease Control and Prevention | |
| http://www.cdc.gov/pregnancy/meds (facts sheets for patients; link to LacMed, a database of medications that might be used during lactation) | |
| Clinical Pharmacology | |
| http://www.clinicalpharmacology.com (medication summaries, used by most pharmacies, membership required) | |
| National Library of Medicine | |
| http://dailymed.nlm.nih.gov/dailymed/about.cfm (information for physicians and patients; more current than U.S. Food and Drug Administration website) | |
| Online Physicians' Desk Reference | |
| http://www.pdr.net (medication reference for physicians, membership required) | |
| Organization of Teratology Information Specialists | |
| http://www.mothertobaby.org/fact-sheets-s13037 (fact sheets for patients) | |
| Reproductive Toxicology Center | |
| http://www.reprotox.org (medication summaries; membership required) | |
| University of Toronto, The Hospital for Sick Children, Motherisk Program | |
| http://www.motherisk.org/women/drugs.jsp (index of medications, including over-the-counter) | |
| http://www.motherisk.org/women/mothernature.jsp (index of herbal products) | |
| U.S. Food and Drug Administration | |
| http://www.fda.gov/ForConsumers/ByAudience/ForWomen/WomensHealthTopics/ucm117976.htm#Medicine_and_Pregnancy (resources for patients) | |
Since 1979, a standard five-letter nomenclature developed by the U.S. Food and Drug Administration (FDA) has been used to assign a pregnancy risk category to prescription and OTC medications (Table 2).2 In response to ongoing criticism of the confusing and simplistic nature of this system, in 2011, the FDA proposed a new rule for labeling that aims to provide more detailed safety data about use in pregnancy and in turn improve clinical decision making.3 The new rule divides information into pregnancy and breastfeeding categories, each with the subcategories of risk summary, clinical considerations, and data. The five-letter system and the new system are both currently available.

| Category | Definition |
|---|---|
| A | Controlled studies in pregnant women fail to demonstrate a risk to the fetus in the first trimester, there is no evidence of risk in later trimesters, and the possibility of fetal harm appears remote. |
| B | Either animal-reproduction studies have not demonstrated a fetal risk but there are no controlled studies in pregnant women, or animal-reproduction studies have shown an adverse effect (other than a decrease in fertility) that was not confirmed in controlled studies in women in the first trimester (and there is no evidence of a risk in later trimesters). |
| C | Either studies in animals have revealed adverse effects on the fetus (teratogenic or embryocidal or other) and there are no controlled studies in women, or studies in women and animals are not available. Drugs should be given only if the potential benefit justifies the potential risk to the fetus. |
| D | There is positive evidence of human fetal risk, but the benefits from use in pregnant women may be acceptable despite the risk (e.g., if the drug is needed in a life-threatening situation or for a serious disease for which safer drugs cannot be used or are ineffective). |
| X | Studies in animals or humans have demonstrated fetal abnormalities or there is evidence of fetal risk based on human experience, or both, and the risk of the use of the drug in pregnant women clearly outweighs any possible benefit. The drug is contraindicated in women who are or may become pregnant. |
OTC medications that are not available as a prescription often do not get safety ratings, and the FDA website is not often updated after a product has initial approval. Multiple websites and databases with conflicting data make counseling women more difficult. Using the lowest dose for the shortest period possible and trying to avoid medication use during the first trimester are reasonable approaches.
Antihistamines
Up to 15% of women use an antihistamine during pregnancy to treat allergic rhinitis or nausea.4 Studies consistently show no significant risk of fetal malformations with first-generation antihistamines, and these agents are considered safe.5–8 The second-generation antihistamines loratadine (Claritin), cetirizine (Zyrtec), and fexofenadine (Allegra) do not appear to increase overall fetal risk. Four studies (n = 1,290) did not find significant fetal risk with cetirizine use.5,9 A slightly higher incidence of hypospadias with loratadine use was shown in one study (n = 1,700), but not in others (n = 2,147).5,8 Fexofenadine has been associated with early pregnancy loss in animal studies but has not been studied in human pregnancy. Fexofenadine is a metabolite of terfenadine, which was removed from the market in 1998 because of a risk of cardiotoxicity. Studies (n = 2,195) on the safety of terfenadine in human pregnancy did not show a significant risk of congenital malformation.5
Data addressing the safety of topical antihistamines in pregnancy are limited to a single study of the ophthalmic agent pheniramine, which is contained in several OTC combinations with naphazoline. No significant malformations were observed in 831 women who used the medication in the first trimester.5 There are no data for other topical antihistamines, such as those in anti-itch creams; however, significant fetal risk is unlikely because of the lack of systemic absorption. Table 3 summarizes the safety of antihistamines in pregnancy.10–16
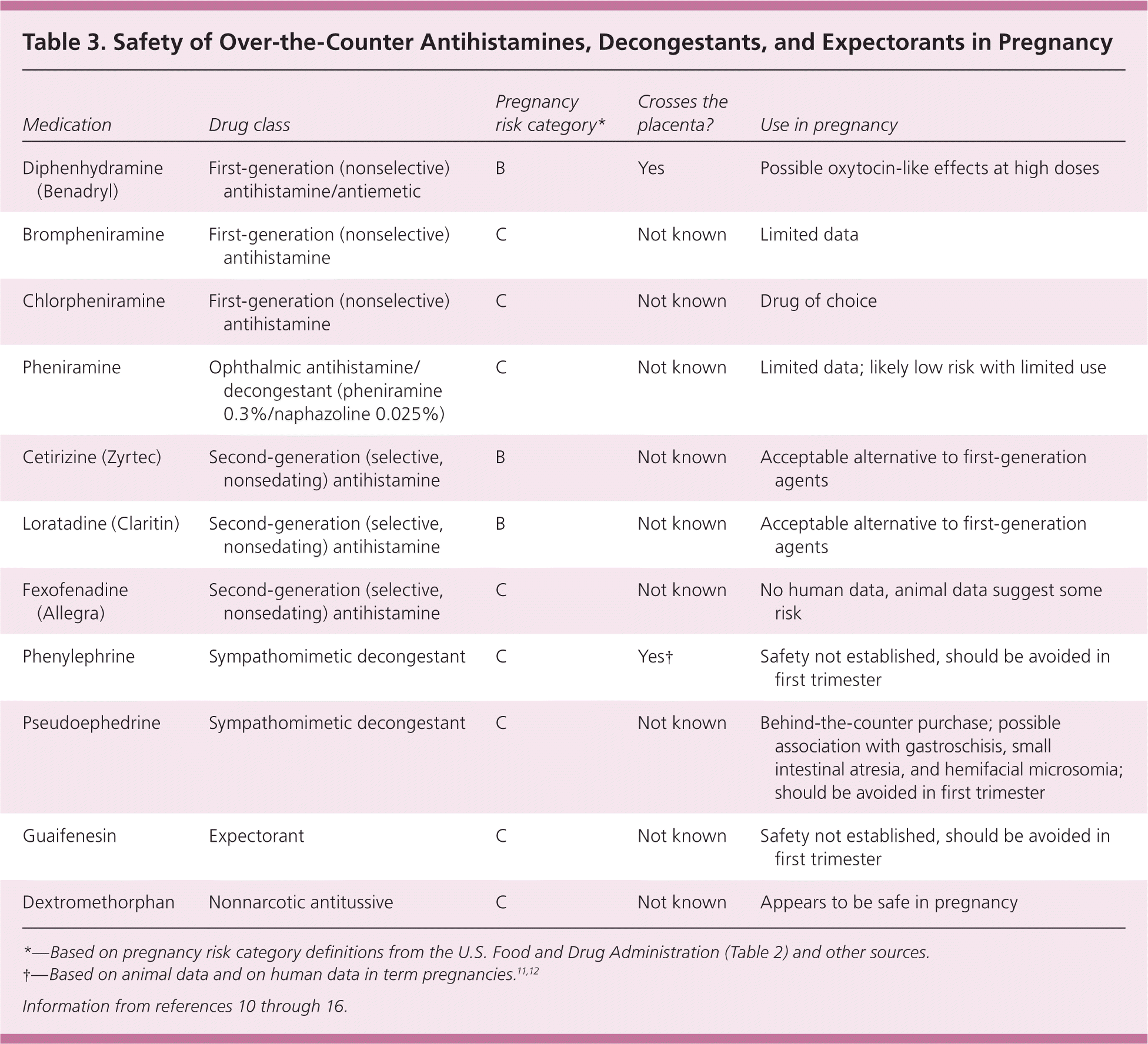
| Medication | Drug class | Pregnancy risk category* | Crosses the placenta? | Use in pregnancy |
|---|---|---|---|---|
| Diphenhydramine (Benadryl) | First-generation (nonselective) antihistamine/antiemetic | B | Yes | Possible oxytocin-like effects at high doses |
| Brompheniramine | First-generation (nonselective) antihistamine | C | Not known | Limited data |
| Chlorpheniramine | First-generation (nonselective) antihistamine | C | Not known | Drug of choice |
| Pheniramine | Ophthalmic antihistamine/decongestant (pheniramine 0.3%/naphazoline 0.025%) | C | Not known | Limited data; likely low risk with limited use |
| Cetirizine (Zyrtec) | Second-generation (selective, nonsedating) antihistamine | B | Not known | Acceptable alternative to first-generation agents |
| Loratadine (Claritin) | Second-generation (selective, nonsedating) antihistamine | B | Not known | Acceptable alternative to first-generation agents |
| Fexofenadine (Allegra) | Second-generation (selective, nonsedating) antihistamine | C | Not known | No human data, animal data suggest some risk |
| Phenylephrine | Sympathomimetic decongestant | C | Yes† | Safety not established, should be avoided in first trimester |
| Pseudoephedrine | Sympathomimetic decongestant | C | Not known | Behind-the-counter purchase; possible association with gastroschisis, small intestinal atresia, and hemifacial microsomia; should be avoided in first trimester |
| Guaifenesin | Expectorant | C | Not known | Safety not established, should be avoided in first trimester |
| Dextromethorphan | Nonnarcotic antitussive | C | Not known | Appears to be safe in pregnancy |
Decongestants
Nearly one in four pregnant women seeks relief from nasal congestion caused by upper respiratory tract infection, allergic rhinitis, or the common phenomenon known as pregnancy rhinitis.4 The safety of oral phenylephrine in pregnancy has not been established. Data that are now about a decade old (n = 2,730) show an increased risk of congenital malformation (relative risk = 0.6 to 1.2) and of eye, ear, and minor limb malformations (relative risk = 2.7) with phenylephrine use during pregnancy.11,12,17 Pseudoephedrine was previously considered low risk in pregnancy based on older cohort studies (n = 1,724) demonstrating no significant teratogenicity.12 However, its safety was brought into question after recent case-control studies observed small associations between pseudoephedrine and birth defects, including gastroschisis, small intestinal atresia, and hemifacial microsomia.12,17–20 The studies are limited by small sample size; retrospective analysis; and potential for confounding factors, such as recall bias. Risk of ventricular septal defects or limb malformations has been observed with decongestants but have not been substantiated.17,21,22
Two studies (n = 5,400) show a decreased risk of preterm birth, low birth weight, and preterm labor among women using a variety of oral decongestants in pregnancy.23,24 There are only a few studies on the safety of topical (nasal and ophthalmic) decongestants, none of which demonstrate increased fetal risk.6,19,20
Overall, available evidence suggests that decongestants (and combination formulations) should be used sparingly in pregnancy, particularly in the first trimester; however, further study is needed. Saline nasal sprays and adhesive nasal strips are safe OTC alternatives for treating nasal congestion. Table 3summarizes the safety of decongestants in pregnancy.10–16
Expectorants and Antitussives
Few studies have addressed the safety of using cough medications during pregnancy. The expectorant guaifenesin has been weakly associated with neural tube defects and inguinal hernias. However, the evidence is not sufficient to determine its safety in pregnancy. It may be prudent to avoid this medication in the first trimester unless the potential benefits outweigh the risks.25 Table 3 summarizes the safety of expectorants in pregnancy.10–16
Dextromethorphan is a nonnarcotic antitussive isomer of codeine that was found to be teratogenic in chicken embryos. However, a human epidemiologic study and a smaller controlled study did not demonstrate elevated risks of congenital malformations.26
Analgesics and Antipyretics
There are no prospective randomized controlled trials to determine the safety of acetaminophen, ibuprofen, or naproxen use in pregnancy. At least two-thirds of women use acetaminophen during pregnancy, and one-half of these women use it in the first trimester.1,4,27 Animal studies suggest that acetaminophen may decrease the diameter of the ductus arteriosus, but experimental conditions prevent reasonable extrapolation to humans.28 More recent studies have looked at chronic acetaminophen use during pregnancy and the risk of tetralogy of fallot, but no definitive connection has been made.29 There is conflicting evidence about the risk of gastroschisis, leukemia, and asthma with acetaminophen use.30 A Danish prospective population-based study (n = 88,142) showed that the hazard ratio for congenital defects was 1.01 for the 26,424 women who took acetaminophen in the first trimester.31 In a follow-up analysis, the hazard ratio for cryptorchidism was 1.38, but only with more than four weeks of regular acetaminophen use in the first and second trimesters.32 Other, newer cohort studies have looked at the possible connection between acetaminophen use and attention-deficit/hyperactivity disorder and other hyperkinetic disorders.33,34
The National Birth Defects Prevention Study (NBDPS), which analyzed data from 16,110 children in the United States exposed to acetaminophen in utero, found no increased risk of birth defects with acetaminophen use. In women using acetaminophen specifically for febrile illness, there were decreased risks of various cranial and facial defects and gastroschisis; acetaminophen may be protective because fever increases the risk of these defects.35 A case series of 300 acetaminophen overdoses in pregnant women found no increased risk of congenital defects, stillbirth, or spontaneous abortions, regardless of trimester. At six weeks of life, the newborns had no evidence of hepatic or renal disease.28 Many trials study acetaminophen in combination with cold remedies, rather than as a single agent, making causality difficult. The available information on acetaminophen use does not establish fetal risks; therefore, as a single agent, it is safe for use during any trimester, especially as single dosing without routine use.
A meta-analysis of aspirin use in the first trimester did not demonstrate an increased risk of congenital anomalies, except for gastroschisis (odds ratio [OR] = 2.37).36,37 Early aspirin use at the time of conception or in the first several weeks of pregnancy does not increase the risk of spontaneous abortion.38 Aspirin has been studied extensively as a treatment for many chronic disorders in pregnant women, including thromboembolism, antiphospholipid disease, and preeclampsia. There can be risks of intrauterine growth retardation and fetal and maternal hemorrhage in the third trimester. Overall, aspirin should be avoided during organogenesis and in the third trimester unless a physician specifically prescribes it and the patient understands the risks and benefits.
In a recent study, neither ibuprofen nor naproxen increased the risk of spontaneous abortion when used in the first six weeks of pregnancy.38 A Swedish study of nonsteroidal anti-inflammatory drug (NSAID) use in early pregnancy did not demonstrate an increased risk of congenital anomalies overall; however, naproxen was associated with orofacial clefts, and all NSAIDs were associated with structural cardiac defects.39 More recent data show a potential association between NSAID use and dextro-transposition of the great arteries, particularly in the first trimester.29 NSAIDs are not recommended in the third trimester because of the risk of premature closure of the ductus arteriosus and subsequent primary pulmonary hypertension in the newborn. Because indomethacin (Indocin) is known to cause oligohydramnios and delay delivery, OTC NSAIDs are assumed to have the same risk. Although NSAID use is generally not recommended during pregnancy, women may ingest these medications inadvertently in many OTC combinations. Prolonged use of NSAIDs, including aspirin, should occur only for specific medical indications during pregnancy. Table 4 summarizes the safety of analgesics and antipyretics in pregnancy.10–16
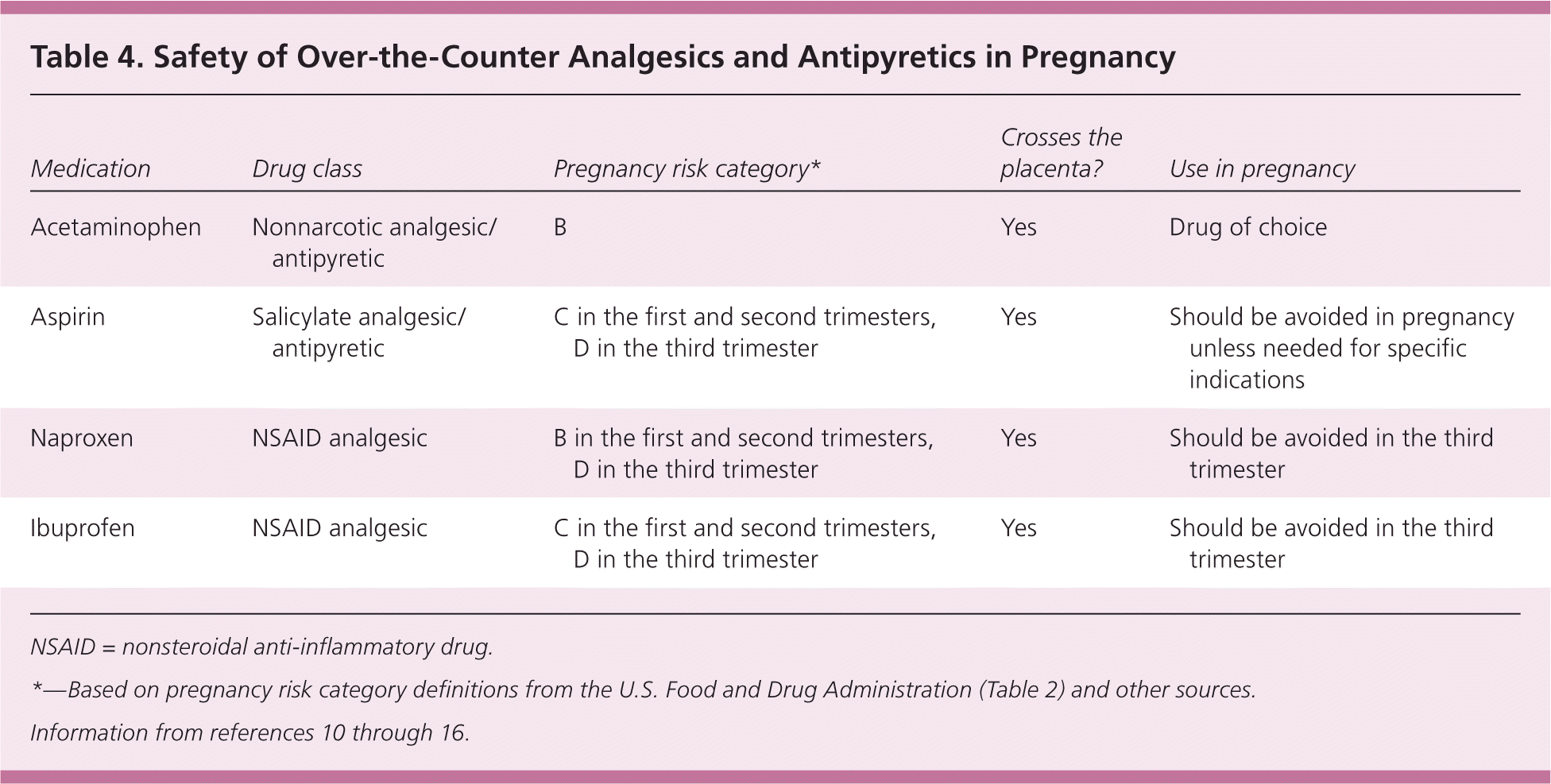
| Medication | Drug class | Pregnancy risk category* | Crosses the placenta? | Use in pregnancy |
|---|---|---|---|---|
| Acetaminophen | Nonnarcotic analgesic/antipyretic | B | Yes | Drug of choice |
| Aspirin | Salicylate analgesic/antipyretic | C in the first and second trimesters, D in the third trimester | Yes | Should be avoided in pregnancy unless needed for specific indications |
| Naproxen | NSAID analgesic | C in the first and second trimesters, D in the third trimester [ corrected] | Yes | Should be avoided in the third trimester |
| Ibuprofen | NSAID analgesic | C in the first and second trimesters, D in the third trimester | Yes | Should be avoided in the third trimester |
Herbals and Dietary Supplements
During pregnancy, herbal remedies are used for nausea, respiratory symptoms, urinary tract infections, pain, and other nonspecific issues.40 However, there are few human data on the safety of herbal remedies in pregnancy. The Dietary Supplement Health and Education Act of 1994 requires manufacturers to ensure the safety of supplements before marketing. However, there is no registration process with the FDA, which takes action only if a supplement is found to be unsafe after marketing.41 Herbals were not included in the NBDPS until the year 2000. According to a subanalysis of the NBDPS, 10.9% of women use herbals during pregnancy, most commonly peppermint, cranberry extract, herbal teas, ginger, chamomile, Echinacea, ginseng, raspberry leaf, and ephedra products.42
St. John's wort is generally not recommended in pregnancy because of a lack of human data.42,43 Echinacea can be used topically or orally. A study with 112 women who used Echinacea in the first trimester showed no increased risk of malformations.44 Feverfew is used for migraine prophylaxis. It inhibits platelet aggregation and prostaglandin production and is contraindicated in pregnancy. Multiple herbals, such as mugwort, blue cohosh, black cohosh, goldenseal, juniper berry, chaste berry, rue, and pennyroyal oil, are uterine stimulants or abortifacients and should be avoided in pregnancy.45 Although ephedra is commonly used during pregnancy according to patient report, it has a significant association with birth defects. According to the NBDPS, ephedra is associated with anencephaly (OR = 2.8).46 Other weight loss products, with or without ephedra, are associated with dextro-transposition of the great vessels and aortic stenosis.46
Glucosamine has been used by pregnant women with painful arthritis and appears to be safe. In a case-control study of 54 women, there was only one major malformation in the glucosamine group, which was comparable to the baseline rate of birth defects, and there was no difference in the risk of stillbirth, abortion, preterm birth, or other maternal morbidity.47 Ginger is commonly used in the first trimester and can be found in some prenatal vitamins. Although there have been concerns about ginger increasing the risk of spontaneous abortion or preterm delivery, this has not been demonstrated in animal studies. Two systematic reviews demonstrated that ginger improves pregnancy-related nausea more than placebo and as effectively as vitamin B6. Its effect on vomiting is less certain. No adverse effects have been noted for the mother or developing fetus.45,48 Ginger is the only dietary supplement that can be recommended based on human studies.45,48
Topical Creams
Topical antifungals are commonly used during pregnancy for treatment of vulvovaginitis. Imidazoles and nystatin are well studied and considered safe during pregnancy.49–51 Systemic absorption of imidazoles varies from 1% with miconazole to 10% with clotrimazole; nystatin is negligibly absorbed. Terbinafine (Lamisil) is sold OTC as a 1% cream. No studies are available for terbinafine cream; however, the oral form is pregnancy category B.51
Hydrocortisone 1% is the only topical corticosteroid cream available OTC. Systemic absorption ranges from 1% to 7%, depending on the area treated and the underlying skin condition.42 Although potent topical corticosteroids may have increased risks in pregnancy, the mild OTC forms are considered safe. As with all steroid use, the lowest dose used for the shortest time possible is recommended.52
Smaller studies have not shown an association between use of the topical antimicrobial bacitracin and fetal malformations.53 There are no studies regarding the safety of benzoyl peroxide use in pregnancy; however, the limited absorption of 5% suggests that it carries minimal risk.42 Overall, topical OTC antifungal, antimicrobial, and steroid creams are safe in pregnancy.
Antacids and Antidiarrheals
Heartburn occurs in up to 80% of pregnant women by the end of the third trimester. Antacids containing aluminum, calcium, or magnesium are often considered first-line treatment in pregnancy. However, at high doses, antacids containing calcium can cause milk-alkali syndrome,54 and antacids with aluminum can cause neurotoxicity. Selective histamine H2 blockers have been used in all trimesters with no known teratogenic effects. In a meta-analysis of 2,398 women taking H2 blockers, the OR for congenital malformations was 1.14.55
Proton pump inhibitors recently became available OTC. Although concerns have been raised about the potential teratogenicity of omeprazole (Prilosec), multiple large cohort studies have demonstrated its safety when taken before conception and during the first trimester.56 In a meta-analysis of 1,530 infants exposed to proton pump inhibitors, the OR for congenital malformations was 1.12 overall and 1.17 for omeprazole alone, and there was no increased risk of preterm birth or spontaneous abortion.57 In another study of proton pump inhibitor use in the first trimester (n = 5,082), the OR for birth defects was 1.10. Proton pump inhibitors and H2 blockers are considered safe in pregnancy.58
Diarrhea and constipation are common during pregnancy. Products containing bismuth, mineral oil, and castor oil should be avoided. Bismuth itself is safe, but it has the same risks as aspirin when combined with salicylate.59 In a study of 89 women, loperamide (Imodium) did not increase the risk of malformation, but was associated with smaller infants.60 However, in a later study of 638 women, loperamide had an OR of 1.43 for congenital malformations. Although the American Gastroenterological Association considers loperamide to be low risk, it should be avoided when possible until further information is available.59,61 Saline laxatives may cause electrolyte sodium retention and should be used sparingly.62 Polyethylene glycol 3350 (Miralax) has minimal systemic absorption and is considered the drug of choice for chronic constipation despite a lack of research.59,62 Table 5 summarizes the safety of OTC antacids, antidiarrheals, and laxatives in pregnancy.10–16
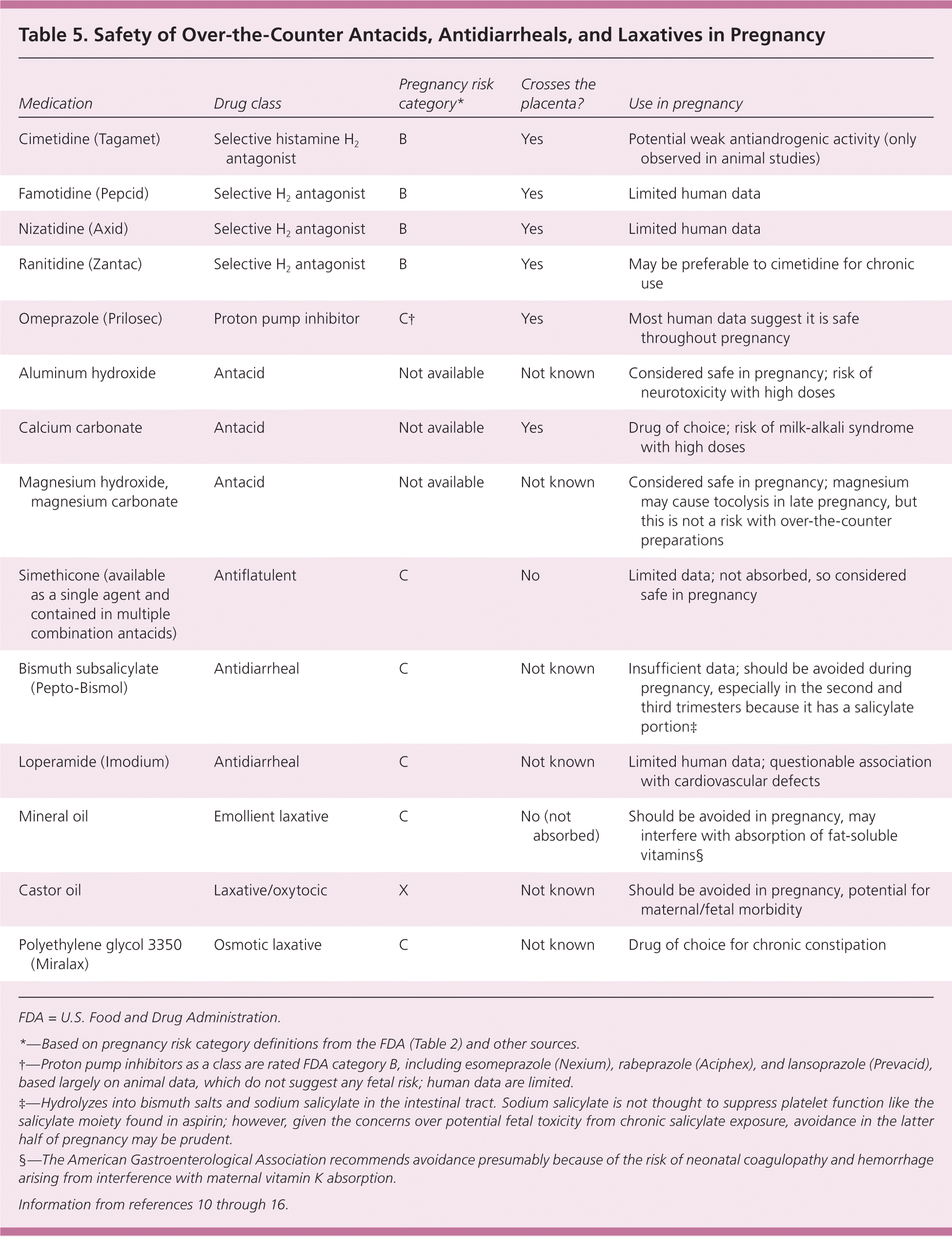
| Medication | Drug class | Pregnancy risk category* | Crosses the placenta? | Use in pregnancy |
|---|---|---|---|---|
| Cimetidine (Tagamet) | Selective histamine H2 antagonist | B | Yes | Potential weak antiandrogenic activity (only observed in animal studies) |
| Famotidine (Pepcid) | Selective H2 antagonist | B | Yes | Limited human data |
| Nizatidine (Axid) | Selective H2 antagonist | B | Yes | Limited human data |
| Ranitidine (Zantac) | Selective H2 antagonist | B | Yes | May be preferable to cimetidine for chronic use |
| Omeprazole (Prilosec) | Proton pump inhibitor | C† | Yes | Most human data suggest it is safe throughout pregnancy |
| Aluminum hydroxide | Antacid | Not available | Not known | Considered safe in pregnancy; risk of neurotoxicity with high doses |
| Calcium carbonate | Antacid | Not available | Yes | Drug of choice; risk of milk-alkali syndrome with high doses |
| Magnesium hydroxide, magnesium carbonate | Antacid | Not available | Not known | Considered safe in pregnancy; magnesium may cause tocolysis in late pregnancy, but this is not a risk with over-the-counter preparations |
| Simethicone (available as a single agent and contained in multiple combination antacids) | Antiflatulent | C | No | Limited data; not absorbed, so considered safe in pregnancy |
| Bismuth subsalicylate (Pepto-Bismol) | Antidiarrheal | C | Not known | Insufficient data; should be avoided during pregnancy, especially in the second and third trimesters because it has a salicylate portion‡ |
| Loperamide (Imodium) | Antidiarrheal | C | Not known | Limited human data; questionable association with cardiovascular defects |
| Mineral oil | Emollient laxative | C | No (not absorbed) | Should be avoided in pregnancy, may interfere with absorption of fat-soluble vitamins§ |
| Castor oil | Laxative/oxytocic | X | Not known | Should be avoided in pregnancy, potential for maternal/fetal morbidity |
| Polyethylene glycol 3350 (Miralax) | Osmotic laxative | C | Not known | Drug of choice for chronic constipation |
Data Sources: We searched PubMed, UpToDate, the National Guideline Clearinghouse, and the Cochrane database using the terms over-the-counter, medicine, and pregnancy; herbals and pregnancy; and individual drug names in combination with pregnancy. Search dates: February to July 2012, July 2014.
The authors thank pharmacist Stephanie Owens for querying the Clinical Pharmacology database.
The opinions herein are those of the authors. They do not represent official policy of the Uniformed Services University of the Health Sciences, the Department of the Air Force, or the Department of Defense.
