
Am Fam Physician. 2013;88(10):710-716
Guideline source: American Academy of Pediatrics
Evidence rating system used? Yes
Literature search described? Yes
Guideline developed by participants without relevant financial ties to industry? No
Published source: Pediatrics, February 2013
Childhood obesity has dramatically increased in North America over the past 30 years, leading to earlier onset of type 2 diabetes mellitus, which previously did not normally occur until later in life. Childhood type 2 diabetes poses a challenge to many physicians who are not used to treating this disease in such young patients.
The American Academy of Pediatrics (AAP) has released a guideline of evidence-based recommendations for treating type 2 diabetes in patients 10 to 18 years of age. The guideline focuses on treatments that have been shown to affect clinical outcomes in these patients.
Definitions
Childhood type 2 diabetes is disease in a child who typically:
Is overweight (body mass index in the 85th to 94th percentile) or obese (body mass index in the 95th percentile or greater)
Has a strong family history of type 2 diabetes
Has substantial residual insulin secretory capacity at diagnosis (reflected by normal or elevated insulin and C-peptide concentrations)
Has insidious onset of disease
Demonstrates insulin resistance (including clinical evidence of polycystic ovary syndrome or acanthosis nigricans)
Lacks evidence for diabetic autoimmunity (negative for autoantibodies typically associated with type 1 diabetes); these patients are more likely to have hypertension and dyslipidemia than are those with type 1 diabetes
Diabetes is defined as one of the following:
A1C level of 6.5% or greater (test performed in an appropriately certified laboratory)
Fasting (no caloric intake for at least eight hours) plasma glucose level of 126 mg per dL (7.0 mmol per L) or greater
Two-hour plasma glucose level of 200 mg per dL (11.1 mmol per L) or greater during an oral glucose tolerance test (using a glucose load containing the equivalent of 75 g anhydrous glucose dissolved in water)
A random plasma glucose level of 200 mg per dL or greater with symptoms of hyperglycemia
Action Statements
Clinicians must ensure that insulin therapy is initiated in children and adolescents with ketosis or diabetic ketoacidosis, in whom the distinction between type 1 and 2 diabetes is unclear. Usually, insulin therapy should be initiated in patients with a random venous or plasma blood glucose concentration of 250 mg per dL (13.9 mmol per L) or greater, or an A1C level greater than 9%.
Type 2 diabetes in children and adolescents may present differently depending on disease stage. In early disease, before diagnostic criteria are met, blood glucose concentrations may be normal much of the time and patients may be asymptomatic. At this stage, the disease may be detected only with abnormal blood glucose measurements during screening. As insulin secretions decline, patients will likely begin to have symptoms of hyperglycemia, sometimes with ketosis or frank ketoacidosis.
Diabetic ketoacidosis is treated with insulin and fluid, as well as electrolyte replacement to prevent the disease from worsening. These patients need immediate inpatient therapy under the care of a physician experienced in treating diabetic ketoacidosis.
Children and adolescents who have type 2 diabetes and poor glycemic control, but no evidence of ketosis or ketoacidosis, may also benefit from insulin therapy, at least short term. Initially, it may be difficult to differentiate type 1 and type 2 diabetes. Insulin therapy should be administered while appropriate testing is performed.
Clinicians should initiate a lifestyle modification program, including nutrition and physical activity, and start metformin (Glucophage) as first-line therapy at the time of type 2 diabetes diagnosis, unless insulin is needed to reverse glucose toxicity in the case of significant hyperglycemia or ketoacidosis.
Because the combination of diet and exercise alone has not been highly successful in children and adolescents with type 2 diabetes, lifestyle modifications should be initiated with metformin. Gastrointestinal adverse effects are common at the beginning of metformin therapy; therefore, it should be started at a low dosage of 500 mg daily, and increased by 500 mg every one to two weeks to a maximum of 2,000 mg daily in divided doses. Extended-release metformin may be considered, although data regarding adverse effects are lacking.
In children and adolescents, a multidisciplinary approach to lifestyle modifications with the involvement of the whole family is most successful. Expert consensus is that less than 10% of children and adolescents will maintain lifestyle changes long term. Patients are more likely to succeed if they are treated simultaneously with medications, possibly because they will have a greater degree of concern for their health than if medication is not needed.
In patients with more modest hyperglycemia (e.g., random blood glucose level of 200 to 249 mg per dL [11.1 to 13.8 mmol per L]) or asymptomatic type 2 diabetes, metformin alone, insulin alone, or metformin and insulin are reasonable options for initial pharmacologic management.
A1C concentrations should be monitored every three months and treatment intensified if results do not meet the goals for finger-stick blood glucose and A1C concentrations.
The A1C target ideally should be less than 7%. If this seems unattainable for a patient, an individualized goal should be set, with the intention of ultimately meeting the guideline target of less than 7%. In the absence of hypoglycemia, a lower goal can be considered.
If goals are not reached, therapy should be intensified if possible. This may include increasing clinic visits, monitoring blood glucose levels more often, adding one or more antidiabetic agents, consulting a registered dietitian or diabetes educator, and increasing attention to diet and exercise regimens. If A1C concentrations remain relatively stable, testing may be extended to every six months.
Clinicians may advise patients to monitor finger-stick blood glucose concentrations if they are taking insulin or other medications with a risk of hypoglycemia; are starting or changing a diabetes treatment regimen; have not met treatment goals; or have comorbid illnesses.
Although the benefit of frequent blood glucose monitoring has not been evaluated in children and adolescents with type 2 diabetes, a fasting blood glucose concentration of 70 to 130 mg per dL (3.9 to 7.2 mmol per L) is a reasonable goal in most adolescents.
Current American Diabetes Association recommendations for finger-stick monitoring may be applied to most children and adolescents with type 2 diabetes: (1) monitoring should be performed at least three times daily for patients using multiple insulin injections or insulin pump therapy; (2) for patients using less frequent insulin injections, noninsulin therapies, or medical nutrition therapy alone, monitoring may be useful as a guide to the success of therapy; and (3) to achieve postprandial glucose targets, postprandial finger-stick monitoring may be appropriate.
All patients with a new diagnosis should perform finger-stick blood glucose testing before meals (including the morning fasting concentration) and at bedtime, regardless of treatment plan. The frequency of testing can be decreased in some patients after the target level is met. Patients and families should receive a written action plan.
In patients using a regimen consisting of a single bedtime insulin injection (basal insulin only), the best way to determine the appropriate dose is by using the morning fasting blood glucose concentration. Therefore, a daily fasting blood glucose measurement is recommended. Although more labor intensive, basal bolus insulin regimens may be appropriate for children and adolescents with type 2 diabetes. The bolus dose is calculated using a correction algorithm for the pre-meal blood glucose concentration and a “carb ratio,” in which 1 U of insulin is given for a certain amount of carbohydrates consumed. In addition, more insulin is given when the blood glucose concentration is above the target level.
For example, if an adolescent has a blood glucose concentration of 250 mg per dL, will eat a meal containing 60 g of carbohydrates, with a carb ratio of 1:10 (1 U of insulin for every 10 g of carbohydrates) and an assigned correction dose of 1:25 > 125 (1 U of insulin for every 25 mg per dL that the glucose level is above the target of 125 mg per dL), the mealtime bolus dose of insulin would be as follows:
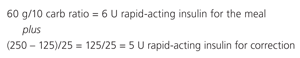
Thus, the total bolus insulin coverage at mealtime would be: 11 U (6 + 5) of rapid-acting insulin.
Clinicians may incorporate the Academy of Nutrition and Dietetics' Pediatric Weight Management Evidence-Based Nutrition Practice Guidelines in their counseling of patients with type 2 diabetes, at the time of diagnosis and as part of ongoing management.
Patients should be referred to a registered dietitian with expertise in the nutritional needs of children and adolescents with type 2 diabetes and advised to follow guidelines from the Academy of Nutrition and Dietetics. These recommendations include a balanced macronutrient diet with lifestyle modifications, nutritional counseling, and caregiver participation.
Clinicians may encourage patients to participate in adequate exercise and limit screen time.
Children and adolescents with type 2 diabetes should participate in moderate to vigorous exercise for at least 60 minutes daily. This may be completed in several, shorter increments (e.g., 10 to 15 minutes) throughout the day. A written exercise prescription that takes into account the patient's physical abilities, preferences, and circumstances may increase adherence. Medication dosages may need to be adjusted when initiating an aggressive physical activity program. Nonacademic screen time should be limited to less than two hours a day.
