Physicians and veterinarians have many opportunities to partner in promoting the well-being of people and their pets, especially by addressing zoonotic diseases that may be transmitted between a pet and a human family member. Common cutaneous pet-acquired zoonoses are dermatophytosis (ringworm) and sarcoptic mange (scabies), which are both readily treated. Toxoplasmosis can be acquired from exposure to cat feces, but appropriate hygienic measures can minimize the risk to pregnant women. Persons who work with animals are at increased risk of acquiring bartonellosis (e.g., cat-scratch disease); control of cat fleas is essential to minimize the risk of these infections. People and their pets share a range of tick-borne diseases, and exposure risk can be minimized with use of tick repellent, prompt tick removal, and appropriate tick control measures for pets. Pets such as reptiles, amphibians, and backyard poultry pose a risk of transmitting Salmonella species and are becoming more popular. Personal hygiene after interacting with these pets is crucial to prevent Salmonella infections. Leptospirosis is more often acquired from wildlife than infected dogs, but at-risk dogs can be protected with vaccination. The clinical history in the primary care office should routinely include questions about pets and occupational or other exposure to pet animals. Control and prevention of zoonoses are best achieved by enhancing communication between physicians and veterinarians to ensure patients know the risks of and how to prevent zoonoses in themselves, their pets, and other people.
Many households in the United States have one or more species of pet animal. The most popular pets are dogs and cats, but rabbits and backyard poultry are increasingly common in domestic settings, and a wide range of reptiles and other “exotic” species are entering our homes. Pets bring enormous social and health-related benefits to people,1 but they can also transmit disease. Physicians and veterinarians have the opportunity to collaborate in their interactions with patients and pets to optimize health for all.
SORT: KEY RECOMMENDATIONS FOR PRACTICE

| Clinical recommendation | Evidence rating | References |
|---|---|---|
| Pets bring enormous social and health-related benefits to their human families. | C | 1 |
| Physicians and veterinarians should take advantage of opportunities to collaborate to improve the health of people and their pets. | C | 5 |
| The clinical history should routinely include questions about whether the family keeps pets or has any other exposure to animals. | C | 5 |
| It is unnecessary for families to rehome pets diagnosed with dermatophytosis (ringworm), scabies, toxoplasmosis, bartonellosis, tick-borne infection, salmonellosis, or leptospirosis if infections are managed appropriately. | C, A for toxoplasmosis | 6–13 |
| Reptiles and amphibians are not recommended pets for children younger than five years because of increased risk of Salmonella infection. | C | 29–31 |
A = consistent, good-quality patient-oriented evidence; B = inconsistent or limited-quality patient-oriented evidence; C = consensus, disease-oriented evidence, usual practice, expert opinion, or case series. For information about the SORT evidence rating system, go to https://www.aafp.org/afpsort.
One area in which physicians and veterinarians are able to readily work together is in addressing infectious diseases that can be transmitted between animals and humans (zoonoses). These diseases can be spread directly, through contact with infected animals; indirectly, through contact with contamination in the animal's environment; or via the vector-borne route (involving transmission by arthropods such as ticks, fleas, flies, or mosquitoes). Often, the clinical disease affects both the person and the pet, but sometimes the pet may appear healthy only to have a subclinical infection or colonization that can lead to illness in a person. Testing of human or animal patients should not be delayed when clinical presentation suggests a zoonosis.
Zoonotic diseases have recently been reviewed2 by the One Health Committee of the World Small Animal Veterinary Association (http://www.wsava.org/educational/one-health-committee), and further information on many of the diseases is available from the Centers for Disease Control and Prevention (http://www.cdc.gov/ncezid/).
The term “One Health” has been used to describe a collaborative effort among veterinarians, physicians, other health care professionals, and scientists to address the well-being of humans, domestic and wild animals, and their shared environment.3,4 In a viable One Health partnership, physicians and veterinarians would be known to each other and communicate professionally for the benefit of the families they serve.5 For example, a veterinarian diagnosing zoonotic disease in the consultation room might inquire if any in-contact family members are unwell and suggest an appointment with their physician's health center, or the veterinarian might offer to make direct contact with the family physician. Similarly, physicians might ask their patients whether they have pets or pet contact, explore whether any presenting symptoms reflect a zoonotic disease, and evaluate the risks that keeping pets may pose for patients who are very young, very old, immunocompromised, or pregnant. The aim of this article is to provide examples of pet-related zoonotic diseases, correct misconceptions about whether some common human diseases are actually zoonotic, and outline the disease risks associated with popular pets. Pets diagnosed with the diseases described in this article do not need to be rehomed if infections are properly managed.6–13
DERMATOPHYTOSIS
Dermatophytosis (ringworm) is caused by a number of skin fungi and can infect both humans and animals14 (Figure 1). Sources of infection include humans, domestic animals, wildlife (primarily rabbits and rodents), and soil. Dogs or cats may show clinical signs or be subclinical carriers of the most common zoonotic species, Microsporum canis, and act as a source of infection for other dogs, cats, or humans.6,15 Some people are more susceptible to dermatophytosis because of genetic factors, preexisting skin disease (e.g., allergic dermatitis), or an immunocompromised state.
Figure 1.
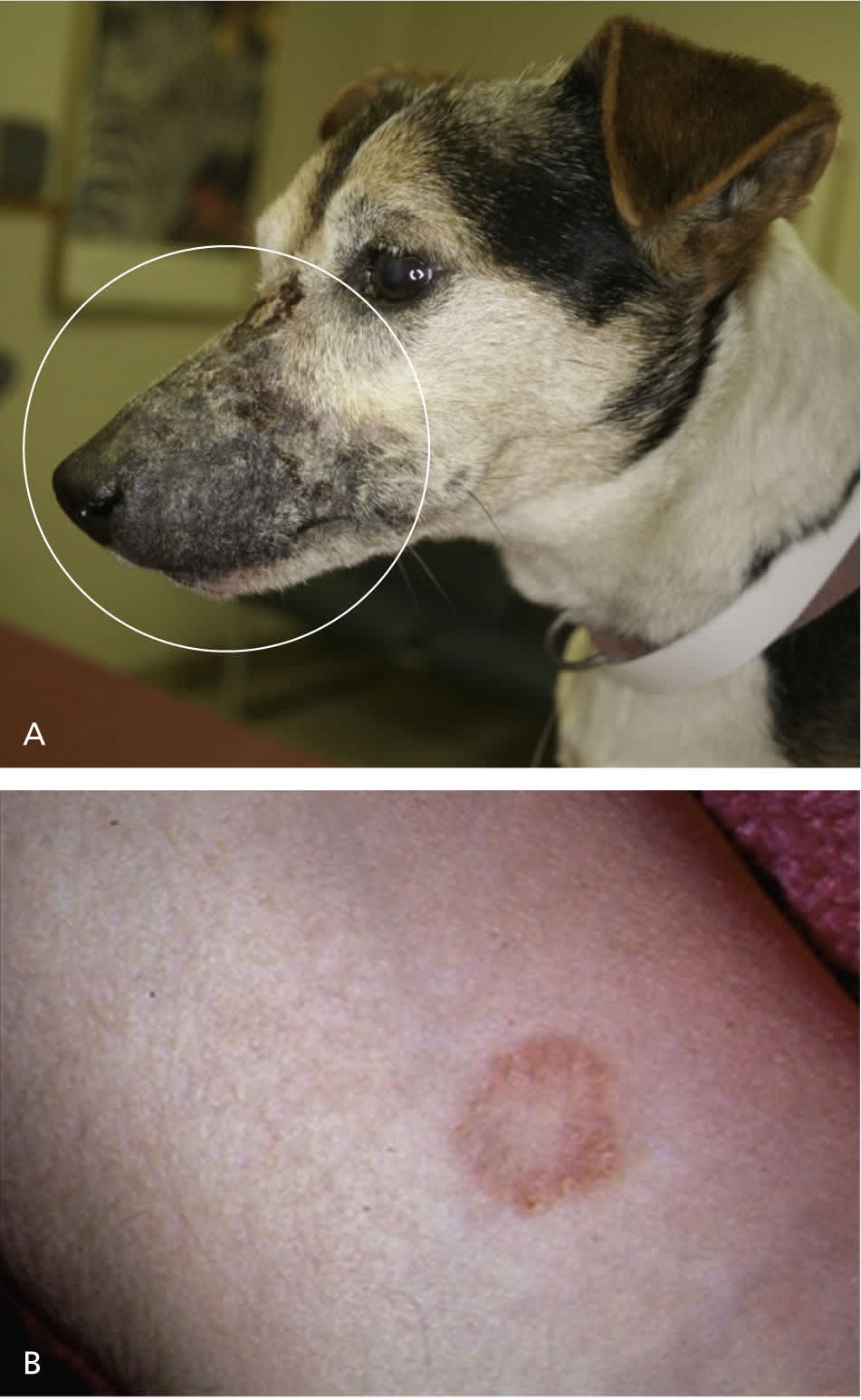
(A) Dermatophytosis in a Jack Russell terrier showing erythema, alopecia, and crusting (circled). (B) Dermatophytosis lesion on the arm of a veterinary student.
Pets with dermatophytosis do not need to be rehomed,15 but physicians should refer pet owners to their veterinarian to develop the best management plan, particularly if any family member is immunocompromised (Table 1).
Table 1. Infections That Can Be Acquired by Direct Contact with Pets

| Infection | Affected pets | Symptoms in humans | Clinical signs in animals | Control measures when diagnosed |
|---|---|---|---|---|
|
| |||
|
|
|
|
|
SCABIES
There is often anguish and some degree of disgust associated with having a dog that has received a scabies diagnosis (sarcoptic mange; Sarcoptes scabiei var canis16; Figure 2). The scabies mite is highly communicable among dogs and may infest humans, but cats are relatively resistant. Persons in contact with an infested dog may be variably affected, and they are only transient hosts for the mites unless there are repeated exposures.17 Mites may persist without a host for two to 21 days, but there is a lesser chance of contracting the infestation from the surrounding environment or fomites than from direct contact with an infested pet.18 If a dog has scabies, it does not need to be rehomed, and there are many effective treatments available from a veterinarian7 (Table 1). The related feline mite Notoedres cati may rarely spread to in-contact humans and cause a pruritic rash, but the infection is self-limited. Scabies in people is more commonly caused by the human itch mite (S. scabiei var hominis), which is transmitted by direct person-to-person contact. The human itch mite does not infect pets.
Figure 2.
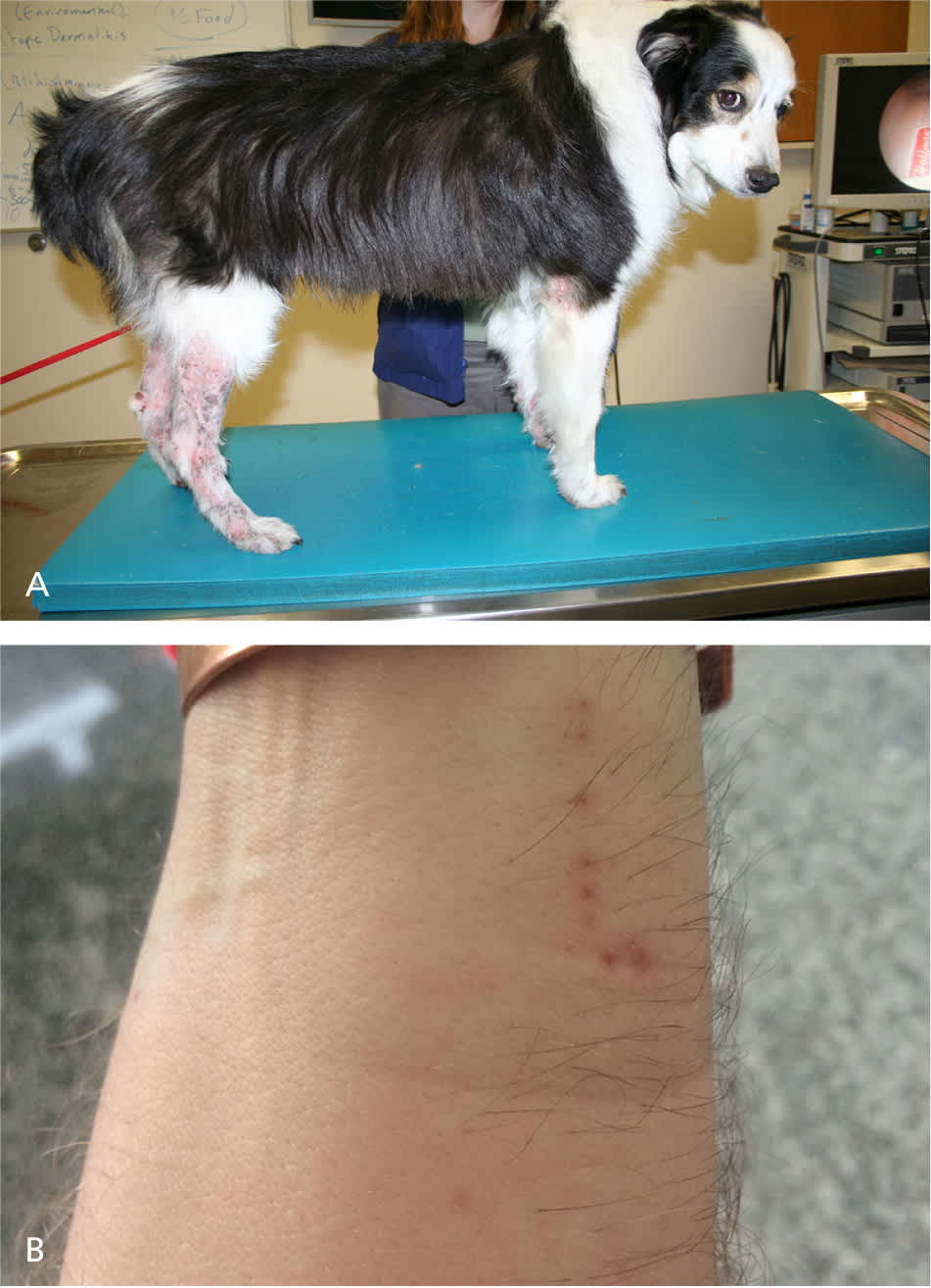
(A) Scabies in a dog showing hair loss, papules, excoriations, and inflammation of the lateral elbows and extremities. (B) Scabies on the arm of a veterinarian. The lesion developed after contact with an infected dog. Note the characteristic linear tract of mite migration.
HEAD LICE
Lice from pets do not infest people, and human lice do not infest pets or other animals. Only humans can acquire and spread human head lice (Pediculus humanus capitis), typically through head-to-head contact as occurs during play at school, home, sports activities, playgrounds, and camps. They are less commonly spread by contact with clothing (e.g., hats, scarves, coats) or other personal items (e.g., combs, brushes, towels).19,20
PINWORM
Human pinworms (Enterobius vermicularis) do not come from pets or other animals, but are intestinal parasitic worms specific to human hosts.21,22 Human pinworm infection can affect persons of all ages and socioeconomic levels, but most commonly occurs in school-aged and preschool-aged children, persons who are institutionalized, and household members and caretakers of patients with pinworm infection. Dogs and cats do not get pinworm infestations, but rabbits may be affected by a species (Passalurus ambiguus) that is not transmissible to humans.23
Which Diseases Could Pet Owners Acquire from Their Pets?
TOXOPLASMOSIS
A common concern for pregnant women is the risk of toxoplasmosis. Because cats are the primary host of Toxoplasma gondii, pregnant women are often concerned about whether they can be infected by their pet cats.
T. gondii is a ubiquitous parasite that completes its life cycle in the intestine of cats. Oocysts that pass in cat feces can contaminate soil, drinking water, and foods such as vegetables, and can initiate infection of humans and other animals if ingested.8 Eating undercooked meat is another common route of infection.24
If T. gondii is acquired during pregnancy and transmitted to the fetus, damage to the fetal central nervous system and eyes can occur. Humans usually do not acquire toxoplasmosis from their pet cats because large numbers of oocysts are shed only once, for less than three weeks, and because most cats groom feces from fur before oocysts become infective.9,25
The application of control measures discussed in Table 2 makes it unnecessary to rehome a pet cat during pregnancy.
Table 2. Pet-Related Infections
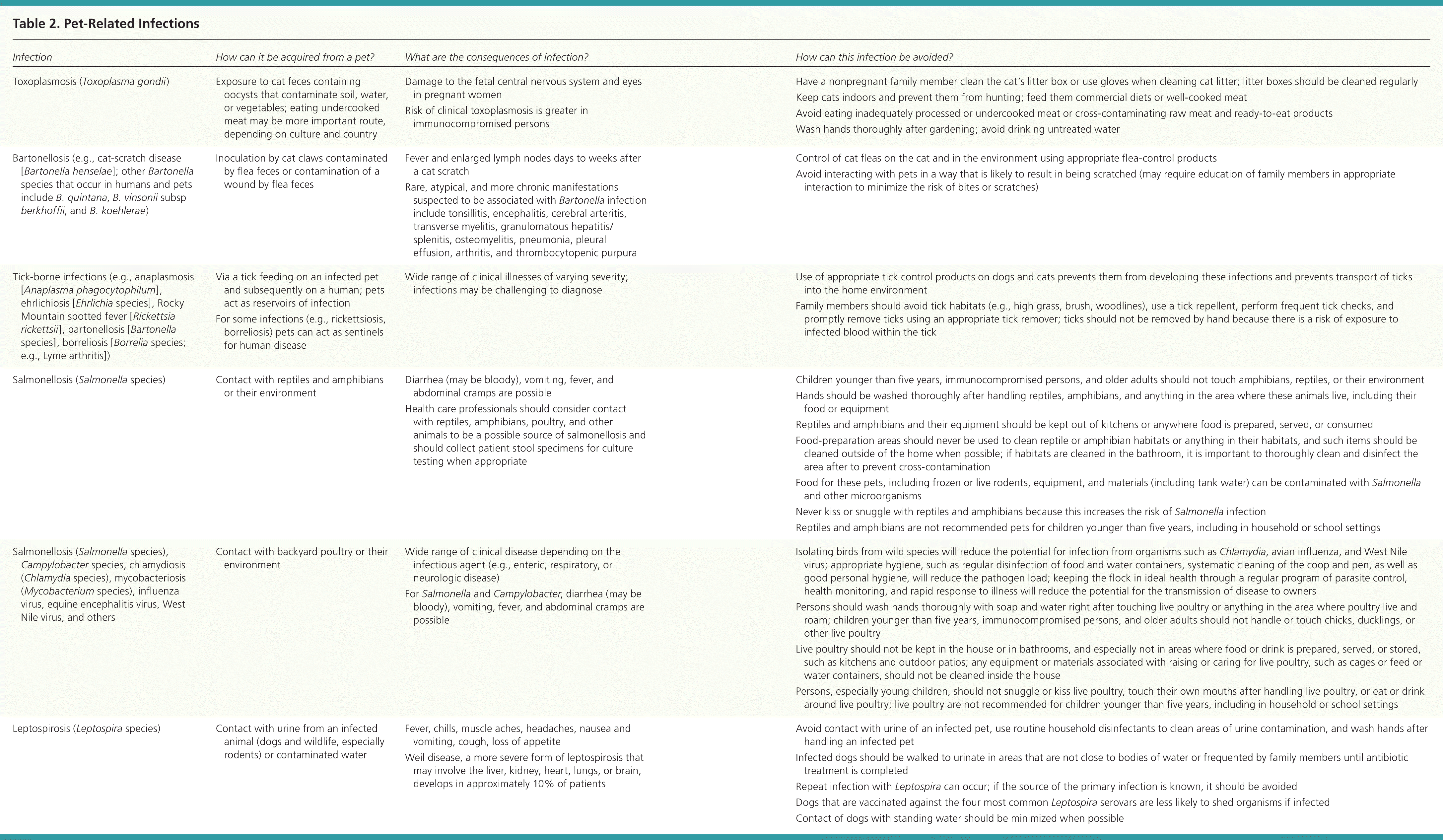
| Infection | How can it be acquired from a pet? | What are the consequences of infection? | How can this infection be avoided? |
|---|---|---|---|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
BARTONELLOSIS
Although other infectious agents can be transmitted via cat scratch, a major concern is transmission of bacteria of one of the Bartonella species. Cat fleas (Ctenocephalides felis) have a role in the transmission of several Bartonella species among cats, dogs, and other small mammals.10 Cats can be chronically bacteremic for months to years after flea transmission, making flea-exposed cats a major reservoir for human infection.
The bacterium can remain viable in flea feces in the environment (i.e., the cat's fur) for at least 15 days. While grooming, flea-infested cats contaminate their claws and saliva with Bartonella-infected flea feces.
Bartonella henselae causes cat-scratch disease in humans (Figure 3). Veterinary workers and persons with occupational animal contact or arthropod exposure are at increased risk of acquiring Bartonella infections.26 It is important for physicians to ask patients about occupational and lifestyle exposure to fleas and cats and to consider the potential role of zoonotic infections in patients with chronic symptoms affecting the joints and the neurologic and vascular systems.27 Prevention of flea infestation of pets is key to preventing these diseases (Table 2).
Figure 3.
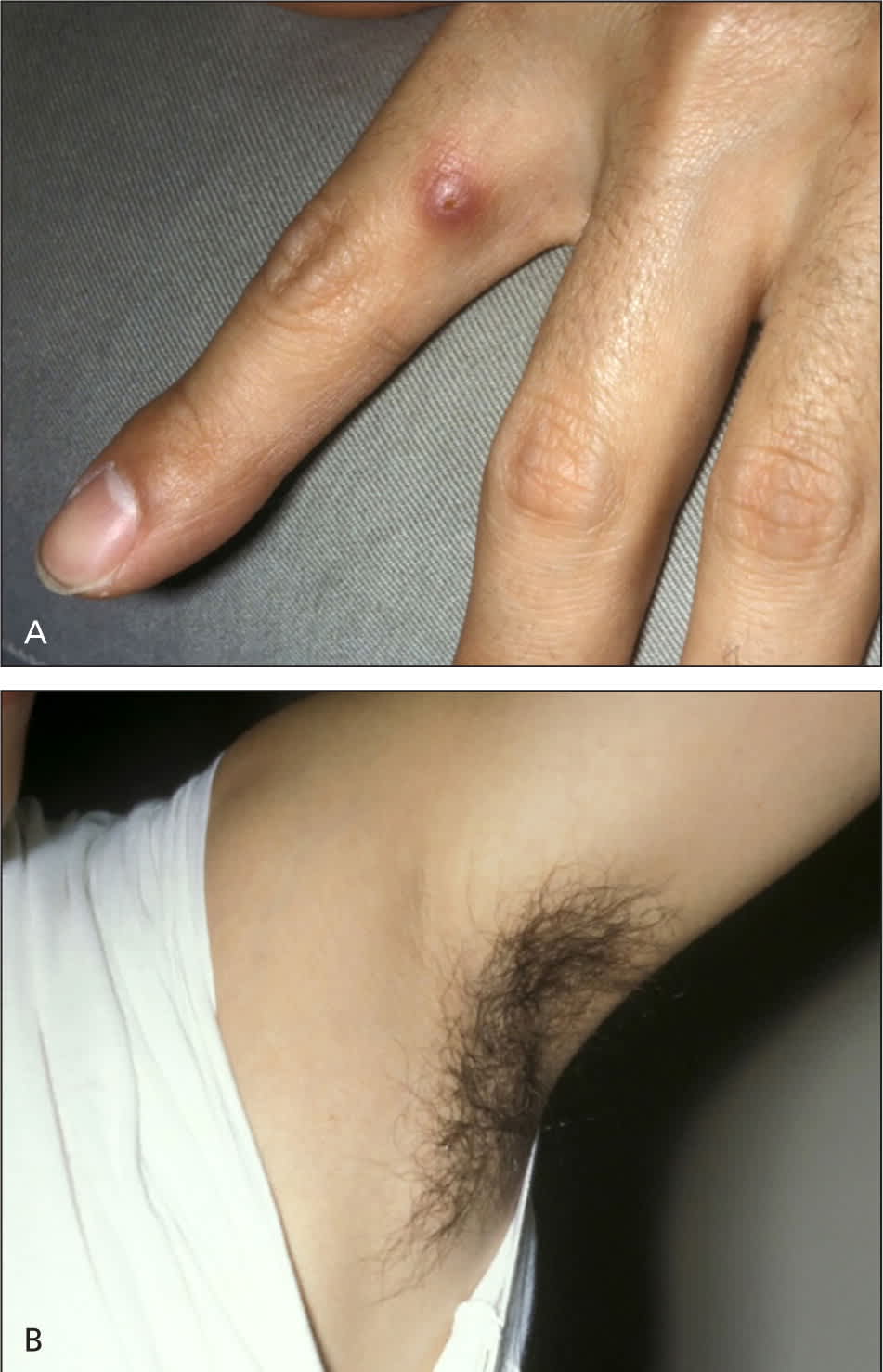
Cat-scratch disease in a human. (A) Papule at the site of cat scratch. (B) Axillary lymphadenopathy.
TICK-BORNE INFECTIONS
Ticks are one of the most important vectors for transmission of infectious agents to people and their pets. Ticks can transmit bacteria, protozoa, rickettsiae, and viruses to pets and humans within the same household.28 In most cases, an infected pet is unlikely to transmit tick-borne infectious agents to humans because pets and humans are both accidental hosts. Transmission of tick-borne agents generally occurs when the tick obtains a blood meal from an animal or human.
Tick species vary in geographic distribution (http://www.capcvet.org/parasite-prevalence-maps/), and different tick species preferentially transmit different pathogens. Pets and humans can be sequentially or simultaneously infested with more than one tick species, and a single tick can transmit more than one organism, leading to coinfection in the tick-infested host.
Pets can be persistently infected (for months to years) with tick-borne zoonotic pathogens and serve as reservoir hosts.11 If a previously infected tick or a tick that acquires an infection from a pet detaches before ingesting a complete blood meal, the tick could subsequently transmit the agent after reattaching to a human. This is a rare event because most attached ticks feed to repletion before detaching, laying eggs, and dying. Pets can also serve as a transport host by carrying ticks from the outdoor environment into the house, where the tick could drop off of the pet and attach to a human. Environmental acquisition of tick-borne disease is more likely than acquisition from a pet. Control measures to prevent tick-borne infection, such as regular use of an ectoparasite control product for pets, are discussed in Table 2.
SALMONELLOSIS FROM REPTILES AND AMPHIBIANS
Reptiles (e.g., turtles, snakes, lizards) and amphibians (e.g., frogs, toads, salamanders) often carry Salmonella bacteria, and the areas where they live can also become contaminated with these bacteria. Contact with contaminated water can lead to illness in humans 29–31 (Table 2).
Children younger than five years, persons who are immunocompromised, and older adults are at a higher risk of illness from Salmonella. Salmonella may spread from the intestine to the bloodstream and then to other body sites, resulting in serious illness or death unless the person is treated promptly with antibiotics. Young children are at increased risk of Salmonella infection because their immune systems are still developing and because they are more likely to put their fingers or other items into their mouths. Therefore, reptiles and amphibians are not recommended pets for children younger than five years.29–31
Salmonellosis should be considered in the differential diagnosis for patients with diarrhea and fever who report a history of reptile or amphibian contact. Physicians and other clinicians should refer patients who own reptiles or amphibians to their veterinarian for advice on appropriate husbandry that will minimize the risk of salmonellosis.
DISEASE RISKS FROM BACKYARD POULTRY
An increasing number of persons keep poultry (e.g., chickens, ducks) as part of a greener, healthier lifestyle or for economic or cultural reasons. Although they provide benefits, backyard poultry carry the risk of illness, especially for children. Disease can result from handling these birds or anything in the areas where they live and roam. Among the birds kept as pets, chickens pose one of the greatest risks of transmitting disease to their owners compared with other bird species, such as parakeets or canaries.32,33 Salmonellosis is the most common zoonotic disease associated with owning backyard chickens.34 Live poultry infected with Salmonella typically appear healthy, but they shed the bacteria intermittently. Other zoonoses that may be spread between humans and poultry are listed in Table 2.
Appropriate preventive measures should be taken to minimize disease transmission to humans35 (Table 2). Physicians should be alert to the possibility of zoonotic disease introduced by a flock of backyard chickens and educate patients on the risks when applicable.
LEPTOSPIROSIS
Multiple Leptospira species can infect dogs, cats, or humans.36 Prevalence rates vary by geographic region, and numerous wildlife reservoirs exist for the bacteria. The organisms are shed in urine, and infection is initiated after exposure to contaminated water. Alternatively, Leptospira infections can occur by direct penetration of intact skin following contact with infected urine, blood, or tissue. Clinical manifestations vary depending on the bacterial strain and mammalian species infected (Table 2).
Infections in the owners of dogs that have been diagnosed with leptospirosis and are undergoing treatment are uncommon, and human leptospirosis is most often acquired from environmental or wildlife exposure.12,13 However, the owner should consult his or her physician and veterinarian for advice about minimizing the risk of infection, particularly if a family member is immunocompromised. Persons who work with animals have a greater risk of contracting leptospirosis.
Data Sources: A literature search was conducted using the Thompson Reuters Web of Science and the search terms zoonosis and dog, zoonosis and cat, zoonosis and reptile, and zoonosis and poultry. Additionally, the individual disease-related sections of the manuscript were critically reviewed by eight internationally recognized specialists (named below) who also recommended relevant reference material. Search date: February 1, 2016.
This article was produced by members of the WSAVA One Health Committee and invited experts. The particular contributions of the following are gratefully acknowledged: Dr. Casey Barton Behravesh (One Health Office, Centers for Disease Control and Prevention, Atlanta, Ga.; head lice, pinworm, salmonellosis, and diseases from backyard poultry), Dr. Ed Breitschwerdt (College of Veterinary Medicine, North Carolina State University, Raleigh; bartonellosis and tick-borne diseases), Dr. Don Harris (Avian and Exotic Medical Center, Miami, Fla.; diseases from backyard poultry), Dr. Peter Karczmar (Coastal Pulmonary and Internal Medicine Associates, East Providence, R.I.; toxoplasmosis), Dr. Mike Lappin (College of Veterinary Medicine and Biomedical Sciences, Colorado State University, Fort Collins; toxoplasmosis and leptospirosis), Dr. Jennifer Schissler (College of Veterinary Medicine and Biomedical Sciences, Colorado State University, Fort Collins; ringworm), Dr. Rod Rosychuk (College of Veterinary Medicine and Biomedical Sciences, Colorado State University, Fort Collins; scabies), and Dr. George Moore (College of Veterinary Medicine, Purdue University, West Lafayette, Ind.; leptospirosis).

