Patients with an inflammatory bowel disease, such as ulcerative colitis or Crohn's disease, have recurrent symptoms with considerable morbidity. Patient involvement and education are necessary components of effective management. Mild disease requires only symptomatic relief and dietary manipulation. Mild to moderate disease can be managed with 5-aminosalicylic acid compounds, including olsalazine and mesalamine. Mesalamine enemas and suppositories are useful in treating proctosigmoiditis. Antibiotics such as metronidazole may be required in patients with Crohn's disease. Corticosteroids are beneficial in patients with more severe symptoms, but side effects limit their use, particularly for chronic therapy. Immunosuppressant therapy may be considered in patients with refractory disease that is not amenable to surgery. Inflammatory bowel disease in pregnant women can be managed with 5-aminosalicylic acid compounds and corticosteroids. Since longstanding inflammatory bowel disease (especially ulcerative colitis) is associated with an increased risk of colon cancer, periodic colonoscopy is warranted.
Inflammatory bowel disease encompasses a number of chronic, relapsing inflammatory disorders involving the gastrointestinal tract.1 The prevalence of these disorders ranges from 20 to 200 per 100,000 U.S. population.2 It is estimated that more than 600,000 people in the United States have some form of inflammatory bowel disease.3 Classically, inflammatory bowel disease includes ulcerative colitis and Crohn's disease.
Ulcerative colitis typically presents with shallow, continuous inflammation extending from the rectum proximally to include, in many patients, the entire colon. Fistulas, fissures, abscesses and small-bowel involvement are absent. Patients with limited disease (e.g., proctitis) typically have mild but frequently recurrent symptoms, while patients with pancolitis more commonly have severe symptoms, often requiring hospitalization.
Unlike ulcerative colitis, Crohn's disease can involve the entire intestinal tract, from the mouth to the anus, with discontinuous focal ulceration, fistula formation and perianal involvement. The terminal ileum is most commonly affected, usually with variable degrees of colonic involvement. Subsets of patients have perianal disease with fissures and fistula formation. Only 2 to 3 percent of patients with Crohn's disease have clinically significant involvement of the upper gastrointestinal tract.4
The pathogenesis of inflammatory bowel disease is not known. A genetic predisposition has been suggested, and a host of environmental factors, including bacterial, viral and, perhaps, dietary antigens, can trigger an ongoing enteric inflammatory cascade. Although ulcerative colitis and Crohn's disease have multiple subtle immunologic differences, no test can yet reliably distinguish between them.2 Approximately 5 to 15 percent of patients have “indeterminate” colitis, a diagnosis that is given when neither Crohn's disease nor ulcerative colitis can be reliably diagnosed based on clinical and pathologic features.5
General Principals of Management
Management efforts in inflammatory bowel disease are directed at decreasing or eliminating symptoms, thereby improving the patient's quality of life. Accomplishing these goals requires careful selection of therapeutic agents based on symptom severity and drug side effects (Figures 1 and 2).
FIGURE 1.
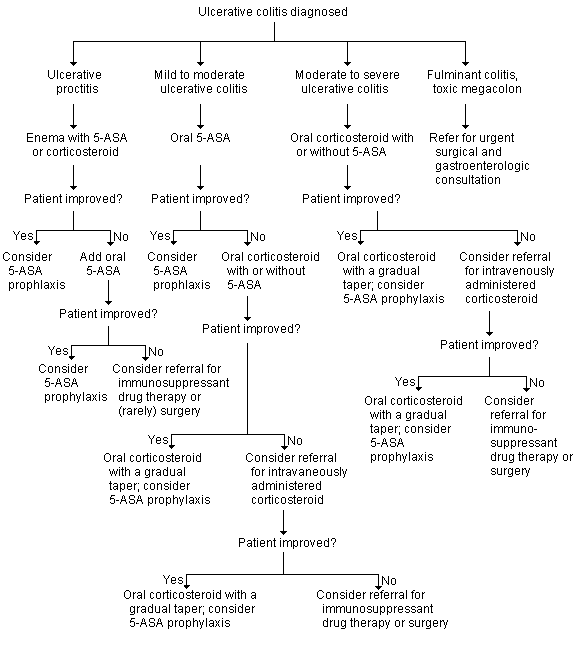
Guidelines for the management of ulcerative colitis, which is diagnosed by clinical evaluation, colonoscopy, barium enema, flexible sigmoidoscopy, laboratory tests and stool studies. Treatment of this illness can require numerous adjustments of therapy, depending on the situation and the individual patient. A close working relationship with a gastroenterologist and/or surgeon is essential (5-ASA = 5-acetylsalicylic acid [mesalamine; Asacol, Pentasa, Rowasa]).
FIGURE 2.
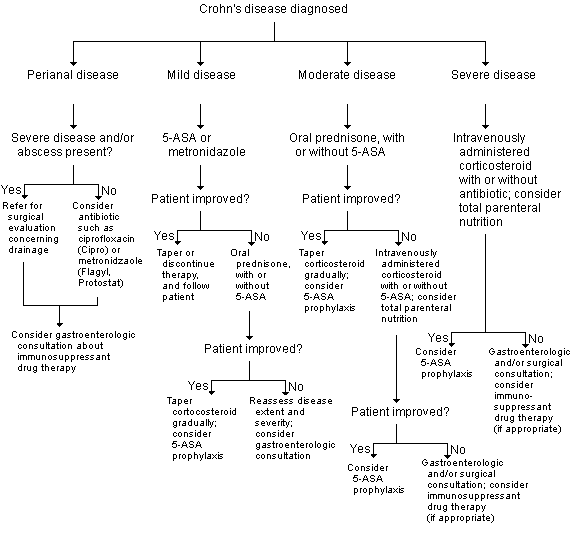
Guidelines for the management of Crohn's disease, which is diagnosed by clinical evaluation, radiographic studies, endoscopy, laboratory tests and stool studies. Treatment of this illness can require numerous adjustments, depending on the situation and the individual patient. A close working relationship with a gastroenterologist and/or surgeon is essential (5-ASA = 5-acetylsalicylic acid [mesalamine; Asacol, Pentasa, Rowasa]).
Endoscopic and radiologic studies are used to diagnose the particular disorder and to determine the extent of the disease. Once the diagnosis has been confirmed, the patient and the physician work together to establish realistic treatment goals. Therapy is then individualized to meet these goals.
When symptoms recur, a careful history, a thorough physical examination and confirmatory laboratory studies are required to make a cost-effective reassessment. Extensive radiologic and endoscopic studies for restaging should be performed only when a major clinical change occurs or a major treatment change is contemplated (e.g., surgery or the addition of corticosteroid or immunosuppressant drug therapy).
Since inflammatory bowel disease is a chronic illness with a frequent, often unpredictable impact on a patient's life, management generally requires much more than the treatment of symptoms. Patient education is crucial. (Sources of information and patient support are provided in the patient information handout at the end of this article.)
Coexisting Problems and Inciting Factors
A landmark study6 of 615 patients with at least a 96 percent seven-year follow-up rate found that 40 to 60 percent of patients with inflammatory bowel disease described their quality of life as fair to poor, despite excellent medical and surgical management. Efforts are under way to develop and standardize a quality of life index for assessing therapeutic efficacy in clinical trials.7
Coexisting problems have an impact on the prognosis and quality of life for patients with inflammatory bowel disease. As with other chronic illnesses, depression is a complicating factor in 30 to 50 percent of patients with severe disease, especially Crohn's disease.8 Up to 30 percent of the depressed patients can develop dependency on prescription narcotics, benzodiazepines and/or alcohol.9 Thus, when coexisting disorders are not treated, patients do poorly regardless of the medical care they receive for inflammatory bowel disease.
Certain lifestyle factors also play a role in inflammatory bowel disease. Smoking may be associated with an increased risk of Crohn's disease, especially postoperative recurrence.10 In contrast, smoking is associated with a decreased risk of ulcerative colitis, and nicotine patches apparently have some benefit in patients with active disease. However, nicotine patches do not help maintain remission of ulcerative colitis.11
Treatment of Symptoms
Loperamide (Imodium) increases both colonic water absorption and internal sphincter function. It can be helpful as adjunctive therapy in patients who have proctitis and diarrhea, patients who have chronic watery diarrhea without active disease and patients who have undergone ileoanal pullthrough surgery. Loperamide should not be used in patients with active pancolitis, bloody diarrhea or suspected enteric infection.
Cholestyramine (Questran) is helpful in patients who develop bile-salt–induced diarrhea after ileal resection. Since this drug binds to many other agents and reduces their bioavailability, it should be used cautiously as long-term therapy. Cholestyramine can markedly worsen steatorrhea in patients with more extensive ileal resection.
Dietary and Nutritional Factors
Lactose malabsorption is present in 35 percent of patients with Crohn's disease12 and 20 percent of patients with ulcerative colitis.13 Since the symptoms of lactose malabsorption can be confused with those of active inflammatory bowel disease, the presence of this digestive disorder should be elicited by the history and, if necessary, appropriate laboratory tests.
Lactose has no effect on the activity of inflammatory bowel disease. Thus, lactose consumption does not need to be prohibited in all patients with this diagnosis. Alternative calcium supplementation should be used in patients who are on a lactose-restricted diet.
Low-fiber diets do not alter the course of inflammatory bowel disease. However, these diets can be helpful in patients with active symptoms, stenoses and/or obstructive symptoms.
Vitamin deficiencies, especially vitamin B12 deficiency, may occur in patients with Crohn's disease or previous ileal resection. Iron deficiency is also relatively common in patients with Crohn's disease. Folate deficiency can occur as a result of poor dietary intake or the interference of sulfasalazine (Azulfidine) with folate metabolism. Occasionally, inflammatory bowel disease can present with an anemia that does not respond to iron replacement therapy. Recently, concomitant administration of synthetic erythropoietin has been shown to be beneficial in treating this refractory anemia.14
In patients with active colitis, significant protein calorie malnutrition with low serum protein and albumin levels is indicative of severe disease that may require hospitalization and intravenous hyperalimentation therapy (total parenteral nutrition). Chronic severe malnutrition in patients with short-bowel syndrome due to massive surgical resection of the small intestines is an indication for home intravenous hyperalimentation therapy.
Enteral hyperalimentation with elemental or oligomeric formulas has been used as primary therapy for active Crohn's disease, especially in Europe. Comparative randomized trials have shown that this nutritional therapy is as effective as or slightly less effective than corticosteroid therapy.15 However, enteral diets tend to be unpalatable and expensive, factors that have limited their use.
5-Aminosalicylic Acid Compounds
A number of products use various means of delivering mesalamine, or 5-aminosalicylic acid (5-ASA; Asacol, Pentasa, Rowasa), a topically active anti-inflammatory agent, to inflamed intestinal mucosa16,17 (Table 1). The newer 5-ASA agents are the drugs of choice in patients with inflammatory bowel disease who are allergic to sulfa. Side effects associated with the 5-ASA agents include interstitial nephritis, pleuropericarditis, leukopenia and pancreatitis. Fortunately, these side effects are quite rare.
TABLE 1 Some Medical Therapies for Inflammatory Bowel Disease
| Medication | Colon activity | Small-bowel activity | Dosage range* | Approximate cost per month (generic)† | Relative side effect risk‡ | |
|---|---|---|---|---|---|---|
| 5-Acetylsalicylic acid compounds | ||||||
| Sulfasalazine (Azulfidine) | +++ | + | 2 to 4 g per day | $49 (23-26) for 180 500-mg tablets | Intermediate | |
| Olsalazine (Dipentum) | +++ | + | 1 to 3 g per day | $124 for 180 250-mg tablets | Low to intermediate | |
| Mesalamine (Pentasa) | ++ | ++ | 2 to 4 g per day | $105 for 180 250-mg capsules | Low | |
| Mesalamine (Asacol) | +++ | + | 1.6 to 4.8 g per day | $111 for 180 400-mg tablets | Low | |
| Mesalamine enema (Rowasa) | +++ | 4 g per day | $93 for 30 4-g enemas | Very low | ||
| Metronidazole (generic) | ++ | ++ | 750 to 1,500 mg per day | $22-26 for 120 250-mg tablets | Intermediate | |
| Prednisone (generic) | +++ | +++ | 10 to 50 mg per day | $5-6 for 100 10-mg tablets | Very high | |
| Corticosteroid enema (Cortenema) | +++ | One enema (100 mg) per day | $229 for 30 100-mg enemas | Low (but avoid long-term use) | ||
| Mercaptopurine (Purinethol) | +++ | +++ | 50 to 100 mg per day | $61 for 50 50-mg tablets | High | |
| Methotrexate (Rheumatrex) | +++ | +++ | 15 to 25 mg per week | $289 (225–262) for 75 2.5-mg tablets | High | |
| $14 for 2 50-mg vials | ||||||
+ = Little activity; ++ = some activity; +++ = strong activity.
*—Relative range of dosages most commonly used, presented for comparative purposes only.
†—Using an arbitrary “middle dose,” estimated cost to the pharmacist is based on average wholesale prices, rounded to the nearest dollar, in Red book. Montvale, N.J.: Medical Economics Data, 1997. Cost to the patient will be higher, depending on prescription filling fee.
‡—Arbitrary scale of risk for long-term use in middle or high doses (developed by the authors of this article).
Sulfasalazine
Sulfasalazine has been in use for more than 30 years. The oldest and least expensive of the 5-ASA compounds, sulfasalazine is composed of sulfapyridine and 5-ASA molecules. Bacteria in the terminal ileum and colon cleave these molecules into sulfapyridine and the topically active 5-ASA. Sulfapyridine is responsible for the sulfa-related adverse effects of sulfasalazine.
Common side effects of sulfasalazine include nausea and headache, which can be avoided by increasing the dosage slowly. Rash, hemolytic anemia and hepatic toxicity are caused by sulfapyridine. If these side effects occur, sulfasalazine should be discontinued immediately. Sulfapyridine can also cause decreased sperm function, a factor that must be considered in patients who are trying to conceive.
Sulfasalazine in a low dosage (e.g., 1 g twice daily) can be used to maintain remission in patients with ulcerative colitis. In a higher dosage (4 to 6 g per day), the drug can be used to treat active ulcerative colitis.
Olsalazine
Olsalazine (Dipentum) delivers intact 5-ASA to the terminal ileum by binding two 5-ASA molecules with a diazo bond, which is cleaved by bacteria. The diazo bond is responsible for ileal secretory diarrhea, a unique side effect of olsalazine therapy. Ileal secretory diarrhea occurs in 5 to 10 percent of patients who receive olsalazine.2 Rarely, the diarrhea can be quite severe.
This drug has been shown to benefit patients with active ulcerative colitis, and it can help maintain disease remission. Little information is available on the use of olsalazine in patients with Crohn's disease, but it is reasonable to assume that the drug is also effective in these patients.
Mesalamine
In one brand of mesalamine tablets (Pentasa), 5-ASA is packaged in ethylcellulose microgranules that are gradually released from the jejunum to the colon. This drug has been found to be superior to placebo in the treatment of active Crohn's disease, as well as in maintaining remission of Crohn's disease (something that has not been well demonstrated with sulfasalazine).18 A dosage of 4 g per day has the most benefit, but it requires taking 16 tablets per day. A dosage of 2 to 3 g per day is effective in patients with active ulcerative colitis. In a dosage of 1 to 2 g per day, mesalamine is equal to sulfasalazine in maintaining remission of ulcerative colitis.19
In another brand of mesalamine tablets (Asacol), the drug is enveloped in a pH-sensitive coating that delivers the drug to the distal ileum and colon. Most of the studies of this mesalamine tablet have been conducted in patients with ulcerative colitis. Dosages ranging from 2.4 to 4.6 g per day have been beneficial in these patients.20 This tablet has been found to be as effective as sulfasalazine in maintaining remission of ulcerative colitis.21 It has also been beneficial in the treatment of active Crohn's disease and, in a dosage of 2.4 g per day, in maintaining remission.22 In enema or suppository form, mesalamine (Rowasa) is the most effective therapy for distal proctosigmoiditis, but it has little efficacy in anorectal Crohn's disease. In proctitis, mesalamine enemas and suppositories are more effective than oral 5-ASA and slightly more effective than corticosteroid enemas or even oral prednisone.23 They work best when they are taken in the evening and retained all night. Although many patients improve after a few days of treatment, full benefit may not be achieved for 12 weeks.
Mesalamine enemas cost considerably more than oral medications. Suppositories are easier to retain in patients with short distal disease. Little or no systemic 5-ASA absorption occurs with mesalamine enemas or suppositories and, as would be expected, they have virtually no side effects.
Antibiotics
Experimental and clinical evidence suggests that bacterial flora play an important role (perhaps as an antigenic substrate) in the pathogenesis of inflammatory bowel disease. Virtually all studies showing a benefit for antibiotic therapy have been performed in patients with Crohn's disease.24
Metronidazole (Flagyl, Protostat) has been the best studied antibiotic. This drug is effective in patients with active Crohn's disease and has a potency similar to that of sulfasalazine. Metronidazole is particularly effective in patients who have perianal Crohn's disease, with the benefits improving as the dosage is increased, up to a maximum of 2 g per day.
In chronic metronidazole therapy, dosages higher than 1 g per day can be associated with irreversible peripheral neuropathy. Gastrointestinal side effects also limit the use of higher doses. Relapse is common when metronidazole is discontinued.
Ciprofloxacin (Cipro) is also effective in patients with Crohn's disease, and it can be combined with metronidazole.
Corticosteroids
Systemic Corticosteroids
For the past 30 years, corticosteroids have been the mainstay of therapy in patients with moderate to severe active inflammatory bowel disease.25 Initial treatment is prednisone, 40 to 60 mg per day. In severely ill hospitalized patients, reasonable initial therapy is hydrocortisone, 100 mg administered intravenously every eight hours. Intravenous therapy generally produces rapid improvement of symptoms, with maximal benefit occurring when the corticosteroid has been administered for six to eight days.
Once improvement has occurred, prednisone is tapered by 5 to 10 mg per week until the dosage is 15 to 20 mg per day. This dosage is then tapered by 2.5 to 5 mg per week until the drug is discontinued. Confusion over the taper schedule can be avoided if patients are given written instructions and 5-mg tablets are used. The goal is to get patients off corticosteroids within a relatively short time but still maintain disease remission. Concomitant use of 5-ASA agents can be helpful. Alternatively, long-term alternate-day corticosteroid therapy can be used in patients with refractory Crohn's disease, although it may be necessary to use dosages of 20 to 25 mg every other day.26
Systemic corticosteroids have an extensive side effect profile. Acute effects include acne and severe mood changes, which are particularly common in young patients. Adrenal insufficiency can be triggered by an intercurrent infection in patients who are receiving low doses or in patients who have just been tapered off of corticosteroids. Visual changes can occur because of steroid-induced hyperglycemia. Early cataract formation is another possible problem. Aseptic joint necrosis, the most dreaded side effect, usually occurs in patients receiving long-term, high-dose corticosteroid therapy. The incidence of this complication is 4.3 percent.27
Topical Corticosteroids
Corticosteroid enemas are beneficial in patients with ulcerative proctosigmoiditis. The foam preparations may facilitate retention and thus may be more effective than the liquid preparations. Both foam and liquid corticosteroid enemas are slightly less effective than 5-ASA enemas and are almost as expensive. Some systemic absorption occurs; adrenal suppression and other corticosteroid side effects rarely occur with long-term use.
Newer Corticosteroids
Budesonide, tixocortol pivolate and fluticasone propionate are among the newer corticosteroid analogs currently under investigation. None of these agents has as yet been approved by the U.S. Food and Drug Administration.
These newer corticosteroids are more rapidly metabolized than traditional corticosteroids, and they offer the promise of efficacy with fewer systemic side effects. The packaging of these agents in a pH-sensitive coating (similar to that used for 5-ASA preparations) offers the possibility of drug delivery to the small bowel and right colon with a minimum of side effects.
Two large prospective studies have shown that budesonide therapy is beneficial in patients with active Crohn's disease.28 Studies are being conducted to determine the effectiveness of budesonide in treating ulcerative colitis and maintaining remission of Crohn's disease. Over time it will become clearer if budesonide and other newer corticosteroids truly provide therapeutic benefits with fewer side effects.29
Immunosuppressant Drugs
Immunosuppressant drugs can be an invaluable adjunct in the treatment of patients with intractable inflammatory bowel disease or complex, inoperable perianal disease.30 Although immunosuppressant agents have significant side effects, they are safer and better tolerated than long-term corticosteroid therapy. However, these agents should not be used in young patients who are candidates for surgery or in patients who are noncompliant and refuse to return for periodic monitoring.
Before immunosuppressant therapy is initiated, side effects and other treatment alternatives should be discussed with the patient. Then it is best to set a definable goal, such as closure of a fistula or tapering from corticosteroids, and a minimum three-month time frame for reaching that goal. The use of immunosuppressant drug therapy requires close communication between patient and physician.
Azathioprine and Mercaptopurine
Since the early 1970s, azathioprine (Imuran) and mercaptopurine (Purinethol) have been used to treat inflammatory bowel disease. These drugs are superior to placebo, but their full effects may not become apparent for as long as three months.31 Azathioprine and mercaptopurine are beneficial in 50 to 70 percent of patients with intractable perianal Crohn's disease.32 Less information is available about their effectiveness in treating ulcerative colitis, although they have been beneficial in patients with this disease.
For both azathioprine and mercaptopurine, the initial dosage is 50 mg per day. Therapeutic benefit usually occurs at dosages of 50 to 100 mg per day for mercaptopurine and 75 to 150 mg per day for azathioprine. Mild leukopenia suggests drug effect and therefore an increased likelihood of benefit. It is prudent to obtain a complete blood count every two weeks during the initial treatment phase in patients with active disease and every three months in patients on maintenance therapy.
Drug-induced pancreatitis occurs in 3 to 5 percent of patients, invariably during the first six weeks of azathioprine or mercaptopurine therapy.33 Pancreatitis is a contraindication to continued use of these agents. One large, retrospective review34 failed to find a significant association between azathioprine and the development of lymphoma or leukemia.
Methotrexate
For more than 20 years, low-dose methotrexate (Rheumatrex) therapy has been used in patients with intractable psoriasis and rheumatoid arthritis. More recently, low doses of this agent have also been given to patients with asthma. One study3 showed that this treatment was beneficial in 70 percent of patients with severe inflammatory bowel disease. The response to methotrexate appeared to be more rapid than the response to mercaptopurine. A recently published multicenter, placebo-controlled trial35 confirmed the significant benefit of low-dose methotrexate therapy in patients with active Crohn's disease.
Methotrexate is given weekly as an intramuscular injection of 15 to 25 mg. Side effects are rare and include leukopenia and hypersensitivity interstitial pneumonitis. Hepatic fibrosis is the most severe potential sequela of long-term therapy. Patients with concomitant alcohol abuse and/or morbid obesity are more likely to develop hepatic fibrosis and therefore should not be treated with methotrexate. It is prudent to obtain a baseline chest radiograph and to monitor complete blood count, liver function and renal function every two weeks until the patient is receiving oral therapy, and every one to three months thereafter. Before methotrexate therapy is initiated, the risks of treatment and the possible need for a liver biopsy should be discussed with the patient.
A pretreatment liver biopsy is indicated in patients who have abnormal liver function tests and in those at potentially increased risk for hepatic toxicity. Follow-up liver function tests are not a good predictor of toxicity. Therefore, it is prudent to perform a liver biopsy after a patient has received a cumulative methotrexate dosage of 1.5 g and periodically thereafter.
Cyclosporine
Cyclosporine (Sandimmune) is a potent immunosuppressive drug used in organ transplantation. Since the mid-1980s, this drug has also been used to treat patients with inflammatory bowel disease. At this time, cyclosporine is most useful in severely ill patients with ulcerative colitis who have not responded to corticosteroid therapy.36 In such patients, intravenously administered cyclosporine is highly effective for rapid disease control, and it may allow patients to avoid surgery. However, by one year, 70 to 80 percent of these patients may still require surgery.37 Thus, in many patients, the role of cyclosporine is to change a risky emergency operation into a less urgent procedure.
Cyclosporine is lipid-bound and thus is associated with an increased risk of seizures when it is administered to acutely ill, severely malnourished patients who have low serum cholesterol/lipid levels. Oral maintenance with cyclosporine has, at best, limited benefit, and the relapse rate is high. The drug has a significant side effect profile that includes renal insufficiency and hypertension.
Future Developments
Leukotriene synthetase inhibitors, which are currently in phase III trials, act on an important intestinal inflammatory pathway. One of these inhibitors, zileuton (Zyflo), has been shown to be effective in inflammatory bowel disease. The FDA has approved the use of this drug for the treatment of asthma. FDA approval for its use in inflammatory bowel disease is pending.38 This leukotriene synthetase inhibitor is not a “breakthrough” drug, and its demonstrated efficacy is similar to that of the 5-ASA preparations.
Other new treatments include monoclonal antibodies to tumor necrosis factor and several inflammatory cytokines. In one small European series,39 infusion of the antitumor necrosis factor chimeric monoclonal antibody (cA2) every four to six weeks resulted in a dramatic resolution of Crohn's disease. This treatment approach is currently being studied in the United States. The drug carries a risk of acute, potentially severe allergic reactions during the infusion. In addition, its long-term effects on the immune system are unknown.
Interleukin 10 and interleukin 11 are cytokines with immunosuppressive and anti-inflammatory activities that, in preliminary studies, have been found to be of benefit in Crohn's disease.40,41 In one recent preliminary study,42 tacrolimus (FK506; Prograf), an immunosuppressant similar to cyclosporine but with better oral absorption, was shown to be beneficial in patients with complex proximal small-bowel Crohn's disease.
Inflammatory Bowel Disease in Pregnancy
Women with inflammatory bowel disease are no longer advised not to have children. Pregnancy is usually uneventful in patients with quiescent inflammatory bowel disease. Patients with active disease are more likely to have a miscarriage, to deliver prematurely or to have an infant with below-normal birth weight. However, medical management results in a satisfactory outcome in most of these pregnancies.43
For obvious reasons, radiologic studies should be avoided in pregnant women. If possible, flexible sigmoidoscopy should also be avoided, because it may stimulate premature labor.
In most patients, the risks to the newborn from untreated disease are much greater than the risks associated with medical therapy. For many years, corticosteroids and sulfasalazine have been used safely in pregnant women with active inflammatory bowel disease. The sulfapyridine moiety of sulfasalazine is tightly bound to serum proteins and therefore does not appear to increase the risk of kernicterus. A recent review44 of 5-ASA compounds in pregnancy suggests that they are also safe.
Metronidazole has been shown to be potentially teratogenic in animal studies. However, a recent meta-analysis of short-term metronidazole therapy (seven to 10 days) in pregnant women with Trichomonas infection suggested that the drug can be used in the first trimester without an increased risk of teratogenicity.45 Because information on long-term effects is lacking, it is prudent to avoid prolonged metronidazole therapy in pregnancy.
If possible, immunosuppressant drugs should not be given to pregnant women. However, azathioprine and mercaptopurine do not appear to increase the risk of congenital malformations in pregnant patients with severe inflammatory bowel disease.46–48 Methotrexate probably should not be used in pregnant women with inflammatory bowel disease, since little is known about the effects of the drug in pregnancy.
Cancer Screening
Ulcerative colitis is associated with an increased risk of colon cancer. An epidemiologic study49 showed no increased risk in patients with proctitis. Compared with normal individuals, however, patients with left-sided colitis were 2.8 times more likely to develop colon cancer, and patients with pancolitis were 15 times more likely to develop this cancer. In patients with pancolitis beginning in childhood, the risk of colon cancer was 162 times higher than the risk in persons without inflammatory bowel disease. Colonoscopy with multiple biopsies for dysplasia can detect cancer early and therefore decrease the risk of mortality. Unfortunately, this surveillance approach is very expensive, and noncompliance is a significant problem.
The risk of malignancy is related to the extent and duration of disease, not its activity. This is especially important in patients with burned-out, quiescent disease who may be reluctant to undergo screening. While dysplasia is a harbinger of malignancy, it is difficult to diagnose in the setting of active inflammatory bowel disease. If possible, patients should be screened for colon cancer at a time when their disease is in remission.
The frequency of colon cancer screening in patients with inflammatory bowel disease remains a subject of debate. Instead of performing yearly colonoscopy after inflammatory bowel disease has been present for seven to 10 years, it may be more effective to tailor screening frequency to the cumulative cancer risk.50 In patients who have had pancolitis or left-sided colitis for seven to 10 years, it is reasonable to perform a baseline surveillance examination to stage the extent of disease. One strategy involves repeating the examination every three years in patients with a disease duration of less than 14 years, every two years in those with disease for 14 to 19 years and every year in those with disease for more than 20 years. More frequent surveillance may be appropriate in patients who have had pancolitis since childhood.
The association between colon cancer and Crohn's disease remains controversial. The overall risk of colon cancer is definitely lower in patients with Crohn's disease than in those with ulcerative colitis. Almost all patients with longstanding, extensive Crohn's colitis have undergone surgical resection of some portion of the colon, which presumably decreases the risk of colon cancer. Screening of surgically treated patients with Crohn's disease is controversial. It is prudent to periodically screen patients with longstanding Crohn's colitis.
Small-bowel cancer is rare in patients with inflammatory bowel disease. However, patients with Crohn's disease are at increased risk for this type of cancer. Screening for small-bowel cancer is neither feasible nor practical.

