A one-minute examination of the placenta performed in the delivery room provides information that may be important to the care of both mother and infant. The findings of this assessment should be documented in the delivery records. During the examination, the size, shape, consistency and completeness of the placenta should be determined, and the presence of accessory lobes, placental infarcts, hemorrhage, tumors and nodules should be noted. The umbilical cord should be assessed for length, insertion, number of vessels, thromboses, knots and the presence of Wharton's jelly. The color, luster and odor of the fetal membranes should be evaluated, and the membranes should be examined for the presence of large (velamentous) vessels. Tissue may be retained because of abnormal lobation of the placenta or because of placenta accreta, placenta increta or placenta percreta. Numerous common and uncommon findings of the placenta, umbilical cord and membranes are associated with abnormal fetal development and perinatal morbidity. The placenta should be submitted for pathologic evaluation if an abnormality is detected or certain indications are present.
Examination of the placenta can yield information that may be important in the immediate and later management of mother and infant. This information may also be essential for protecting the attending physician in the event of an adverse maternal or fetal outcome.
Although some experts argue that all placentas should be examined by a pathologist,1 most hospitals do not mandate this examination. Instead, the delivering physician is usually responsible for determining when pathologic interpretation is necessary. In some urgent situations, decisions must be made before pathologic interpretation is available or has been completed. Therefore, it is essential that the delivering physician perform a thorough, accurate examination of the placenta.
The examination of normal placentas and most abnormal placentas can be accomplished within one minute. Universal examination of the placenta in the delivery room, with documentation of findings and submission of tissue for pathologic evaluation based on abnormal appearance or certain clinical indications, is standard medical practice.3(pp701–3)
Clinical Characteristics of the Normal Placenta
The usual term placenta is about 22 cm in diameter and 2.0 to 2.5 cm thick. It generally weighs approximately 470 g (about 1 lb). However, the measurements can vary considerably, and placentas generally are not weighed in the delivery room.
The maternal surface of the placenta should be dark maroon in color and should be divided into lobules or cotyledons. The structure should appear complete, with no missing cotyledons. The fetal surface of the placenta should be shiny, gray and translucent enough that the color of the underlying maroon villous tissue may be seen.
At term, the typical umbilical cord is 55 to 60 cm in length,3 with a diameter of 2.0 to 2.5 cm. The structure should have abundant Wharton's jelly, and no true knots or thromboses should be present. The total cord length should be estimated in the delivery room, since the delivering physician has access to both the placental and fetal ends.
The normal cord contains two arteries and one vein. During the placental examination, the delivering physician should count the vessels in either the middle third of the cord or the fetal third of the cord, because the arteries are sometimes fused near the placenta and are therefore difficult to differentiate.
Fetal membranes are usually gray, wrinkled, shiny and translucent. The membranes and the placenta have a distinctive metallic odor that is difficult to describe but is easily recognized with experience. Normally, the placenta and the fetal membranes are not malodorous.
Potential Abnormalities of the Placenta
The placental abnormalities that may be detected in the delivery room are discussed in the following sections. Photographs of a number of these abnormalities are presented (Figures 1 through 6). The examination of the placenta and the significance of clinical findings are summarized in Table 1.3–11
FIGURE 1.
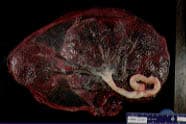
Succenturiate lobe.
FIGURE 2.
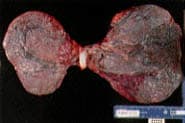
Bilobed placenta.
TABLE 1 Examination of the Placenta in the Delivery Room
| Factors to assess | Condition | Appearance | Clinical significance |
|---|---|---|---|
| Placental completeness | Intact, complete | All cotyledons present | No apparent retained placental fragments |
| No velamentous vessels; vessels taper to periphery of placenta | |||
| Incomplete | Cotyledons missing | Probable retained placental tissue (e.g., in cases of placenta accreta) | |
| Velamentous vessels present (see Figure 6) | Probable retained placental tissue (e.g., in cases of retained succenturiate lobe of placenta) | ||
| Retained tissue is associated with postpartum hemorrhage and infection | |||
| Placental size | Normal | Diameter: about 22 cm | |
| Thickness: 2.0 to 2.5 cm | |||
| Weight: about 470 g (roughly 1 lb) | |||
| Thin placenta | Less than 2 cm | Possible placental insufficiency with intrauterine growth retardation | |
| Placenta membranacea (rare condition in which the placenta is abnormally thin and spread out over a large area of the uterine wall; associated with bleeding and poor fetal outcome) | |||
| Thick placenta | More than 4 cm | Maternal diabetes mellitus | |
| Fetal hydrops | |||
| Intrauterine fetal infections | |||
| Abnormalities of shape | Multiple lobes (bilobate, bipartite, succenturiate, accessory) | See Figures 1 and 2 | Probable retained placenta, with surgical removal required |
| Increased incidence of postpartum infection and hemorrhage | |||
| Placenta membranacea | Hemorrhage and poor fetal outcomes | ||
| Placenta accreta and placenta percreta | Probable retained placenta, with surgical removal required | ||
| Increased incidence of postpartum infection and hemorrhage | |||
| Abnormalities of the maternal placental surface and substance | Placental infarcts | Firm pale or gray areas | Old infarcts |
| Pregnancy-induced hypertension | |||
| Systemic lupus erythematosus | |||
| Advanced maternal age | |||
| Dark areas | Fresh infarcts | ||
| Pregnancy-induced hypertension | |||
| Systemic lupus erythematosus | |||
| Advanced maternal age | |||
| Fibrin deposition | Firm gray areas | No clinical significance unless extensive, in which case there may be placental insufficiency with intrauterine growth retardation or other poor fetal outcome | |
| Placental bleeding (e.g., abruption) | Clot, especially an adherent clot toward the center of the placenta, with distortion of placental shape | Associated with abruption | |
| Fresh clot located along the margin, with no distortion of placental shape | Marginal hematoma: no clinical significance if the clot is small | ||
| Chorioangioma | Fleshy, dark red | If small, probably of no clinical significance | |
| If large, may be associated with fetal hydrops | |||
| Choriocarcinoma | Resembles a fresh infarct | Very rare with a normal gestation | |
| Hydatidiform mole | Grape-like cluster of edematous villi | Very rare with a normal gestation | |
| Abnormalities of the fetal placental surface | Fetal anemia | Pale fetal surface | Anemia in newborn |
| Fetal hydrops | |||
| Hemorrhage requiring transfusion | |||
| Circumvallate placenta | Thick ring of membranes (see Figure 3) | Prematurity | |
| Prenatal bleeding | |||
| Abruption | |||
| Multiparity | |||
| Early fluid loss | |||
| Circummarginate placenta | Inner membrane ring thinner than circumvallete placenta (see Figure 4) | Probably of no clinical significance, but may be associated with an increase in fetal malformations | |
| Amnion nodosum | Multiple tiny white, gray or yellow nodules (see Figure 5) | Oligohydramnios | |
| Renal agenesis | |||
| Pulmonary hypoplasia | |||
| Squamous metaplasia | Multiple tiny white, gray or yellow nodules especially around the cord insertion | Common and probably of no clinical significance | |
| Fetus papyraceus and fetus compressus | One or several nodules or thickenings | Deceased twin | |
| May be associated with otherwise unexplained fetal demise | |||
| Amnionic bands | Delicate or robust bands of amnion | Amputation of fetal parts | |
| Fetal death | |||
| Abnormalities of the umbilical cord | Cord length | Measure cord length and include the fetal and maternal ends (normal length: about 40 to 70 cm) | |
| Short cord | Less than 40 cm | Poorly active fetus | |
| Down syndrome | |||
| Werdnig-Hoffmann disease | |||
| Decreased intelligence quotient | |||
| Fetal malformations | |||
| Myopathic and neuropathic disease | |||
| Cord rupture, hemorrhage or stricture | |||
| Breech or other fetal malpresentation | |||
| Prolonged second stage of labor | |||
| Abruption | |||
| Uterine inversion | |||
| Long cord | More than 100 cm | Fetal hyperkinesis | |
| Increased risk of fetal entanglement | |||
| Increased risk of torsion and knots | |||
| Thromboses | |||
| Thin cord and decreased amount of Wharton's jelly | Narrow areas in the cord (normal cord has a relatively uniform diameter of 2.0 to 2.5 cm) | Postmaturity and oligohydramnios | |
| Torsion and fetal death | |||
| Edema | Diffuse | Hemolytic disease | |
| Prematurity | |||
| Cesarean section | |||
| Maternal preeclampsia | |||
| Eclampsia | |||
| Maternal diabetes mellitus | |||
| Transient tachypnea of the newborn | |||
| Idiopathic respiratory distress | |||
| Focal | Trisomy 18 syndrome | ||
| Patent urachus | |||
| Omphalocele | |||
| Necrotizing funisitis | Distinctive segmental resemblance to a barber's pole | Syphilis and other acute, subacute and chronic infections | |
| Possible swelling, necrosis, thrombosis and calcifications | |||
| Velamentous cord insertion | See Figure 6 | Increased risk of fetal hemorrhage from the unprotected vessels, as well as vascular compression and thrombosis | |
| Advanced maternal age | |||
| Diabetes mellitus | |||
| Smoking | |||
| Single umbilical artery | |||
| Fetal malformations | |||
| Cord knot | Fetal compromise if the knot is tight | ||
| Entanglement | Fetal compromise, especially at delivery | ||
| Abnormal number of vessels | Expect two arteries, one vein | If only one artery is present, up to nearly a 50 percent incidence of fetal anomalies | |
| Count the number of vessels at more than 5 cm from the placental end of the cord | Cord more prone to compression | ||
| Other thromboses | Clot in vessel(s) on cut section | Fetal compromise | |
| Amnionic web at the base of the cord | Fetal compromise | ||
| Abnormalities of the membranes | Color | Green | Meconium staining |
| Old blood from an earlier bleeding event | |||
| Infection (myeloperoxidase in leukocytes) | |||
| Smell | Malodorous | Possible infection | |
| Fecal odor: possibly Fusobacterium or Bacteroides infection | |||
| Sweet odor: possibly Clostridium or Listeria infection |
Placental Completeness
Evaluating placental completeness is of critical, immediate importance in the delivery room. Retained placental tissue is associated with postpartum hemorrhage and infection.
The maternal surface of the placenta should be inspected to be certain that all cotyledons are present. Then the fetal membranes should be inspected past the edges of the placenta. Large vessels beyond these edges indicate the possibility that an entire placental lobe (e.g., succenturiate or accessory lobe) may have been retained (Figure 1).
All or part of the placenta is retained in placenta accreta, placenta increta and placenta percreta. In these conditions, the placental tissues grow into the myometrium to lesser or greater depths. Manual exploration and the removal of retained placental tissue are necessary in these cases.
Placental Size
Placentas less than 2.5 cm thick are associated with intrauterine growth retardation of the fetus.4 Placentas more than 4 cm thick have an association with maternal diabetes mellitus, fetal hydrops (of both immune and nonimmune etiology) and intrauterine fetal infections.5(pp423–36,476,542–613)
An extremely thin placenta may represent placenta membranacea. In this condition, the entire uterine cavity is lined with thin placenta. Placenta membranacea is associated with a very poor fetal outcome.
Placental Shape
Extra placental lobes are important, primarily because they may lead to retained placental tissue (Figure 2).
Blood may be adherent to the maternal surface of the placenta, particularly at or near the margin. If the blood is rather firmly attached, and especially if it distorts the placenta, it may represent an abruption. The dimensions and volume of the placenta should be estimated.
Placental Consistency and Surfaces
The placenta should be palpated, and the fetal and maternal surfaces should be carefully examined.
Maternal Surface. In a term infant without anemia, the maternal surface of the placenta should be dark maroon. In a premature infant, the placenta is lighter in color. Pallor of the maternal surface indicates the presence of fetal anemia, which may be a sign of hemorrhage. With prompt recognition of fetal hemorrhage (such as occurs in vasa previa), lifesaving transfusion can be performed.
Clots on the maternal surface, particularly adherent centrally located clots, may represent placental abruption. It should be emphasized, however, that abruption is a clinical diagnosis.
Fetal Surface. A thick ring of membranes on the fetal surface of the placenta may represent a circumvallate placenta (Figure 3), which is associated with prematurity, prenatal bleeding, abruption, multiparity and early fluid loss.5(pp386–91) A similar but thinner ring of membrane tissue represents a circummarginate placenta (Figure 4). A circummarginate placenta is probably of no clinical significance, although one study found an association between this structural anomaly and an increase in fetal malformations.5(pp386–91)
FIGURE 3.
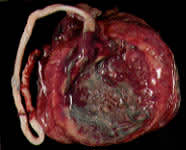
Circumvallate placenta.
FIGURE 4.

Circummarginate placenta.
Numerous small, firm, white, gray or yellow nodules on the fetal surface may represent either amnion nodosum (Figure 5) or squamous metaplasia. Amnion nodosum is associated with oligohydramnios, renal agenesis and poor fetal outcome. Squamous metaplasia is common and is probably of no fetal significance.
FIGURE 5.
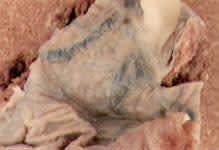
Amnion nodosum.
A nodule or thickening on the fetal surface may represent a vanished twin or a fetus papyraceus. A deceased twin sometimes coexists with a normal fetus, but it may also be associated with demise of the second twin, and this second death may be of uncertain cause.5(pp684–70)
Delicate or more robust bands of amnionic tissue may strangle and amputate fetal parts, including digits, entire limbs, head, neck or trunk. In such cases, amnion may be missing from the placenta but present on the cord.5(pp162–8) When fetal parts are missing or amputated, careful pathologic examination of the placenta is warranted.
Placental Parenchyma
A diffusely soft placenta may represent infection, particularly if the structure is also thickened. Firm areas in the placenta may represent fibrin deposition or infarction. Fresh infarcts are red, while older infarcts are gray. Fibrin deposits are gray and, if extensive, may be associated with intrauterine growth retardation and other poor fetal outcomes. If infarcts or fibrin occupy less than 5 percent of the placental mass, they are usually unimportant.
Focal fleshy, dark-red areas may represent chorioangiomas.5(pp423–36) These benign hemangiomas occur in 1 percent of placentas. While small chorioangiomas are usually of no clinical significance, large chorioangiomas are associated with fetal anemia, thrombocytopenia, hydrops, hydramnios, intrauterine growth retardation, prematurity and stillbirth.5(pp841–6)
Gestational trophoblastic neoplasia, including benign hydatidiform moles, invasive moles and choriocarcinoma, only rarely coexist with viable gestations. Moles appear as grape-like clusters of edematous villi, while choriocarcinoma may look much like an infarct.6
Apparent hemorrhage deep to the fetal membranes or a dark-colored cyst may represent Breus' mole, which is associated with Turner's syndrome (45, X) and with fetal demise.5(pp293–6)
Any suspicious specimen must be examined by a pathologist, with follow-up as indicated by the disease process.
Umbilical Cord
While opinions of authorities differ with regard to the limits of normal for cord length, 40 to 70 cm would appear to be a reasonable range.3,5(pp183–5),7 The typical umbilical cord is long enough (55 to 60 cm) to allow the infant to begin nursing before placental delivery. This provides a release of oxytocin to facilitate uterine contractions and both the shearing and delivery of the placenta.3
In part, cord length is genetically determined. However, the length of the umbilical cord is also increased by the tension the fetus places on the cord. Hence, a short cord is associated with a less active fetus, fetal malformations, myopathic and neuropathic diseases, Down syndrome and oligohydramnios.
Short cords may result in cord rupture, hemorrhage and stricture. Cords of insufficient length may also result in breech and other fetal malpresentations, a prolonged second stage of labor, abruption and uterine inversion.3
The umbilical cord may become excessively long because of fetal hyperkinesis. Long cords are associated with entanglements, torsion, knots and thromboses.
Abnormalities of cord length are clearly associated with an array of longstanding intrauterine factors and consequences, some of which may become apparent only much later in a child's life. Hence, cord length should be documented for every delivery.
Cord Diameter and Inflammation
Throughout its length, the typical umbilical cord has a fairly uniform diameter (2.0 to 2.5 cm). Narrow areas may represent a focal deficiency of Wharton's jelly and are associated with torsion and fetal death.3
Diffuse edema of the cord is associated with hemolytic disease, prematurity, cesarean section, maternal preeclampsia, eclampsia and diabetes mellitus. Cord edema may also be associated with either transient tachypnea of the newborn or idiopathic respiratory distress syndrome. Focally edematous cords are associated with trisomy 18 syndrome, patent urachus and omphalocele.
Necrotizing funisitis is a severe inflammation of the cord that sometimes has a distinctive segmental resemblance to a barber's pole.8 This inflammatory condition may represent syphilis or some other acute, subacute or chronic infection. Swelling, necrosis, thrombosis and calcifications may be present.5(pp278–80)
Cord Insertion
The umbilical cord typically inserts into the placenta near its center. About 90 percent of cord insertions are central or eccentric. About 7 percent of umbilical insertions occur at the placental margin. Marginal insertions are generally benign.
In about 1 percent of singleton fetuses, cord insertion is velamentous (Figure 6). This type of cord insertion is associated with an increased risk of fetal hemorrhage from the unprotected vessels, as well as vascular compression and thrombosis. Velamentous cord insertion is also associated with advanced maternal age, diabetes mellitus, smoking, a single umbilical artery and fetal malformations.
FIGURE 6.
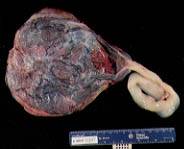
Velamentous cord insertion.
Webs of amnion at the base of the cord may compromise circulation to the fetus.
Cord Knots
A true cord knot occurs when the fetus passes through a loop of umbilical cord, usually early in pregnancy. In most cases, a knot does not cause fetal compromise. However, if sufficient tension is placed on the cord before or during labor and delivery, blood flow may be cut off, and signs of fetal asphyxia may occur.
Cord Vessels
The umbilical cord typically contains two arteries and a single vein. If only one artery and one vein are grossly visible, the fetal anomaly rate is nearly 50 percent.11 These anomalies may affect the cardiovascular, genitourinary or gastrointestinal system, and other systems as well.5(pp183–5),9
Thromboses
Thrombosis of cord vessels is often overlooked by both delivering physicians and pathologists. This is an important cause of fetal injury.10
Free Fetal Membranes
Fetal membranes should be thin, gray and glistening. Thick, dull, discolored or foul-smelling membranes indicate the possibility of infection. The nature of the odor may provide a clue to the infecting organism: a fecal odor may indicate Fusobacterium or Bacteroides, while a sweet odor may indicate Clostridium or Listeria.5(pp542)
Green-colored fetal membranes are frequently the result of meconium staining. However, a green color may be imparted by changing blood pigments from an earlier bleeding event or by the myeloperoxidase in leukocytes in the case of infection.
Thick green slime that easily rinses off the membranes is meconium. Any other pigmentation requires histologic determination.
Pathologic Examination of the Placenta
When any abnormality of potential clinical significance is identified, the placenta should be sent for pathologic examination. Cultures or additional special studies should be obtained before the placenta is sent to pathology.
Indications for pathologic examination include a poor pregnancy outcome (prematurity, intrauterine growth retardation, perinatal death and asphyxia), systemic maternal disorders, third-trimester bleeding and evidence of fetal or maternal infection.2(pp701–3)
In patients with multiple gestations, the placenta must be examined for zygosity, vascular anastomoses and numerous common abnormalities. The individual cords should be labeled for the pathologist (i.e., twin A and twin B).
Controversy exists as to whether the placental specimen should be fixed in formalin or refrigerated. Therefore, the guidelines of the delivering physician's institution should be followed. If the specimen is to be fixed, copious amounts of formalin (at least 10 times as much formalin as placental tissue) should be used. Refrigerating the specimen in a container the size of a large ice cream carton preserves the placenta very well for a few days and allows it to be available if the infant shows some abnormal clinical findings during the first few days of life.
The pathologist should be notified of the clinical situation, and questions should be answered. Interest in placental pathology has increased in recent years, and the pathologist can contribute to the diagnosis and treatment of the mother and infant.2(pp660–721)
Ultrasonography and Fetal Surgery
Ultrasonography can be used to identify many of the abnormalities described in this article. Indeed, ultrasonography and the possibility of surgical intervention have been partly responsible for the resurgence of interest in placental pathology. Discussions of ultrasound examination and fetal surgery, as well as the development of the placenta, are beyond the intended scope of this article. However, an excellent review article on placental ultrasonography is available.4
Final Comment
Pathology fees for placental examination vary but are similar to those for a surgical specimen. The question of whether pathologic examination of the placenta will change outcome must be considered.
Documentation of the delivery room examination of the placenta, plus the pathologic examination when indicated clinically or by hospital policy, will preserve valuable information.12 A suggested form for documenting the clinical findings and the indications for pathologic evaluation is presented in Figure 7. Since some indications for placental examination are not apparent at the time of delivery, it may be wise to save the placenta (labeled and refrigerated or preserved in formalin) until the neonatal outcome is determined.
FIGURE 7. Description of the Placenta and Associated Structures

Suggested form for documenting the clinical findings of the placental examination, as well as the indications for pathologic examination.
It has been suggested that all placentas should be sent for complete pathologic examination,7 but this approach may represent an overuse of resources. In any event, if placentas are sent to pathology for the indications that have been discussed in this article, few necessary examinations are likely to be missed.1 At minimum, the hospital's requirements for the handling of placentas should be followed.

