Management of the most common type of dementia—Alzheimer's disease—is becoming increasingly sophisticated. Differentiation of Alzheimer's disease from vascular dementia has become therapeutically important, since the choice of treatments depends on the diagnosis. Two cholinesterase inhibitors, donepezil and tacrine, are labeled for use in patients with Alzheimer's disease. Other therapies, such as estrogen, nonsteroidal anti-inflammatory drugs and vitamin E, are sometimes used and show promise in delaying the progression of this dementia. Behavior problems, which often accompany the disease, can be managed using environmental modification, alterations in caregiving and medication. In the terminal phase of the illness, quality care involves implementing advance directives, communicating with the family, individualizing care and attending to patient comfort.
Until recently, the family physician's approach to patients with dementia consisted primarily of helping family members cope with the burden of caregiving and providing information about services such as adult day care programs and nursing homes. Over the past decade, however, research and innovations in care have increased the options available to physicians who care for demented patients and their families. While no dramatic breakthroughs have occurred, the overall management of dementia is becoming increasingly sophisticated, and it is likely that this trend will continue during the next decade.
Progress in Understanding the Mechanisms of Disease
Dementia is an acquired syndrome in which intellectual ability decreases to the point that it interferes with daily function. The prevalence of dementia increases markedly after 75 years of age, making it largely a disease of older persons. Dementia is not the same as delirium, in which the onset of cognitive impairment tends to be more acute, is due to reversible causes such as drugs or acute illness, and involves alterations in consciousness.
Currently, it is believed that there are two major causes of dementia in older persons: Alzheimer's disease and vascular dementia. Reliable estimates of the prevalence of the various causes of dementia in primary care settings are not available, but the best estimates indicate that approximately 60 percent of dementing illnesses are due to Alzheimer's disease, 15 percent are due to vascular dementia and many of the remainder involve concurrent Alzheimer's disease and vascular dementia (“mixed dementia”).1,2 Recently, some authorities have added a third diagnosis—Lewy body disease—to the list of common causes of dementia. However, it is uncertain whether this new designation represents a distinct diagnostic entity or a subtype, since many patients diagnosed with Lewy body dementia appear to have co-existing Alzheimer's disease.1
Alzheimer's disease is associated with diffuse neuron injury and death, with characteristic pathologic findings of senile plaques and neurofibrillary tangles. Clinically, persons with Alzheimer's disease demonstrate a slow, progressive decline in multiple areas, including cognition and the ability to perform activities of daily living. Behavior disturbances, ranging in severity from repeated questioning to physical violence, are common. The average duration of the disease is 10 years, during which afflicted persons progress from mild memory loss to the need for 24-hour supervision to total dependency and death (Figure 1). It is unclear whether Alzheimer's disease represents a single entity or several variants. Some experts believe that there are distinct subtypes of Alzheimer's disease, such as Lewy body disease (in which parkinsonian signs, visual hallucinations or alterations in alertness or attention, or all of these symptoms, are conspicuous) and frontotemporal dementia (in which disinhibition, misconduct or apathy, or all of these signs, are prominent). The well-established risk factors for Alzheimer's disease are age, a family history of the disease and Down syndrome.2
FIGURE 1.
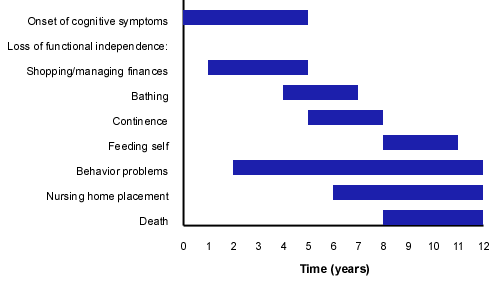
Typical occurrence of manifestations of Alzheimer's disease.
The etiology of Alzheimer's disease remains elusive, although considerable progress has been made in understanding its biochemical and genetic mechanisms. It is known that the 42-amino-acid fragment of ß-amyloid precursor protein is critically important to the pathogenesis of senile plaques and that most forms of familial Alzheimer's disease (including the dementia of Down syndrome) are associated with overproduction of ß-amyloid precursor protein.3,4 Several of the proteins that comprise neurofibrillary tangles—most notably phosphorylated tau protein and ubiquitin—have been identified, but the relationship between plaque formation, neurofibrillary tangle formation and cell injury remains unclear.3 It is known that the gene for the apolipoprotein E ∈ 4 allele is found about three times as frequently in persons with Alzheimer's disease as in age-matched control subjects and that persons who are homozygous for the gene are at higher risk for the disease than those who are not homozygous for the gene. However, the specificity and sensitivity of the test are too low to recommend it for screening in the general population.5
The pace of research in these areas is rapid, and it is likely that these research directions will lead to more effective treatments in the future. Figure 2 represents a summary of current understanding of the mechanism(s) of Alzheimer's disease and the site of action of common therapies.
FIGURE 2.
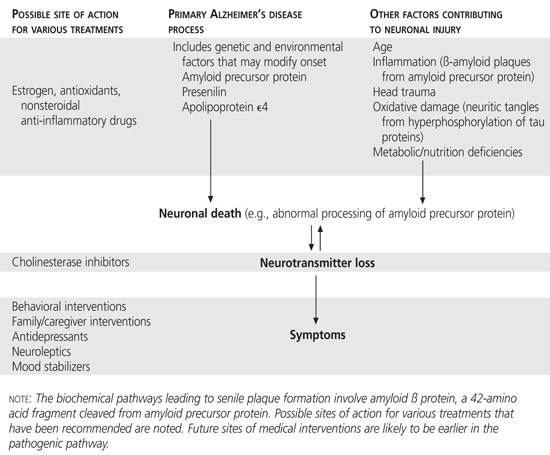
Possible pathogenesis of Alzheimer's disease, based on recent research.
Vascular dementia is generally one of two types: multi-infarct dementia or subcortical vascular dementia. In cases of multi-infarct dementia, the neurologic examination often reveals focal, asymmetric abnormalities, and multiple strokes are evident on computed tomography (CT) or magnetic resonance imaging (MRI) of the brain. Subcortical vascular dementia (also known as Binswanger's disease) is characterized by vascular disease that predominantly affects tiny arterioles in the region of the midbrain. In patients with Binswanger's disease, gait problems are often prominent early in the course, and CT or MRI scans appear normal except for increased signal in the deep white matter (a nonspecific sign). The causes of and risk factors for vascular dementia are not well known but are likely to be similar to those for arteriosclerosis and small vessel arterial disease elsewhere in the body: hypertension, diabetes mellitus, smoking and hypercholesterolemia.
Diagnostic Evaluation
Establishment of the differential diagnosis of Alzheimer's disease can usually be performed in the primary care setting. The most important diagnostic tools remain the history given by the patient, family members and other caregivers, and the office-based clinical examination. The evaluation of a patient with suspected dementia should be implemented whenever an older person is reported to have gradually increasing difficulty or change in at least one of the following areas: learning and retaining new information, handling complex tasks, reasoning ability, spatial ability and orientation, language ability or behavior.2 The clinical evaluation should be used to review changes in daily function; to quantify the presence and degree of cognitive impairment through use of a standardized instrument, such as the Mini-Mental State Examination6; to identify localizing neurologic findings; and to differentiate between delirium, Alzheimer's disease and vascular dementia7 (Table 1).1,2,6
TABLE 1 Selected Diagnostic Criteria for Delirium, Dementia, Alzheimer's Disease and Vascular Dementia
| Diagnostic criteria for delirium |
| 1. A change in cognition or development of a perceptual disturbance is present and not explained by a preexisting, established or evolving dementia. |
| 2. The disturbance developed over a short period of time (usually hours to days) and tends to fluctuate. |
| 3. The level of consciousness (awareness of the environment) is disturbed or fluctuates. |
| 4. There is evidence that a drug, acute illness or metabolic disturbance is present that could explain the change in cognition. |
| Diagnostic criteria for dementia |
| 1. Cognitive impairment is present, as demonstrated by: (1) memory loss and (2) impairment of language, praxis, recognition or abstract thinking. |
| 2. The cognitive impairment is chronic and progressive and has resulted in functional decline. |
| 3. Delirium has been ruled out. |
| Diagnostic criteria for Alzheimer's disease |
| 1. Dementia is present. |
| 2. History, physical and mental status examinations are consistent with Alzheimer's disease. |
| 3. Screening blood tests (CBC, BUN, calcium, liver function, thyroid function, vitamin B12 and others as indicated) and review of medications do not reveal any major untreated cause of cognitive impairment. |
| 4. Brain imaging study (CT or MRI) is normal or shows atrophy (some authorities do not recommend neuroimaging studies except in uncertain cases). |
| Diagnostic criteria for vascular dementia |
| 1. Dementia is present. |
| 2. Two or more of the following are present: (1) focal neurologic signs on physical examination; (2) an onset that was abrupt, stepwise or stroke-related; (3) brain imaging study (CT or MRI) shows multiple strokes. |
CBC = complete blood cell count; BUN = blood urea nitrogen; CT = computed tomography; MRI = magnetic resonance imaging.
The establishment of a specific diagnosis is especially challenging early in the course of dementing illness. In such cases, consultation with a neuropsychologist, a neurologist or a geriatric psychiatrist may be helpful. Testing a blood sample for apolipoprotein E may be used as an adjunct to other diagnostic procedures; possession of two ∈4 alleles indicates an increased likelihood that the dementia is due to Alzheimer's disease.5 However, as previously mentioned, this test cannot be used to make the diagnosis in the absence of a strong suspicion of Alzheimer's disease and is not recommended for diagnosis but only as an additional confirmatory test in some patients.
In the past, the evaluation of patients with suspected dementia focused on looking for reversible or potentially reversible causes of the syndrome. It is now acknowledged that “reversible” dementia rarely occurs and that family physicians should instead rule out delirium and search for co-existing conditions that worsen an existing dementia. In practice, this means reviewing patients' medications, screening carefully for depression and ruling out metabolic conditions such as nutritional deficiencies, diabetes mellitus, uremia, alterations in electrolytes and thyroid disease. Observation over time is often required before the diagnosis can be certain, so physicians should be careful not to label a patient with Alzheimer's disease until a proper assessment has been completed.
Use of Medication to Delay Symptom Progression
High-quality primary care remains the cornerstone of disease management: regular visits at least every four to six months, frequent contact with family members, availability by telephone, general health maintenance (including attention to vision and hearing) and minimization of medications with potential effects on mood or cognition (Table 2). However, specific drug treatment options are available and can be offered to patients and their families. Currently, two cholinesterase inhibitors are available for use in treating Alzheimer's disease: tacrine (Cognex) and donepezil (Aricept). Several others, such as metrifonate8 and rivastigmine, are in the late stages of development and testing. Effects are modest: a good response to either drug will return the patient's function to the level that was present between six and 12 months before medication was started. Beneficial effects on behavior symptoms also have been reported in some patients, and the use of tacrine has been shown to delay nursing home placement.1 Figure 3 provides guidelines for the use of cholinesterase inhibitors in the treatment of Alzheimer's disease.9
TABLE 2 Medications That May Cause or Worsen Cognitive Impairment
| Antiarrhythmic agents |
| Antibiotics |
| Anticholinergic agents |
| Anticonvulsants |
| Antidepressants |
| Antiemetics |
| Antihistamines/decongestants |
| Antihypertensive agents |
| Antimanic agents |
| Antineoplastic agents |
| Antiparkinsonian agents |
| Cardiotonic agents* |
| Corticosteroids |
| Histamine H2-receptor antagonists |
| Immunosuppressive agents |
| Muscle relaxants |
| Narcotic analgesics |
| Nonsteroidal anti-inflammatory drugs* |
| Radiocontrast agents |
| Sedatives |
*—In overdose.
FIGURE 3.
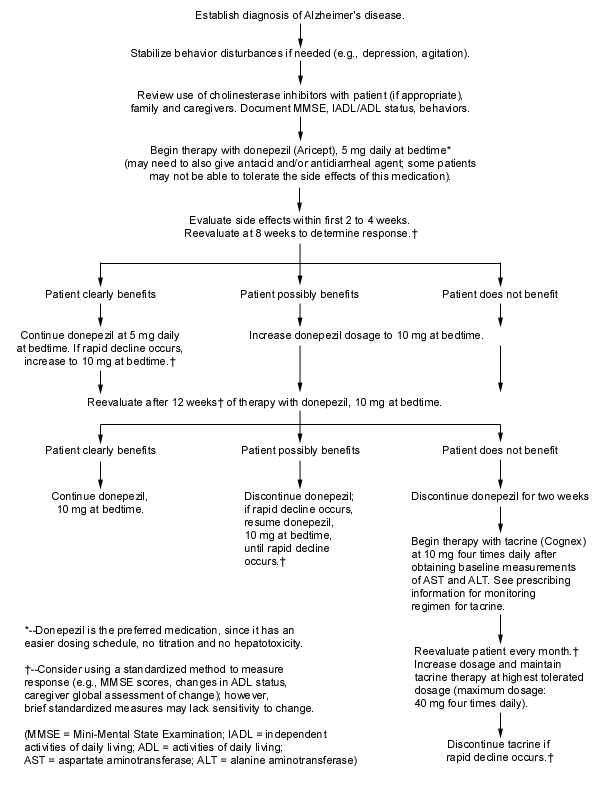
Practical guidelines for use of cholinesterase inhibitors in patients with Alzheimer's disease.
In addition to cholinesterase inhibitors, several nonspecific drug therapies appear to have a role in delaying the progression of dementia. Three longitudinal studies of estrogen replacement therapy have found a significant protective effect, with the risk of Alzheimer's disease being inversely related to dosage and duration of use.10–12 It may be, however, that estrogen use does not prevent Alzheimer's disease but merely delays its expression. Although estrogen use may slightly increase the risk of breast cancer, estrogen's beneficial effects on bone and the cardiovascular system could encourage many women to consider its use if Alzheimer's disease is of particular concern. Further research is under way.
Nonsteroidal anti-inflammatory drugs (NSAIDs) such as aspirin and ibuprofen also have been associated with a lower incidence of dementia. Whether this lower incidence is due to a reduction in inflammation associated with Alzheimer's disease or to protection against stroke is uncertain.13,14 Because of the risks of NSAID use, which include gastrointestinal and renal toxicity, these agents cannot be routinely recommended for use as a preventive measure against Alzheimer's disease at the present time.15
A recent clinical trial indicated that therapy with vitamin E supplementation or selegiline (Eldepryl) may significantly slow the progression of moderate Alzheimer's disease. Since vitamin E had far fewer adverse effects and was associated with comparable efficacy to that of selegiline, this study supports the consideration of vitamin E supplementation in persons with dementia or those who are at risk for the disease.16 Dosages of up to 2,000 IU daily are used.
Ginkgo biloba extract, which contains antioxidants similar to vitamin E, has been recently reported to delay symptom progression in dementia, but little is known about the long-term effects of the extract, the precise pharmacologic content of the extract or the active ingredient.17
In summary, family physicians who care for persons with dementia should consider prescribing medication to slow disease progression in most patients who are not in the terminal stages of disease. Potential adverse effects must be considered (e.g., bleeding disorders and increased risk of falls with high-dose vitamin E, gastrointestinal bleeding with NSAIDs). Table 38,15,17,18 summarizes the recommendations that are supported by randomized clinical trials (although these medications are not necessarily labeled for use in patients with Alzheimer's disease). In the future, additional medications are expected to become available.
TABLE 3 Drug Treatments to Be Considered in Patients with Dementia
| Dementia diagnosis | Medication | Typical dosage | Comments | |
|---|---|---|---|---|
| Alzheimer's disease | Donepezil (Aricept) | 5 to 10 mg once daily | Equal efficacy and fewer side effects than tacrine; elevated hepatic transaminase levels are rare; diarrhea and abdominal pain occur occasionally. | |
| or | ||||
| Tacrine (Cognex) | 10 mg four times daily, increased at 6-week intervals to 40 mg four times daily | Elevated hepatic transaminase levels are common; check ALT every 2 weeks during dosage titration and if nausea is present; diarrhea is also common. (See prescribing information about other monitoring recommendations.) | ||
| Ibuprofen (Motrin)* | 400 mg two to three times daily | Use with caution because of risk of gastrointestinal or renal toxicity. | ||
| Conjugated estrogens (Premarin)* | 0.625 mg daily | Prescribe for women only; consider cyclic progestin for patients with an intact uterus. | ||
| Vitamin E (alpha or mixed tocopherols)* | 800 to 2,000 IU daily | Mild anticoagulant effects. | ||
| Vascular dementia | Antihypertensive medication | Seek to maintain systolic blood pressure below 150 to 160 mm Hg, but titrate dosage to maintain diastolic blood pressure in the 85 to 95 mm Hg range | Because of widespread cerebral atherosclerosis in many patients, treatment that lowers diastolic blood pressure below 85 to 90 may worsen cognitive impairment. | |
| Enteric-coated aspirin | 81 to 325 mg daily | Consider warfarin (Coumadin) if atrial fibrillation is present. | ||
| Vitamin E (alpha or mixed tocopherols) | 800 to 2,000 IU daily | Mild anticoagulant effects. | ||
*—Although these medications cannot be recommended for treatment of Alzheimer's disease at present, current research is promising.
ALT = alanine aminotransferase; IU = international units.
Treating Behavior Disturbances Associated with Dementia
The psychiatric and behavior disturbances associated with dementia often cause the greatest problems for caregivers. These disturbances include personality changes, behaviors associated with memory loss (e.g., asking repeated questions), agitation, resistiveness and physical combativeness in care situations, socially inappropriate behaviors, mood disturbances, delusions and hallucinations. Over the past 15 years, considerable progress has been made in developing systematic approaches to behavior problems in dementia. Table 4 provides an overview of recommended approaches.
TABLE 4 Management of Behavior Problems in Patients with Alzheimer's Disease
| Use the five “P's” for effective assessment and planning |
| Describe the problem accurately—what happens, who is involved, when it occurs, what precedes the behavior and what consequences result for the patient and caregivers. |
| Assess the patient—are there unmet needs that may be triggering the behavior? Is an undetected medical problem present (e.g., pain)? What stresses are acting on the patient? Are delusions or other psychotic signs present? Is the patient depressed? |
| Brainstorm possible causes—an interdisciplinary team meeting is the best setting in which to do this. |
| Develop a plan. |
| Implement the plan, reassess it, modify it if necessary and pass it on to all caregivers if it is successful. |
| First-line treatment: behavioral and environmental therapy |
| The key to success is changing what the patient experiences. |
| Caregiver action (such as a calm demeanor, validation of feelings and beliefs, and use of techniques such as redirection and distraction) is important. |
| Environmental modification can often remove or lessen the provoking stimuli. |
| Indications for medication, with recommended initial drugs |
| Medicate early if delusions, depression or pain is present; otherwise, medicate only if agitation cannot be controlled by behavioral methods. |
| If medication is used, remember that older persons in general require lower dosages and that persons with dementia are especially susceptible to adverse drug effects. |
| If the main problem is delusions, use an antipsychotic agent such as haloperidol (Haldol) or risperidone (Risperdal). |
| If the main problem is agitation, use a short-acting antianxiety agent such as lorazepam (Ativan) or buspirone (Buspar*). |
| If the main problem is depression, use a selective serotonin reuptake inhibitor, beginning at one half the starting dosage for a young adult. If sedation is also desirable, consider using trazodone (Desyrel). |
| If the main problem is pain, use a standard dosage of acetaminophen or, in cases of severe pain and/or disruptive behaviors accompanied by pain, a very low-dose narcotic, possibly accompanied by a low-dose antipsychotic or antianxiety agent. |
*—May not be immediately effective.
One of the most useful and often over-looked strategies to reduce the occurrence of problem behaviors is environment modification. Hazards should be minimized; for example, persons with moderate dementia should not drive.15 In addition, persons with dementing illness often overreact to features of the environment that are unpleasant or overstimulating. Examples of environmental factors that can lead to agitation and aggression include noise (e.g., a police siren), situations that offer too many choices (e.g., deciding what to wear from a closet full of clothes) and normal activities that are misunderstood (e.g., people walking or talking outside a room may arouse suspicion). For these reasons, careful attention to the environment can help family and institutional caregivers manage problem behaviors without medication. For example, a person who becomes disruptive during meals in a noisy nursing-home dining room may eat quietly when placed at a four-person table set with formal dinnerware in a small room.
In the institutional setting, special care units for patients with Alzheimer's disease have become increasingly popular.19 These units attempt to provide up-to-date management of behavior problems with a minimum of physical and pharmacologic restraints. Critical evaluation of such units has yielded mixed results, however, and a recent large study failed to show any slowing in the rates of functional decline among persons with dementia who reside in special care units.20
Late-Stage Care
For family caregivers and their physicians, the terminal phases of dementing illness present some of the most difficult challenges. In late dementia, the patient has few or no verbal abilities, does not recognize family or care-givers, is incontinent and usually nonambulatory, and often demonstrates agitated behaviors such as resistiveness, moaning or shouting. The quality of life for many such patients appears poor, yet they cannot verbalize their wishes regarding how and to what extent they would want to be treated. Thus, it falls on family members and physicians to try to provide the kind of care the patient would request if he or she could reason and speak.
The best approach to care of patients with late-stage dementia is for family physicians to counsel all patients to make advance directives before they become this ill. These advance directives should include the following points: whether and when cardiopulmonary resuscitation (CPR) should be withheld (here the family physician may wish to counsel patients that the rate of successful [that is, survival until discharge from the hospital] CPR in nursing home patients with significant functional impairment is less than 5 percent); the circumstances under which other life-supporting measures—such as feeding tubes, antibiotics, hospitalization and surgical procedures—would not be desired; and who should be legally designated as durable power of attorney for health care decisions, to assist the physician and family in determining what the patient's wishes would have been in case the patient becomes unable to indicate this, as happens in late-stage dementia.
Care for patients with late-stage dementia should not consist merely of waiting for a terminal event, however. Instead, the goal should be enhancement of the quality of life for patients and their loved ones. Comfort care for end-stage dementia is often inadequate. For example, 62 percent of persons with mild to moderate dementia report pain, most commonly in the back, knee, foot, shoulder, neck, wrist, head or hip, yet few persons with end-stage dementia, who may be experiencing the same types of pain, receive pain medication.21
One of the most difficult areas is feeding. Persons with end-stage dementia nearly always become difficult to feed, often refusing or spitting out food. Studies indicate that tube feedings do not prolong life and are associated with discomfort and medical complications; therefore, tube feeding is generally not recommended for persons in the end stage of dementing illness.22,23 Alternatives to tube feeding include switching consistency (e.g., from regular to soft or from pureed to liquid), adding thickening agents if aspiration is a problem, feeding favorite foods (and abandoning restrictions such as reduced salt or low-fat diets) and allotting adequate time to feed the person. Table 518, 22–26 lists some of the most salient components of quality care for patients with end-stage dementia. The family physician can play a crucial role in this stage of illness by advising and guiding caregiving staff and family in the management of everyday situations and in the crises that inevitably develop.
TABLE 5 Principles of Quality Care for Patients with End-Stage Dementia
| Communicate to the family and caregivers when you consider the patient to have end-stage illness. | |
| Emphasize that the exact prognosis is uncertain but that goals of care often are different in this stage. Criteria for end-stage disease are: | |
| • Severe confusion (e.g., Mini-Mental State Examination score <3), and | |
| • Inability to dress, toilet or walk independently, and | |
| • Extremely limited speech (usually <5 words), and | |
| • At least one of the following medical complications occurring within the previous 3 months: dehydration, pneumonia, urosepsis or refusal/inability to consume adequate calories despite encouragement by staff | |
| Meet with the family to determine care goals. | |
| • Review advance directives | |
| • Discuss palliative care and how it can be provided (including if and when hospice can be involved) | |
| • Determine preferences for care should acute illness arise, and document them | |
| • Let family members know that you will be available | |
| Provide basic palliative care to all end-stage patients. | |
| This should include the following: | |
| • Companionship for the patient until the end | |
| • Someone for the family to talk with during the terminal stage and after death | |
| • Regular, gentle range-of-motion exercises (unless painful) | |
| • Minimization of physical restraint | |
| • Limiting medications to those that may be helpful to the patient in the short run | |
| • Skin care to maximize comfort and prevent breakdown (e.g., protection of bony prominences, frequent repositioning, attention to changing wet linens/incontinence pads, massage with lubricating lotion) | |
| • Activities that the patient can still enjoy (e.g., gentle bathing and dressing, comfortable linens and bedclothes, familiar music, favorite foods) | |
| • Identification and relief of pain (most patients probably have some musculoskeletal pain) | |
| • Minimization of constipation and fecal soiling by avoiding cathartics and constipating agents, facilitating defecation and removing stool accumulation with enemas if necessary | |
| • Relief of dry mouth with sips of liquids and mouth care (cleaning, use of various swabs, ice chips and lubricants) | |
| • Feeding that is appropriate for the goals of care, with emphasis on a food consistency and delivery that are accepted by the patient, with adequate time allowed for patients who eat slowly and require feeding | |
The Future
Alzheimer's disease is projected to increase in prevalence during the coming decades, as the U.S. population continues to live longer. At the same time, advances in basic and clinical research will make management of the disease more sophisticated. Tests are likely to become available to identify persons at risk for Alzheimer's disease as well as those who have very early disease. More drugs will become available, and it is possible that disease progression may be delayed for many years. Thus, the family physician's role will become increasingly pivotal, as early diagnosis and aggressive treatment are added to the care coordination and caregiver support roles that family physicians have traditionally provided for persons with dementia.

