Type 2 diabetes mellitus (formerly called non–insulin-dependent diabetes) causes abnormal carbohydrate, lipid and protein metabolism associated with insulin resistance and impaired insulin secretion. Insulin resistance is a major contributor to progression of the disease and to complications of diabetes. Type 2 diabetes is a common and underdiagnosed condition that poses treatment challenges to family practitioners. The introduction of new oral agents within the past three years has expanded the range of possible combination regimens available for treating type 2 diabetes. Despite the choice of pharmacologic agents, physicians must stress the nonpharmacologic approaches of diet modification, weight control and regular exercise. Pharmacologic approaches must be based on patient characteristics, level of glucose control and cost considerations. Combinations of different oral agents may be useful for controlling hyperglycemia before insulin therapy becomes necessary. A stepped-care approach to drug therapy may provide the most rational, cost-efficient approach to management of this disease. Pharmaco-economic analyses of clinical trials are needed to determine cost-effective treatment strategies for management of type 2 diabetes.
Diabetes mellitus affects approximately 16 million people in the United States and accounts for about one sixth of all expenditures for health care.1 Ninety percent of patients with diabetes have type 2 diabetes (formerly known as non–insulin-dependent diabetes) and often require oral agents or insulin for glucose control. The mortality rate in patients with diabetes may be up to 11 times higher than in persons without the disease.1 Diabetes is a leading cause of blindness, renal failure, and foot and leg amputations in adults. Managed care and budgeted resources challenge clinicians to provide comprehensive health care to patients with diabetes.
Within the past three years, the introduction of new oral agents has prompted questions regarding the most cost-effective approach for management of type 2 diabetes. Since pharmaco-economic data concerning antidiabetic regimens are limited, clinicians must select the most appropriate agent(s) based on patient characteristics, level of glucose control and cost. A rational approach for managing patients with varying stages of disease requires an understanding of features that lead to disease progression, and a thorough review of the new oral agents for the treatment of type 2 diabetes and the clinical and economic basis for appropriate drug selection.
Disease Progression
Diabetes is a group of metabolic diseases with characteristic hyperglycemia associated with defects in insulin secretion, insulin action, or both. Type 1 diabetes (formerly known as insulin-dependent diabetes) is characterized by beta cell destruction, usually leading to absolute insulin deficiency. Its etiology is either immune mediated, related to physical destruction of the pancreas (as in pancreatitis or pancreatic cancer) or idiopathic. Type 2 diabetes presents as a spectrum of metabolic abnormalities with prominent insulin resistance and relative insulin deficiency.2 The effect of diabetes is not limited to carbohydrate metabolism. Lipid and protein metabolism play an important role in the progression of the disease.3 Abnormal glucose metabolism accounts for poorly regulated biochemical processes that glycosylate hemoglobin and other proteins and lipids throughout the body. The progression of diabetes is caused by numerous metabolic events that occur over a period of years. By controlling these metabolic events, the progression of the disease may be slowed or stopped.
The prevalence of diabetes in persons 45 to 64 years of age is 7 percent, but the proportion increases significantly in persons 65 years of age or older.4 Certain minority populations have even higher rates. Despite its high prevalence, diabetes is largely underdiagnosed (Table 1).5 It is estimated that over 8 million people in the United States alone are unaware that they have the disease.1 There is evidence that retinopathy begins to develop at least seven years before the clinical diagnosis of type 2 diabetes is made.6 Patients with undiagnosed diabetes mellitus are at serious risk for coronary heart disease, stroke and peripheral vascular disease, and have a greater likelihood of dyslipidemia, hypertension and obesity.
Criteria for diagnosing diabetes in asymptomatic, undiagnosed patients are outlined in Table 2.5 These criteria are associated with the following factors: the steep rise in the incidence of the disease after 45 years of age, the negligible likelihood of developing any diabetic complications within three years after a negative screening test, and knowledge of documented risk factors for the disease (Table 3) .7 Studies have defined the glycosylated hemoglobin A1c (HbA1c) level above which the likelihood of having or developing macrovascular or microvascular disease increases. Because HbA1c and fasting blood glucose (FBG) are the measurements of choice for monitoring diabetes, decisions about when and how to implement treatment strategies are based on these parameters.8
TABLE 3 Major Risk Factors for Type 2 Diabetes Mellitus
| Family history of diabetes (parents or siblings with diabetes) |
| Obesity (≥120% over desired body weight or BMI ≥27 kg per m2) |
| Race/ethnicity (e.g., black, Hispanic, native American, Asian American, Pacific Islander) |
| Age ≥45 years |
| Previously identified IFG or IGT |
| Hypertension (≥140/90 mm Hg) |
| HDL cholesterol level ≤35 mg per dL (0.90 mmol per L) and/or a trigyceride level ≥250 mg per dL (2.83 mmol per L) |
| History of gestational diabetes mellitus or delivery of babies over 4,032 g (9 lb) |
IFG = impaired fasting glucose; IGT = impaired glucose tolerance; HDL = high-density lipoprotein.
Information from Screening for type 2 diabetes. Diabetes Care 1998;21(suppl 1):S20-2.
The National Diabetes Data Group9 developed the current classification and diagnosis of diabetes that is used in the United States. Other diabetic states also may occur that have etiologies differing from those of type 1 and type 2 diabetes. These diabetic states include: genetic defects in beta cell function; genetic defects in insulin action; diseases of the exocrine pancreas (e.g., pancreatitis); trauma; cystic fibrosis; endocrinopathies (e.g., acromegaly, Cushing's syndrome, hyperthyroidism, pheochromocytoma); drug- or chemical-induced; infection; uncommon forms of immune-mediated diabetes; genetic syndromes (e.g., Down syndrome, Klinefelter's syndrome, Turner's syndrome); and gestational diabetes mellitus.
Insulin resistance is defined as an impaired biologic response to exogenous or endogenous insulin in the tissues. Insulin resistance and impaired insulin secretion are the defining features of type 2 diabetes. Patients with insulin resistance syndrome have a group of related clinical and laboratory findings, including glucose intolerance, central obesity, dyslipidemia, hypertension and altered fibrinolysis.10
Insulin resistance occurs because of defective insulin-mediated glucose uptake and utilization, which reflects the inhibition of a glucose transport. Both nonpharmacologic and pharmacologic therapies are used to reduce insulin resistance. Nonpharmacologic approaches include a low-calorie diet, weight loss and regular vigorous exercise. Pharmacologic approaches include the use of metformin (Glucophage) and troglitazone (Rezulin).
Since insulin resistance is hypothesized to be a major factor in the development of type 2 diabetes, treatment of resistance may prevent or delay the onset of diabetes. Presently, the Diabetes Prevention Program11 is under way to ascertain which treatment for insulin resistance may help prevent or delay the onset of diabetes. These treatments include intensive lifestyle changes (7 percent reduction of body weight through caloric restriction and exercise) and use of metformin. Drug treatment for primary prevention of diabetes is not currently warranted.
Insulin resistance is a major factor that contributes to hyperglycemia. Macrovascular complications are strongly associated with hyperinsulinemia that is due to insulin resistance. These complications include cardiovascular, cerebrovascular and peripheral vascular diseases. A number of mechanisms, independent of glucose metabolism, are believed to accelerate the progression of diabetic complications. Insulin-mediated biochemical pathways lead to enhanced vascular smooth muscle proliferation, platelet adhesiveness and vasoconstriction.12
The risk of cardiovascular disease increases with diabetes and is greater in patients with coexisting dyslipidemia. This combination of disease states is associated with significant rates of morbidity and mortality from cardiovascular events. Near-normal or improved glycemic control has been shown to significantly diminish the risk of long-term complications in patients with type 2 diabetes.13 The treatment of nonpregnant patients with “impaired fasting glucose” should begin with lifestyle modification, including meal planning and exercise. Initiation of pharmacologic therapy in these patients has not been shown to improve their prognosis, and the use of insulin and sulfonylureas may lead to complications of hypoglycemia.8
Oral Agents for Type 2 Diabetes
SULFONYLUREAS
With the labeling of tolbutamide (Orinase) by the U.S. Food and Drug Administration in 1962, the sulfonylurea class of drugs quickly became the mainstay of treatment for type 2 diabetes. Although newer agents have recently entered the marketplace, sulfonylureas still play a primary role in pharmacologic management of type 2 diabetes. Patients who respond best to treatment with sulfonylureas include those with a diagnosis of type 2 diabetes before 40 years of age, duration of disease less than five years before initiation of drug therapy and a fasting blood glucose level of less than 300 mg per dL (16.7 mmol per L).14
Approximately two thirds of patients who begin therapy with a sulfonylurea respond, although up to 20 percent of them eventually require additional medication. Few patients with uncontrolled diabetes receive clinical benefit when switched from one sulfonylurea agent to another.14 The use of agents with a longer half-life (e.g., chlorpropamide [Diabinese]) in the elderly and in patients with renal impairment is discouraged because the risk of hypoglycemia is increased.
METFORMIN
Metformin is a biguanide agent that lowers blood glucose primarily by decreasing hepatic glucose output and reducing insulin resistance. Metformin is used as monotherapy or in combination with sulfonylureas for management of type 2 diabetes. When used as monotherapy, metformin does not cause hypoglycemia and is thus termed an “antihyperglycemic.” The use of metformin is contraindicated in patients with renal insufficiency (i.e., a serum creatinine level of 1.5 mg per dL [130 μmol per L] in men and 1.4 mg per dL [120 μmol] in women, or abnormal creatinine clearance) or acute or chronic metabolic acidosis. Metformin should be temporarily withheld before any procedure involving intravascular administration of iodinated contrast media. Normal renal function should be confirmed 48 hours after the procedure before restarting metformin therapy. There is no known reason to discontinue metformin therapy during other parenteral contrast studies.
Extreme caution should be used in patients with severe hepatic dysfunction, hypoxemic states (e.g., severe chronic obstructive pulmonary disease, congestive heart failure), moderate to severe illness and excessive alcohol intake. In these patients, the use of metformin may contribute to the development of lactic acidosis, a condition that is fatal in about 50 percent of patients who develop it (one episode per 100,000 patient-years).15 Cimetidine (Tagamet) decreases the renal clearance of metformin and may potentiate its effects. Patients receiving oral anticoagulant therapy and metformin may require a higher dosage of warfarin (Coumadin) to achieve a therapeutic antithrombotic effect.16 Hemogloblin, hematocrit, red blood cell indexes and renal function should be monitored at least annually in patients taking metformin.
ALPHA-GLUCOSIDASE INHIBITORS
Alpha-glucosidase inhibitors, such as acarbose (Precose) and miglitol (Glyset), are indicated as monotherapy or in combination with sulfonylureas for management of type 2 diabetes. These agents inhibit the breakdown of complex carbohydrates and delay the absorption of monosaccharides from the gastrointestinal tract.17 Acarbose and miglitol should be titrated over two to three weeks to minimize flatulence and other gastrointestinal side effects that commonly lead to discontinuation of these agents. Alpha-glucosidase inhibitors are contraindicated in patients with inflammatory bowel disease, partial intestinal obstruction, a predisposition to intestinal obstruction, colonic ulceration and other gastrointestinal disorders.17
Dose-dependent hepatotoxicity is associated with this drug class, so liver function tests should be carefully monitored in patients receiving higher dosages of these medications (e.g., more than 50 mg three times daily). Transaminase elevations are reversible with discontinuation of the drug and are often asymptomatic. Serum transaminase levels should be checked every three months for the first year patients take the medication and periodically thereafter. Drugs that are susceptible to binding with other agents (e.g., cholestyramine [Questran]) should be taken two to four hours apart from alpha-glucosidase inhibitors to avoid drug interactions. Intestinal absorbents and digestive enzyme preparations should not be administered with acarbose.
TROGLITAZONE
The thiazolidinediones are a unique drug class of “insulin sensitizers” that promote skeletal muscle glucose uptake.18 Troglitazone is the first agent of this drug class to be introduced in the U.S. market and, like metformin, it reduces insulin resistance. Troglitazone is beneficial in patients requiring large daily amounts of insulin (more than 30 units per day) whose diabetes is still uncontrolled. A reduction of up to 50 percent in total daily insulin dosage is possible with drug titration. Troglitazone is also effective when used in combination with other oral agents,19 thereby potentially delaying the need to start insulin therapy.
The U.S. Food and Drug Administration recently ruled that troglitazone should only be used in combination with other diabetic therapies. The effectiveness of oral contraceptives may be decreased with troglitazone administration. Over 150 case reports of hepatotoxicity have been reported with troglitazone, so liver function must be monitored every month for the first eight months of treatment and every other month for four months thereafter.18 Periodic transaminase measurements should be obtained as long as the patient is taking troglitazone.
REPAGLINIDE
Repaglinide (Prandin) is a benzoic acid derivative and the first of the non-sulfonylurea meglitinides introduced in early 1998. The mechanism of action and side effect profile of repaglinide are similar to those of the sulfonylureas.15 This agent has a rapid onset of action and should be taken with meals two to four times daily. Repaglinide is a suitable option for patients with severe sulfa allergy who are not candidates for sulfonylurea therapy. The drug is used as monotherapy or in combination with metformin. It should be titrated cautiously in elderly patients and in those with renal or hepatic dysfunction.
Treatment Rationale
All treatment strategies should emphasize cardiovascular risk reduction, focusing particularly on hypertension control, smoking cessation and correction of dyslipidemia. Diet, exercise and weight reduction should be the cornerstone of management. Before selecting a medication to improve control of diabetes, the family physician should understand the comparative glucose-lowering effects of available agents. The dose-response for the oral agents on levels of FBG, post-prandial glucose and HbA1c is described in Table 4.16–18 The goals of therapy for type 2 diabetes are outlined in Table 5.20
TABLE 4 Dose-Response of Oral Agents for Type 2 Diabetes Mellitus
| Agent | Average FBG reduction (%) | Average PPG reduction (%) | Average HbA1creduction (%) |
|---|---|---|---|
| Sulfonylureas | 25 to 40 | 20 | 2.0 |
| Alpha-glucosidase inhibitors* | 10 to 20 | 40 to 45 | 0.5 to 1.5 |
| Metformin (Glucophage)* | 20 to 40 | 25 | 1.5 to 2.0 |
| Troglitazone (Rezulin)* | 20 | 25 | 1.0 to 1.5 |
FBG = fasting blood glucose; PPG = postprandial glucose; HbA1c = glycosylated hemoglobin A1c
*—Combined with another oral agent or insulin.
TABLE 5 Goals of Therapy for Patients with Diabetes Mellitus*
| Biochemical index | Nondiabetic value | Goal value | When additional action is suggested |
|---|---|---|---|
| Preprandial glucose measurement† | <110 mg per dL (6.1 mmol per L) | 80 mg per dL (4.4 mmol per L) to 120 mg per dL (6.7 mmol per L) | <80 mg per dL (4.4 mmol per L) or >140 mg per dL (7.8 mmol per L) |
| Bedtime glucose measurement† | <120 mg per dL (6.7 mmol per L) | 100 mg per dL (5.6 mmol per L) to 140 mg per dL (7.8 mmol per L) | <100 mg per dL (5.6 mmol per L) or >160 mg per dL (8.9 mmol per L) |
| HbA1c(%) | <6 | <7 | >8 |
HbA1c = glycoslylated hemoglobin.
*—The values shown in this table are by necessity generalized to the entire population of persons with diabetes. Patients with co-morbid diseases, the very young and older adults, and others with unusual conditions or circumstances may warrant different treatment goals. These values are for nonpregnant adults. “Additional action suggested” depends on individual patient circumstances. Such actions may include enhanced diabetes self-management education, co-management with a diabetes team, referral to an endocrinologist, change in pharmacologic therapy, initiation of or increase in self-monitored blood glucose testing, or more frequent contact with the patient. HbA1c is referenced to a non-diabetic range of 4.0 to 6.0 percent.
†—Measurement of capillary blood glucose.
Information from Standards of medical care for patients with diabetes mellitus. Diabetes Care 1998;21(suppl 1):S23–31.
Few clinical trials have been conducted to evaluate the possibility of a “ceiling effect” with select antidiabetic agents. A dose-dependent reduction in HbA1c was observed with glimepiride (Amaryl) in one clinical trial.21 Splitting the total daily dosage of sulfonylurea into two separate doses may be necessary to achieve optimal glycemic control in most patients on medium to high daily dosages of these agents. Patients with type 2 diabetes become less responsive over time to one agent alone and frequently require combination therapy to adequately control their disease.
The current approach to management of drug therapy in patients with type 2 diabetes is to begin insulin therapy if a combination of two oral agents fails to provide adequate glycemic control. For every 10 units of insulin administered, an average of 1 kg of weight may be gained.22 This weight gain, associated with the lipotrophic effect of insulin, promotes greater insulin resistance in patients with severe disease. The result of escalating doses of insulin is a perpetual cycle of weight gain and uncontrolled diabetes. With the advent of the newer oral agents, combination therapy may delay insulin use in patients who traditionally would require insulin early in the course of the disease. Reasonable combinations of oral agents based on mechanism of action include sulfonylurea plus metformin, sulfonylurea plus an alpha-glucosidase inhibitor, sulfonylurea plus troglitazone, repaglinide plus metformin, troglitazone plus metformin, insulin plus metformin, and insulin plus troglitazone.
Daily home glucose monitoring is strongly encouraged in all patients with diabetes who undergo therapy with insulin or oral agents. Patients with uncontrolled disease and those requiring medication adjustment need more intensive monitoring. The frequency and timing of glucose monitoring should be individualized for each patient. The optimal frequency of self-monitoring of blood glucose in patients with type 2 diabetes is not known, but monitoring should be performed often enough to facilitate reaching treatment goals. Efforts should be made to substantially increase appropriate use of self-monitoring, providing both the patient and the practitioner with substantial information necessary to achieve glycemic goals. The family practitioner should negotiate appropriate monitoring for patients with finger soreness, hectic lifestyles or more stable diabetes.
A stepped-care approach to drug therapy provides a rational, effective method of disease management. All oral agents available for treatment of type 2 diabetes are indicated for use as monotherapy after diet, exercise and weight reduction have failed to control hyperglycemia. Currently, there is no clinical advantage in selecting a sulfonylurea, metformin or insulin as initial therapy, although certain causes of hyperglycemia or patient characteristics may make one preferable to another (Figure 1). The United Kingdom Prospective Diabetes Study (UKPDS) showed a similar reduction in microvascular complications in patients treated with these drug classes. No difference in cardiovascular outcomes was apparent.13 Because alpha-glucosidase inhibitors and troglitazone were not evaluated in the UKPDS, it is not known how these agents affect diabetes-related complications.
FIGURE 1. Causes of Hyperglycemia and Action of Oral Antidiabetic Agents
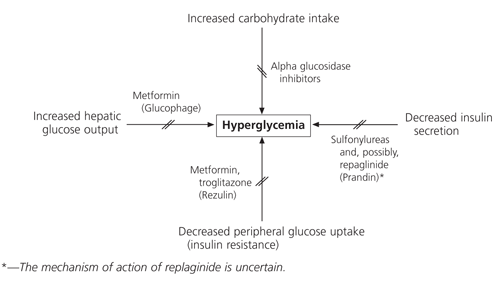
Causes of hyperglycemia and sites of action of oral antidiabetic agents.
When possible, sulfonylureas should be prescribed as initial therapy in nonobese patients since they are less expensive than the newer oral agents. Metformin is an excellent initial agent for obese patients (i.e., those greater than 120 percent of ideal body weight) or as add-on therapy in patients whose disease is not controlled with sulfonylurea therapy. An alpha-glucosidase inhibitor or troglitazone may be an alternative to a sulfonylurea or metformin as add-on therapy in patients with uncontrolled disease or significant renal dysfunction.
Patients generally require some form of insulin treatment if their diabetes cannot be managed adequately with maximum dosages of two oral agents. The use of three oral agents in combination to control blood glucose is of limited benefit. Since metformin and troglitazone are currently the only antidiabetic medications that decrease insulin resistance, they are logical choices for use with insulin regimens. In patients whose diabetes remains uncontrolled even with moderate daily dosages of insulin (i.e., more than 30 units per day), metformin and troglitazone are effective in reducing insulin requirements and improving glycemic control. Currently, no pharmaco-economic analyses have been performed to evaluate the cost-effectiveness of various antidiabetic regimens.
Recommended components of initial and follow-up office visits for patients with diabetes are outlined in Tables 6 and 7.20
TABLE 6 Components of Initial Office Visit for Patients with Diabetes Mellitus
| Medical history |
| Symptoms, laboratory results related to diagnosis |
| Nutritional assessment, weight history |
| Previous and present treatment plans |
| Medications |
| Medical nutrition therapy |
| Self-management training |
| Self-management blood glucose results |
| Current treatment program |
| Exercise history |
| Acute complications |
| History of infections |
| Chronic diabetic complications |
| Medication history |
| Family history |
| Risk factors for coronary heart disease |
| Psychosocial/economic factors |
| Physical examination |
| Height and weight |
| Blood pressure |
| Ophthalmoscopic examination |
| Thyroid palpation |
| Cardiac examination |
| Evaluation of pulses |
| Foot examination |
| Skin examination |
| Neurologic examination |
| Oral examination |
| Sexual maturation (if peripubertal) |
| Laboratory evaluation |
| Fasting plasma glucose (optional) |
| Glycohemoglobin |
| Fasting lipid profile |
| Serum creatinine |
| Urinalysis |
| Urine culture (if indicated) |
| Thyroid function tests (if indicated) |
| Electrocardiogram (adults) |
| Management plan |
| Short- and long-term goals |
| Medications |
| Medical nutrition therapy |
| Lifestyle changes |
| Self-management education |
| Monitoring instructions |
| Annual referral to eye specialist |
| Specialty consultations (as indicated) |
| Agreement on continuing support/follow-up |
Information from Standards of medical care for patients with diabetes mellitus. Diabetes Care 1998;21(suppl 1):S23–31.
TABLE 7 Components of Follow-Up Visits for Patients with Diabetes Mellitus
| Contact frequency | |
| Daily for initiation of insulin or change in regimen | |
| Weekly for initiation of oral glucose-lowering agent(s) or change in regimen | |
| Routine diabetes visits | |
| Quarterly or more often for patients who are not meeting goals | |
| Semi-annually for other patients | |
| Medical history (assessment of treatment regimen) | |
| Frequency/severity of hypoglycemia or hyperglycemia | |
| SMBG results | |
| Patient regimen adjustments | |
| Adherence problems | |
| Lifestyle changes | |
| Symptoms of complications | |
| Other medical illnesses | |
| Medications | |
| Psychosocial issues | |
| Physical examination | |
| Physical examination annually | |
| Dilated eye examination annually | |
| Every regular diabetes visit | |
| Weight | |
| Blood pressure | |
| Previous abnormalities on the physical examination | |
| Foot examination | |
| Laboratory evaluation | |
| Hemoglobin A1c | |
| Quarterly if treatment changes or patient is not meeting goals | |
| Twice per year if stable | |
| Fasting plasma glucose (optional) | |
| Fasting lipid profile annually | |
| Urinalysis for protein annually | |
| Microalbumin measurement annually (if urinalysis is negative for protein) | |
| Management plan review | |
| Each visit | |
| Short- and long-term goals | |
| Medications | |
| Glycemia | |
| Frequency/severity of hypoglycemia | |
| SMBG results | |
| Complications | |
| Control of dyslipidemia | |
| Blood pressure | |
| Weight | |
| Medical nutrition therapy | |
| Exercise regimen | |
| Adherence to self-management training | |
| Follow-up of referrals | |
| Psychosocial adjustments | |
| Annually | |
| Knowledge of diabetes | |
| Self-management skills | |
SMBG = self-monitoring of blood glucose.
Information from Standards of medical care for patients with diabetes mellitus. Diabetes Care 1998;21(suppl 1): S23–31

