Vaccination is the primary measure for preventing morbidity and mortality from influenza. During the influenza season, family physicians must distinguish influenza from the common cold and other flu-like illnesses. Signs and symptoms of influenza include abrupt onset of fever, severe myalgias, anorexia, sore throat, headache, cough, and malaise. Clinical diagnosis can be difficult or nonspecific when patients have other symptoms (e.g., stuffy nose, sneezing, cough, sore throat) that can be caused by various respiratory viruses or bacterial pathogens. Family physicians can improve diagnostic accuracy by being aware of the epidemiology of influenza. During outbreaks of influenza, commercially available rapid assays can be used to identify type A and B viruses. On average, rapid in-office tests are more than 70 percent sensitive and 90 percent specific for viral antigens. The assays vary in complexity, specificity, sensitivity, time to obtain results, specimen analyzed, and cost. The results of rapid viral tests can guide treatment decisions.
The administration of influenza vaccine to appropriate risk groups remains a high priority for family physicians. Nevertheless, from late fall through early spring of every year, family physicians must distinguish among the many types of “common cold” viruses, bacteria, and influenza viruses that cause respiratory tract symptoms. The diagnostic and treatment challenge may be difficult because all of these pathogens can cause nonspecific respiratory and somatic symptoms.
In the clinical setting, rapid, accurate diagnosis of influenza is based on the history, physical examination and, in some instances, laboratory testing before treatment is initiated. This article reviews the clinical diagnosis of influenza and provides information about rapid in-office diagnostic tests.
Clinical Course of Uncomplicated Influenza
Most physicians use a cluster of signs and symptoms to diagnose influenza. Influenza viruses, common cold viruses, and bacteria cause similar upper respiratory tract symptoms, but the intensity, severity, and frequency of the symptoms vary. Features of influenza and the common cold are compared in Table 1.1,2
TABLE 1 Comparison of Influenza and the Common Cold
| Features | Influenza | Common cold |
|---|---|---|
| Onset* | Abrupt | More gradual |
| Fever* | Common: 37.7°C to 40.0°C (100°F to 104°F) | Uncommon or only 0.5°C (1°F) increase |
| Myalgia* | Severe, common | Uncommon |
| Arthralgia | Severe, common | Uncommon |
| Anorexia | Common | Uncommon |
| Headache | Severe, common | Mild, uncommon |
| Cough (dry)* | Common, severe | Mild to moderate |
| Malaise | Severe | Mild |
| Fatigue, weakness | More common than with the common cold; lasts 2 to 3 weeks | Very mild, short lasting |
| Chest discomfort | Common, severe | Mild to moderate |
| Stuffy nose | Occasional | Common |
| Sneezing | Occasional | Common |
| Sore throat | Occasional | Common |
*—Clusters of more severe or common features may be more likely to predict influenza. Information from references 1 and 2.
Influenza may present as a mild respiratory illness similar to the common cold. It also can present as severe prostration without characteristic signs and symptoms. Lack of specificity can make clinical diagnosis difficult.
The first sign of influenza may be the abrupt onset of fever (temperature of 37.7°C to 40.0°C [100°F to 104°F]) and dry cough. The temperature gradually declines, but fever may be present for up to a week (Figure 1).3 Other symptoms may include chills, anorexia, generalized or frontal headache, severe myalgia and arthralgia, weakness, and fatigue. Symptoms of respiratory illness may predominate, and patients also may have a sore throat and a persistent nonproductive cough, with or without substernal or pleuritic chest pain.1,4–6
FIGURE 1. Natural Course of Influenza
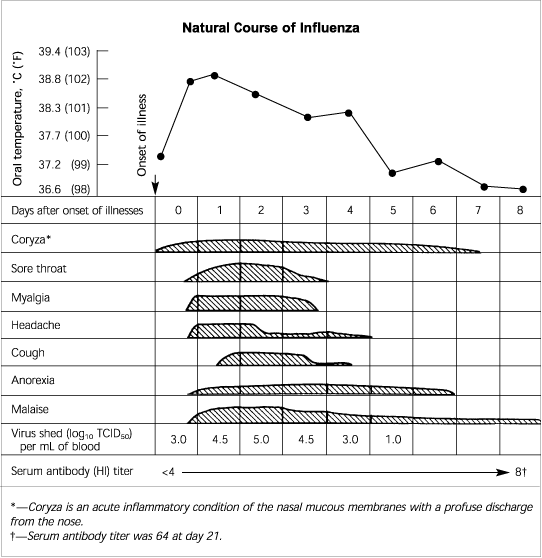
Clinical characteristics of naturally occurring influenza A in an otherwise healthy 28-year-old male patient. (TCID50 = median tissue culture infective dose; HI = hemagglutination inhibition)
Adapted from Dolin R. Influenza: current concepts. Am Fam Physician 1976;14(3):74.
Patients with influenza may be flushed and have conjunctival injection, pain on eye motion, or photophobia. Nonexudative pharyngitis, rhonchi, or scattered rales may be present. In elderly patients, the skin may be hot, dry, or diaphoretic. The leukocyte count, if obtained, may be normal or decreased. The chest radiograph typically reveals no acute infiltrates.1,4–6
Identifying Influenza Based on Clinical Findings
Determining the presence of influenza on solely clinical grounds is complicated by the poor specificity of clinical diagnosis, poor sensitivity of clinical findings, other pathogens that cause similar symptoms, and influenza subtypes that cause different symptoms.7–10
The identification of influenza is improved when physicians are aware that influenza virus is present in their area. Data from one study11 that attempted to predict influenza virus infections during epidemics indicated that cough and fever (temperature higher than 38°C [100.4°F]) had a positive predictive value of 86.8 percent, a negative predictive value of 39.3 percent, a sensitivity of 77.6 percent, and a specificity of 55 percent. In another study,12 data collected from clinical trials on the use of zanamivir (Relenza) in patients who presented within 48 hours of symptom onset indicated that cough and fever had the best positive predictive value (79 percent; P <0.001); furthermore, the higher the temperature, the stronger the positive predictive value was. In this study,12 the presence of myalgias and sore throat did not significantly improve the positive predictive value. Other studies4,12,13 have reported a possible sensitivity of 77 to 85 percent when fever (temperature higher than 38°C [100.4°F]), cough, and myalgias are present during the influenza season.4,13
Study findings suggest that the likelihood of accurate diagnosis increases when epidemiologic data support the clinical suspicion of influenza virus infection. Physicians can obtain epidemiologic information on influenza from their local or state health department, as well as a number of Web sites (Table 2). From October through May, weekly influenza surveillance data can be obtained from the Centers for Disease Control and Prevention (CDC)—through voice system information: 888-232-3228; or by fax: 888-232-3299 [request document number 361100]). Information from the CDC and other sources can make physicians aware of influenza viruses and subtypes that are circulating locally and nationally. Information on influenza viruses throughout the United States can be helpful for patients who may be traveling to or from high-risk influenza areas.
TABLE 2 Internet Resources for Information on Influenza
| CDC influenza information*:www.cdc.gov/ncidod/diseases/flu/weekly.htm,www.cdc.gov/ncidod/diseases/flu/fluvirus.htm |
| National Institute of Allergy and Infectious Diseases:www.niaid.nih.gov |
| World Health Organization:www.who.int/health-topics/influenza.htm |
| U.S. Department of Health and Human Services:www.dhhs.gov |
| American Lung Association:www.lungusa.org/diseases/luninfluenz.html |
| National Foundation for Infectious Disease:www.nfid.org/factsheets/inflfacts.html |
| Consumer health information from sources such as Aetna InteliHealth, featuring information from Harvard Medical School:www.intelihealth.com |
CDC = Centers for Disease Control and Prevention.
*—From October through May, the CDC collects and reports influenza surveillance data (voice information telephone system: 888-232-3228; fax: 888-232-3299 [request document number 361100]).
Complications of Influenza
Epidemiology, time course, illness severity, and the presence of underlying medical conditions are important points in distinguishing between uncomplicated and complicated viral or postviral illnesses. Although other complications of influenza can occur, pneumonia is the most common (Table 3).1,5,14 Influenza may be followed by viral pneumonia, secondary bacterial pneumonia, or mixed viral-bacterial pneumonia.
Healthy children six to 23 months of age, adults 65 years and older, and persons six months to 64 years with cardiopulmonary, respiratory, renal, metabolic, or immunodeficient conditions, as well as pregnant women, are predisposed to greater morbidity and mortality from influenza.15 Consequently, these patients should be considered at high risk for complications.
Laboratory Testing
Symptoms of influenza-like illness can be caused by multiple respiratory viruses, including respiratory syncytial virus, parainfluenza virus, adenovirus, rhinovirus, and corona-virus. Data indicate that up to 70 percent of patients with influenza-like illness may not be infected with an influenza virus.16 Once the physician has made the tentative diagnosis of influenza, laboratory testing may be an appropriate confirmatory step.
RAPID VIRAL TESTS
Office-based techniques for detecting influenza A and B viruses include enzyme immunoassay and direct immunofluorescence. Using these sophisticated, yet relatively easy techniques, the physician can diagnose influenza A and B in 10 to 20 minutes.
At present, five office-based assays for detecting influenza virus A and/or B are available commercially: BD Directigen Flu A+B, BD Directigen Flu A, FLU OIA, QuickVue Influenza A/B Test, and ZstatFlu (Table 4).4,17–26 The assays vary in sensitivity, specificity, complexity, and skill and time required for performance and interpretation.4,21,27 Another assay, the Influenza A/B Rapid Test, cannot be obtained commercially but is available to physicians who are participating in influenza surveillance studies.
TABLE 4 Commercial Rapid Viral Tests
| Features | BD Directigen Flu A+B17,18 | BD Directigen Flu A4,18–20 | FLU OIA4,18,20–23 | QuickVue Influenza A/B Test17,18,24 | ZstatFlu4,18,20,21,25,26 |
|---|---|---|---|---|---|
| Approved for use in CLIA-waived laboratories | No | No | No | Yes* | Yes |
| Distinguishes between influenza A and B viruses | Yes | No | No | No | No |
| Cost per test | $20 to $24 | $17 to $20 | $12 to $17 | $15 to $18 | $20 |
| Assay time | 15 minutes | 15 minutes | 15 minutes | 10 minutes | 20 minutes |
| Number of steps | 9 | 11 | 7 | 3 | 4 |
| Preferred specimen | Nasopharyngeal aspirate | Nasopharyngeal washing or aspirate | Nasopharyngeal swab | Nasopharyngeal washing or aspirate | Throat swab |
| Sensitivity | 67% to 96% | 60% to 77% | 62% to 88% | 73% to 81% | 51% to 72% |
| Specificity | 88% to 97% | 80% to 90% | 52% to 80% | 96% to 99% | 93% to 100% |
| Shelf life | <1 year | Variable | 1 year | 1 year | 3 years |
| Storage | Room temperature | Room temperature | 4°C to 8°C (39.2°F to 46.4°F) | Room temperature | Room temperature |
| Batching | No | No | No | No | Yes |
| Portability | Yes, with media | Yes, liquid transport | 1 hour only | Yes, must be refrigerated | >24 hours without media |
CLIA = Clinical Laboratory Improvement Amendments; CPT = Current Procedural Terminology.
*—Requires modifier QW coding to be recognized as a CLIA-waived test for CPT coding.
BD Directigen Flu A+B
This is the only test that distinguishes between influenza A and B viral antigens. The assay costs about $20 and takes about 15 minutes to perform. A swab is used to obtain a specimen from nasopharyn-geal washings or aspirates or directly from the nose or throat; results vary based on the specimen type. Compared with cell culture, the assay has an overall sensitivity of 86.2 percent for influenza A and 80.8 percent for influenza B; overall specificity is 90.7 percent for influenza A and 99.5 percent for influenza B. Evaluations of the test in office laboratories have shown a 93 percent accuracy for influenza A and a 92 percent accuracy for influenza B.17,18
BD Directigen Flu A
This enzyme immunoassay performs best when nasopharyngeal washings and aspirates are used. The assay involves 11 steps and takes about 15 minutes to complete. It detects only influenza A, with an overall sensitivity of 91 percent and an overall specificity of 95 percent compared with viral culture. Cost is less than $20.4,18–20
FLU OIA
This enhanced optical immunoassay detects influenza A and B nucleoproteins but does not distinguish between influenza A and B viruses from various specimens. Experienced personnel can perform this test and obtain results in 15 minutes at a cost of less than $18. The test is reported to have an overall sensitivity of 80 percent and an overall specificity of 73 percent.4,18,20–23
QuickVue Influenza A/B Test
This assay uses monoclonal antibodies to detect influenza A and B viral nucleoproteins but does not distinguish between them. The test is approved for use in Clinical Laboratory Improvement Amendments (CLIA)-waived facilities. The type of specimen influences the results. Sensitivity is greater for nasal washes or aspirates than for nasal swabs (81 percent versus 73 percent); specificity is also greater (99 percent versus 96 percent). Compared with culture, the test has a sensitivity of 75 percent and a specificity of 97 percent. The assay can be performed in about 10 minutes at a cost of approximately $15.17,18,24
ZstatFlu
This enzyme-based assay detects influenza A and B viral neuraminidase activity in throat swab specimens. It does not distinguish between influenza A and B viruses. This test has been approved for use in CLIA-waived laboratories. Compared with culture, the assay has a sensitivity of 65 percent for influenza A virus and 57 percent for influenza B virus; comparative specificity ranges from 95 to 100 percent. The test costs about $20, and results are available in about 20 minutes.4,18,20,21,26
Comments
During the time of year when outbreaks of influenza-like illnesses are common, rapid office testing can be performed to determine if influenza is the cause of a patient's symptoms, to support clinical decision-making, and to define treatment choices. However, physicians should be aware of the limitations of rapid viral tests. The CDC estimates that up to 30 percent of samples may produce negative rapid test results, yet be positive by viral culture methods; rapid tests may also yield false-positive results.18 Furthermore, although several rapid viral tests can identify both influenza A and B viruses, at this time only the BD Directigen Flu A+B test distinguishes between A and B viruses, and this test is not approved for use in CLIA-waived facilities. The QuickVue Influenza A/B test is approved for use in CLIA-waived facilities.
A recent study28 comparing FLU OIA, QuickVue Influenza A/B Test, ZstatFlu, and BD Directigen Flu A in a pediatric practice found that FLU OIA and ZstatFlu required, on average, twice as much technician time. Test sensitivities ranged from 72 to 95 percent, with ZstatFlu being significantly less sensitive; specificities ranged from 76 to 86 percent. The positive predictive value ranged from 80 to 86 percent; the negative predictive value ranged from 75 to 94 percent, with ZstatFlu having the lowest negative predictive value (75 percent).
Influenza test selection should be based on CLIA status, sensitivity and specificity, required specimens, time to obtain results, operator skill, and cost. If the chosen test is positive, antiviral drug therapy may be considered if symptoms have been present for less than 48 hours. A negative test result may support the decision to withhold antiviral drug therapy.
OTHER TESTS
Although viral culture is the most accurate method for identifying specific viral strains and subtypes, it is an impractical choice for office-based physicians who must prescribe antiviral drug therapy within 48 hours of symptom onset. Viral culture results are not available for two to 10 days, depending on methodology. Furthermore, viral culture is costly (about $100) and requires special laboratory equipment and procedures, as well as skilled technical expertise for correct performance. In the future, polymerase chain reaction (PCR) techniques may replace viral culture for identifying viral strains and subtypes.27
In sophisticated laboratories, nucleic acid detection techniques (e.g., PCR or nucleic acid amplification), electron microscopy, cytology, and histology may be used to diagnose viral illnesses. These tests are more commonly performed in reference or hospital laboratories. They do not help with outpatient management.
In clinical practice, rapid viral tests would be most helpful in deciding whether to initiate treatment for influenza. However, antibody titers (viral serology) may provide some retrospective assessment of symptoms 10 to 14 days after the initial serologic specimen was obtained. A fourfold rise in titer provides confirmatory evidence of infection long after empiric antiviral treatment would have been completed.
Leukocytosis may be present in patients with influenza. A white blood cell count higher than 15,000 per mm3 (15 ×109 per L), with or without a left shift, may be a sign of secondary bacterial infection or pneumonia.1
Final Comment
The symptoms of uncomplicated influenza are somewhat unique in terms of their abrupt onset, the time of year in which influenza presents, and the clinical course of the illness. The presence of an influenza epidemic in a given city or state makes the diagnosis of this illness more likely and will increase the sensitivity and positive predictive value of any rapid test that is performed. On average, rapid in-office tests are more than 70 percent sensitive and 90 percent specific for viral antigens.18
Accurate diagnosis and prompt treatment with antiviral agents can have a positive effect on public health and reduce the economic impact of influenza. Initiation of antiviral therapy within 48 hours of symptom onset is recommended for more rapid reduction of symptoms and reduced viral shedding.29 [Evidence level C, consensus/expert guidelines]
Antiviral agents include amantadine (Sym-metrel), rimantadine (Flumadine), zanamivir, and oseltamivir (Tamiflu). Amantadine and rimantadine are recommended for the treatment of influenza A, but they are not active against influenza B virus infections. However, if both influenza A and B are present in the community, use of a rapid assay test that distinguishes the influenza B virus would justify treatment with the more expensive neura-minidase inhibitors (oseltamivir and zanamivir), which are active against both influenza A and B viruses.29 Whatever the treatment, physicians must always be alert for postinfluenza complications that can increase morbidity and mortality from influenza.
Administering influenza and pneumonia vaccines should be one of the highest priorities in primary care medicine.30 Family physicians can prevent the spread of influenza and treat this illness more effectively by tracking epidemiologic data, knowing the presenting symptoms of this illness, making the correct diagnosis, initiating treatment promptly, and remaining alert for potential complications.

