Each year, more than 1 million patients are admitted to U.S. hospitals because of unstable angina and non–ST-segment elevation myocardial infarction (UA/NSTEMI). To help standardize the assessment and treatment of these patients, the American College of Cardiology and the American Heart Association convened a task force to formulate a management guideline. This guideline, which was published in 2000 and updated in 2002, highlights recent medical advances and is a practical tool to help physicians provide medical care for patients with UA/NSTEMI. Management of suspected UA/NSTEMI has four components: initial evaluation and management; hospital care; coronary revascularization; and hospital discharge and post-hospital care. Part I of this two-part article discusses the first two components of management. During the initial evaluation, the history, physical examination, electrocardiogram, and cardiac biomarkers are used to determine the likelihood that the patient has UA/NSTEMI and to aid in risk assessment when the diagnosis is established. Hospital care consists of appropriate initial triage and monitoring. Medical treatment includes anti-ischemic therapy (oxygen, nitroglycerin, beta blocker), antiplatelet therapy (aspirin, clopidogrel, platelet glycoprotein IIb/IIIa inhibitor), and antithrombotic therapy (heparin, low-molecular-weight heparin).
The term “acute coronary syndrome” encompasses unstable angina and non–ST-segment elevation myocardial infarction (UA/NSTEMI) and ST-segment elevation myocardial infarction (STEMI). UA/NSTEMI is the combination of two closely related clinical entities (i.e., a syndrome), whereas STEMI is a distinct clinical entity. UA/NSTEMI is characterized by an imbalance between myocardial oxygen supply and demand. Most often, the syndrome develops because of decreased myocardial perfusion resulting from coronary narrowing caused by nonocclusive thrombus formation subsequent to disruption of an atherosclerotic plaque. In contrast, STEMI results from an occlusive thrombus.
Strength of Recommendations
| Key clinical recommendation | SOR labels | References |
|---|---|---|
| The updated ACC/AHA guideline recommends use of the thienopyridine clopidogrel (Plavix) in patients who cannot tolerate aspirin (ACC/AHA class I). | A | 2, 3, 17 |
| The updated ACC/AHA guideline considers the use of clopidogrel in addition to aspirin to have a class I indication in patients with UA/NSTEMI who are undergoing an early noninterventional or interventional approach and are not at high risk for bleeding. | A | 2, 3, 18, 19 |
| When elective coronary artery bypass grafting is planned, clopidogrel should be withheld for five to seven days. | C | 2, 3 |
| Platelet glycoprotein IIb/IIIa inhibitors have an ACC/AHA class I indication in patients for whom catheterization and percutaneous coronary intervention are planned. These agents should be administered in addition to aspirin and heparin. | A | 20–23, 25 |
| Anticoagulation with unfractionated heparin or low-molecular-weight heparin for patients with UA/NSTEMI has an ACC/AHA class I indication. The heparin is to be used in addition to aspirin or clopidogrel. | A | 27 |
ACC = American College of Cardiology; AHA = American Heart Association; UA/NSTEMI = unstable angina and non–ST-segment elevation myocardial infarction.
Each year, more than 5 million patients present to U.S. emergency departments with chest pain and related symptoms.1 Approximately 1.4 million of these patients are admitted for management of UA/NSTEMI.1 Because of the scope of the problem, it is important for family physicians to understand the diagnosis, risk assessment, and treatment of this syndrome.
To help standardize the assessment and treatment of patients with UA/NSTEMI, the American College of Cardiology (ACC) and the American Heart Association (AHA) convened a task force to produce a management guideline. The ACC/AHA guideline, which was published in 20001 and updated in 2002,2,3 divides the management of suspected UA/NSTEMI into four components: initial evaluation and management; hospital care; coronary revascularization; and hospital discharge and post-hospital care. This two-part article focuses on the major management recommendations in the guideline, using the ACC/AHA classification of recommendations (Table 1).3 Recent advances in management are highlighted. Part I reviews the first two components of management, and part II4 reviews the other two components.
TABLE 1 ACC/AHA Classification of Evidence Used in the UA/NSTEMI Guideline
| Class I: conditions for which there is evidence or general agreement that a given procedure or treatment is useful and effective |
| Class II: conditions for which there is conflicting evidence or a divergence of opinion about the usefulness or efficacy of a procedure or treatment |
| Class IIa: weight of evidence or opinion favors usefulness or efficacy |
| Class IIb: usefulness or efficacy is less well established by evidence or opinion |
| Class III: conditions for which there is evidence or general agreement that a given procedure or treatment is not useful or effective, and in some cases may be harmful |
ACC = American College of Cardiology; AHA = American Heart Association; UA/NSTEMI = unstable angina and non–ST-segment elevation myocardial infarction. Information from reference 3.
Initial Evaluation and Management
Two important issues arise in the initial evaluation of the patient with a suspected acute coronary syndrome: the likelihood that the clinical presentation represents an acute coronary syndrome (Table 2)3,5 and the risk of adverse outcomes (Table 3).3,5 The initial clinical evaluation to address both issues should include a history, a physical examination, an electrocardiogram, and a cardiac biomarker measurement (a cardiac-specific troponin level [preferred in the ACC/AHA guideline2,3] or an MB isoenzyme of creatine kinase level). Data from this evaluation aid in the assessment of risk and in decisions about the required intensity of monitoring (intensive care unit versus “step-down” unit), choice of therapeutic agents, and use of cardiac catheterization and revascularization.
RISK PREDICTION RULE
The 2002 ACC/AHA guideline2,3 includes the use of a risk prediction rule for early assessment. Multiple risk scores have been developed to predict the likelihood of adverse outcomes in patients presenting with UA/NSTEMI.6–8 One example is the seven-point Thrombolysis in Myocardial Infarction (TIMI) risk score for UA/NSTEMI (Figure 1).6
Figure 1
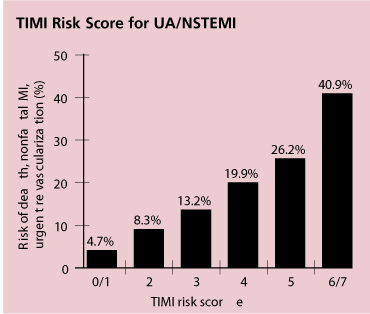
Thrombolysis in Myocardial Infarction (TIMI) risk score for unstable angina and non–ST-segment elevation myocardial infarction (UA/NSTEMI). Adapted with permission from Antman EM, Cohen M, Bernink PJ, McCabe CH, Horacek T, Papuchis G, et al. The TIMI risk score for unstable angina/non-ST elevation MI: a method for prognostication and therapeutic decision making. JAMA 2000;284:835–42.
The TIMI risk score integrates historical factors, frequency of symptoms, electrocardiographic findings, and cardiac biomarker levels.6 Higher scores are associated with an increased risk of adverse outcomes such as death, (re)infarction, or recurrent ischemia requiring revascularization. The risk of these outcomes ranges from approximately 5 percent with a TIMI risk score of zero or one point to approximately 41 percent with a risk score of six or seven points. The risk score may be used to help guide therapeutic decisions. Patients with higher risk scores have been shown to derive greater benefit from specific pharmacologic therapies (enoxaparin [Lovenox],6 platelet glycoprotein IIb/IIIa inhibitor9) and an early cardiac catheterization (invasive) strategy.10
BIOMARKERS
Recent studies have examined the role of nontraditional biomarkers in the risk stratification of patients with acute coronary syndrome. High-sensitivity C-reactive protein (hs-CRP), a marker of inflammation, has been shown to provide prognostic information in patients with acute coronary syndromes, independent of clinical factors and traditional markers of necrosis.11–13 B-type natriuretic peptide (BNP) has been associated with heart failure, as well as adverse clinical outcomes (predominantly mortality), in patients with acute coronary syndromes.14 The study findings11–14 suggest that future risk stratification in patients with acute coronary syndrome may involve a panel of biomarkers.
One investigative team15 has proposed a simplified method of combining the information provided by biomarkers. From zero to three points are assigned, depending on the number of elevated biomarkers (cardiac-specific troponin, hs-CRP, BNP). The risk of death, recurrent myocardial infarction, or congestive heart failure has been found to be 4.5 times higher when all three biomarkers are elevated than when no biomarker is elevated.15 However, more data are needed before use of hs-CRP and BNP can be recommended for risk stratification in UA/NSTEMI.
Hospital Care
Patients with UA/NSTEMI should be admitted to an inpatient unit, where they should undergo continuous monitoring for arrhythmias and recurrent ischemia. Patients with high-risk indicators, such as recurrent pain or hemodynamic disturbance (Table 4),3 should be admitted to a coronary care or intensive care unit capable of more extensive monitoring. Therapy should include anti-ischemic, antiplatelet, and antithrombotic agents, as well as a care plan that includes consideration of an early invasive strategy (Figure 2).3
TABLE 4 High-Risk Indicators Favoring an Early Invasive Strategy in Patients with UA/NSTEMI
| Recurrent angina or ischemia at rest or with low-level activity, despite intensive anti-ischemic therapy |
| Elevated cardiac-specific troponin level (troponin I or T) |
| New or presumably new ST-segment depression |
| Recurrent angina or ischemia with symptoms of congestive heart failure, an S3 gallop, pulmonary edema, worsening rales, or new or worsening mitral regurgitation |
| High-risk findings on noninvasive stress testing |
| Depressed left ventricular function (e.g., ejection fraction < 40% on noninvasive study) |
| Hemodynamic instability |
| Sustained ventricular tachycardia |
| Percutaneous coronary intervention within previous six months |
| Previous coronary artery bypass grafting |
UA/NSTEMI = unstable angina and non–ST-segment elevation myocardial infarction.
Information from reference 3.
Figure 2
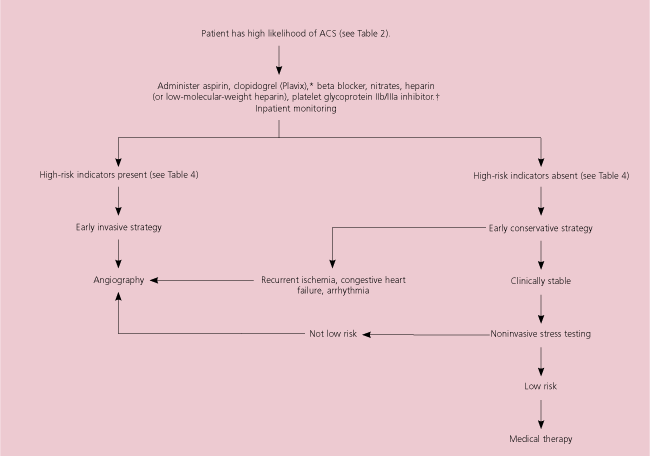
Evaluation and management of the patient with a high likelihood of having an acute coronary syndrome (ACS). (ACC = American College of Cardiology; AHA = American Heart Association) Adapted with permission from ACC/AHA 2002 guideline update for the management of patients with unstable angina and non-ST-segment elevation myocardial infarction. A report of the American College of Cardiology/American Heart Association Task Force on Practice Guidelines (Committee on the Management of Patients with Unstable Angina). Accessed online May 11, 2004, at: http://www.americanheart.org/presenter.jhtml?identifier=3001260.
ANTI-ISCHEMIC THERAPY
ACC/AHA class I anti-ischemic interventions include supplemental oxygen, sublingually or intravenously administered nitroglycerin for relief of recurrent ischemia and associated symptoms and, in the absence of contraindications, an intravenously administered beta blocker for management of ongoing chest pain followed by an orally administered beta blocker.2,3 Beta blockers should be used cautiously in patients with marked first-degree atrioventricular block, second- and third-degree atrioventricular block without a pacemaker, asthma, severe left ventricular dysfunction with congestive heart failure, significant chronic obstructive pulmonary disease, and significant sinus bradycardia or hypotension.2,3
ANTIPLATELET THERAPY
Three classes of antiplatelet agents have important roles in the management of UA/NSTEMI: aspirin, thienopyridines, and platelet glycoprotein IIb/IIIa inhibitors.
Aspirin
A cornerstone of management in acute coronary syndromes, aspirin has been shown to reduce cardiovascular events by 50 to 70 percent.16 In the absence of known contraindications, aspirin therapy should be used in all patients with suspected, probable, or definite acute coronary syndrome.
Clopidogrel
The updated ACC/AHA guideline2,3 recommends use of the thienopyridine clopidogrel (Plavix) in patients who cannot tolerate aspirin17 (ACC/AHA class I). Because of its safety profile (compared with ticlopidine [Ticlid]), clopidogrel currently is the preferred thienopyridine.1 Clopidogrel is a potent antiplatelet agent that acts by irreversibly blocking the P2Y12 adenosine diphosphate receptor on the platelet surface, thereby interrupting platelet activation and aggregation.
One of the major changes in the care of patients with UA/NSTEMI has been the ACC/AHA class I indication for use of clopidogrel in addition to aspirin in patients with acute coronary syndromes.2,3 This change occurred because of the findings of recent major clinical trials. The Clopidogrel in Unstable Angina to Prevent Recurrent Ischemic Events (CURE) trial18 randomized more than 12,000 patients with UA/NSTEMI to receive clopidogrel or placebo in addition to aspirin. Patients were followed for three to 12 months. In the CURE trial, death, myocardial infarction, or stroke occurred in 9.3 percent of the patients treated with clopidogrel, compared with 11.5 percent of those who received placebo. The improvement occurred at the cost of a small, but significant increase in bleeding (relative risk: 27 percent), especially in patients who underwent coronary artery bypass grafting within five days of discontinuing clopidogrel therapy.
In an analysis of patients undergoing percutaneous coronary intervention (PCI-CURE study),19 patients were treated with clopidogrel or placebo for a median of 10 days before the intervention (all patients also received aspirin). After the intervention, patients in the PCI-CURE study received open-label clopidogrel or ticlopidine for four weeks, followed by the initial study drug (clopidogrel or placebo) for an average of eight months. The clopidogrel-treated patients had fewer early (30-day) and long-term cardiovascular events.
As a result of the study findings, the 2002 ACC/AHA guideline2,3 considers the use of clopidogrel in addition to aspirin to have a class I indication in patients with UA/NSTEMI who are undergoing an early noninterventional or interventional approach and are not at high risk for bleeding. Clopidogrel therapy is recommended for at least one month and may be continued for up to nine months. Aspirin, unless contraindicaetd, should be continued for life. When elective coronary artery bypass grafting is planned, clopidogrel should be withheld for five to seven days.
Platelet Glycoprotein IIb/IIIa Inhibitors
These agents constitute a third class of antiplatelet agents that may be used in patients hospitalized with UA/NSTEMI. Three agents in this class currently are available for clinical use: abciximab (ReoPro), which is a monoclonal anti-body; and eptifibatide (Integrelin) and tirofiban (Aggrastat), which are “small molecule” glycoprotein IIb/IIIa inhibitors. These potent inhibitors of platelet aggregation are administered intravenously.
Clinical trials have shown that platelet glycoprotein IIb/IIIa inhibitor therapy is beneficial in selected patients with UA/NSTEMI.20 However, benefit appears to be greater in patients for whom an early invasive strategy is planned (i.e., cardiac catheterization and percutaneous coronary intervention)20,21 and patients who have elevated cardiac-specific troponin levels22 or other high-risk indicators such as an elevated TIMI risk score9 or diabetes mellitus.23 In patients for whom an early invasive strategy is not planned, the Global Utilization of Strategies to Open Occluded Coronary Arteries IV–Acute Coronary Syndromes (GUSTO IV-ACS) randomized trial24 showed no benefit for abciximab compared with placebo. As a result of the GUSTO IV-ACS study findings, use of abciximab is contraindicated in patients for whom an early invasive strategy is not planned (ACC/AHA class III).
Based on the combined study findings,20–23,25 platelet glycoprotein IIb/IIIa inhibitors have an ACC/AHA class I indication in patients for whom catheterization and percutaneous coronary intervention are planned. These agents should be administered in addition to aspirin and heparin. An ACC/AHA class IIa recommendation is given to the use of eptifibatide or tirofiban in addition to aspirin and heparin in patients with continuing ischemia, elevated cardiac-specific troponin levels, or other high-risk features for whom an invasive strategy is not planned.
ANTITHROMBOTIC THERAPY
The final component of medical therapy to consider in patients with UA/NSTEMI is antithrombotic/anticoagulant therapy. Unfractionated heparin results in anti-coagulation by facilitating the action of antithrombin which, in turn, inactivates factor IIa (thrombin), factor IXa, and factor Xa. Treatment with unfractionated heparin has been shown to be superior to aspirin therapy alone in patients with UA/NSTEMI.26 Low-molecular-weight (LMW) heparin (e.g., enoxaparin) is obtained through cleavage of heparin to provide chains with different molecular weights. Compared with unfractionated heparin, the LMW heparins are relatively more potent inhibitors of factor Xa. LMW heparins also have a more predictable pharmacology, which means that laboratory monitoring of anticoagulation status is not needed.26 Anticoagulation with unfractionated heparin or LMW heparin has an ACC/AHA class I indication.27 One of these agents is to be used in addition to aspirin or clopidogrel.
Recent trial data28 have shown superior results with the use of enoxaparin compared with unfractionated heparin in patients with UA/NSTEMI. Consequently, the guideline2,3 indicates a ACC/AHA class IIa recommendation for the use of enoxaparin, rather than unfractionated heparin, in patients with UA/NSTEMI. Although data are not conclusive, recent trials have shown that LMW heparin is safe to use in combination with glycoprotein IIb/IIIa inhibitors29,30 and in percutaneous coronary intervention.31–33

