
A more recent article on acute shoulder injuries in adults is available.
Am Fam Physician. 2004;70(10):1947-1954
The shoulder is the most mobile joint in the human body. The cost of such versatility is an increased risk of injury. It is important that family physicians understand the anatomy of the shoulder, mechanisms of injury, typical physical and radiologic findings, approach to management of injuries, and indications for referral. Clavicle fractures are among the most common acute shoulder injuries, and more than 80 percent of them can be managed conservatively. Humeral head fractures are less common and usually occur in elderly persons; 85 percent of them can be managed nonoperatively. Common acute soft tissue injuries include shoulder dislocations, rotator cuff tears, and acromioclavicular sprains. Acromioclavicular injuries are graded from types I to VI. Types I and II are treated conservatively, types IV to VI are treated surgically, and there is debate about the best approach for type III. Eighty percent of shoulder dislocations are anterior. Diagnosis of this injury is straightforward. The injury usually can be reduced by employing a number of nonsurgical techniques. Traumatic or acute rotator cuff tears can be managed conservatively or surgically, depending on the patient and the degree of injury.
| Key clinical recommendation | Label | References |
|---|---|---|
| Figure-of-eight bandages for clavicle fractures are more uncomfortable, have a higher incidence of complications, and do not improve outcomes when compared with an arm sling. | A | 4 |
| Surgical intervention in displaced proximal humerus fractures improves clinical outcomes. | B | 8 |
| In young athletes, surgical intervention for shoulder dislocation reduces the risk for recurrent dislocations and improves function. | A | 12–15 |
The shoulder includes the proximal humerus, the clavicle and the scapula, and their connections to each other, to the sternum (clavicle), and to the thoracic rib cage (scapula). Together, these elements form the most mobile joint in the human body (Figure 1). It allows the upper extremity to rotate up to 180 degrees in three different planes, enabling the arm to perform a versatile range of activities. This mobility comes at a cost: it leaves the shoulder prone to injury. Family physicians often encounter patients with shoulder injuries. It is important to understand the anatomy of the shoulder, mechanisms of injury, evaluation and management of injuries, and indications for referral.
Clavicle Fractures
The only bony connection between the axial skeleton and the upper extremity occurs through the clavicle, which is held securely in place by ligaments at the sternum and acromion. The clavicle overlies and protects the brachial plexus, pleural cap, and great vessels of the upper extremity. Clavicle fractures are among the most common injuries, accounting for one in 20 adult fractures.1 The injury usually is caused by a fall on the lateral shoulder or, less commonly, by a direct blow or by falling on an outstretched arm.
The clavicle is relatively superficial and easily palpable along its entire length. Clavicle fractures usually can be diagnosed by careful inspection and palpation. Acute complications are uncommon, although pneumothorax, hemothorax, and injuries to the brachial plexus or subclavian vessels have been reported.2 Neurovascular and lung examinations should be performed to screen for these complications. A routine anteroposterior view usually is the only radiograph needed to confirm the fracture and specify its location. Nondisplaced fractures, however, may be difficult to detect on an anteroposterior view, particularly in children. In such cases, a 20-degree (Zanca view) or 45-degree cephalic tilt view usually demonstrates the fracture.
Clavicle fractures are classified by Allman1 into three groups by dividing the clavicle into thirds. Group 1 (middle one third of the clavicle) is the most common type of break (Figure 2)3 and represents 80 percent of clavicle fractures.1 Group 1 fractures are treated conservatively with an arm sling for comfort, even if significant displacement is present. Historically, a figure-of-eight bandage was applied, but they are uncomfortable, have a higher incidence of complications, and do not improve functional or cosmetic results.4 Ice and analgesics are helpful in the initial treatment. Elbow range-of-motion exercises should be started as soon as pain permits. Shoulder range-of-motion and strengthening exercises should begin once the fracture heals.
Nondisplaced group 2 (lateral one third of the clavicle) fractures usually can be treated conservatively. Group 2 fractures that extend to the articular surface, even if nondisplaced, often lead to osteoarthritis of the acromioclavicular (AC) joint.5 Displaced group 2 fractures generally require operative treatment because they are unstable and have a high incidence of nonunion. Surgical treatment generally results in good function.6
Displaced group 3 fractures (medial one third of the clavicle) and sternoclavicular dislocations require orthopedic referral. These injuries have a fairly high rate of significant intrathoracic or neurovascular injury that may require emergency surgery. Nondisplaced group 3 fractures without associated injuries can be treated conservatively with a sling for comfort.
Proximal Humerus Fractures
Proximal humerus fractures occur most commonly in elderly persons. They usually result from a fall onto an outstretched arm. In young adults, direct blows are a more common cause. Up to 85 percent of proximal humerus fractures can be treated nonoperatively.2 Evaluation of a patient with a proximal humerus fracture starts with a careful and focused physical examination. Neurologic and vascular examinations of the upper extremity should be completed and documented. Occasionally, the axillary nerve or axillary artery may be injured; rarely, the brachial artery, brachial plexus, or another nerve may be injured. Identification of an anterior or posterior bulge may suggest a dislocation. Tenderness and swelling often are diffuse, making it difficult to detect clear point tenderness.
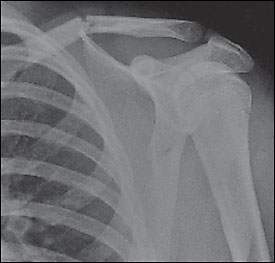
Appropriate radiographs are an important part of diagnosing and evaluating proximal humerus fractures. A standard shoulder series includes anteroposterior, transscapular (Y—Figure 3), and axillary views.3 Instead of a true shoulder series, radiologic technicians sometimes obtain only internal and external rotation views of the humerus. Although these views may demonstrate the fracture, they are suboptimal for detecting associated fractures and shoulder dislocations.
Because of its bony structure and the insertion of the rotator cuff tendons, the proximal head of the humerus generally fractures with four predictable cleavage lines (Figure 4).3 Regardless of the number of fragments, proximal humerus fractures are classified by the displacement and degree of angulation.7 Neer 1-part fractures have no more than 1-cm displacement of any fragment and no more than 45 degrees of angulation. More than 80 percent7 of proximal humerus fractures are nondisplaced (i.e., Neer 1-part fractures) and can be treated conservatively, if stable.2 Open fractures and fractures with neurologic or vascular deficits require emergent orthopedic referral. Patients with displaced proximal humerus fractures should be referred because surgical intervention appears to improve the outcome.8 Fracture-dislocations and fractures of the anatomic neck (indicated by the line just below the humeral head in (Figure 4)3 also should be referred.
Treatment of Neer 1-part fractures includes a sling for comfort and early range-of-motion exercises, which should be started as soon as tolerated (about five to 10 days after the injury). Patients should begin with pendulum exercises with the injured arm in the sling. They perform this movement by bending at the waist, allowing the arm to fall toward the floor, and rotating it in a circle. With time, the size of the circle is increased, and the sling is removed during the exercise.
After two to three weeks, abduction (progressively walking fingers up the wall) and internal rotation (first touching hip, then progressing to mid-back) exercises are added. Elderly patients tend to lose elbow range of motion rapidly. To avoid this, patients should be encouraged to remove the sling and flex and extend the elbow as soon as this movement can be tolerated. To speed recovery and avoid iatrogenic loss of elbow range of motion, use of the sling should be discontinued completely by four to six weeks after injury. Formal physical therapy can be helpful in maximizing future function of the shoulder.
Glenohumeral Dislocation
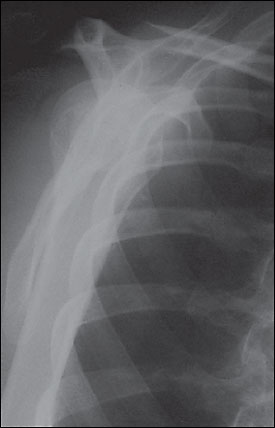
The glenohumeral joint is one of the most commonly dislocated joints. Ninety percent of shoulder dislocations are anterior (Figure 5),3 with most of the others being posterior.9 Inferior dislocations (luxatio erecta) are rare and often are accompanied by neurovascular injury and fracture. In younger patients, most shoulder dislocations are caused by direct trauma and sports injuries. In elderly persons, falls are the predominant cause, and the dislocation often is accompanied by a fracture. An excessive external rotation or abduction force usually causes anterior dislocations, while posterior dislocations usually occur when the humeral head is driven posteriorly with great force.
Patients with shoulder dislocations typically are unwilling to move the affected arm and tend to cradle it with the other arm. The diagnosis of an anterior dislocation often is suggested by simple inspection. A bulge may be noticeable where the humeral head rests, with emptiness beneath the acromion where the humeral head should be.
Management begins with a careful examination to rule out neurologic or vascular deficits. The standard three-view shoulder series should be obtained to ascertain or confirm the diagnosis and to rule out associated fractures. Muscle spasm sets in shortly after dislocation, making reduction more difficult. Therefore, reduction should be performed promptly after radiographs are obtained and interpreted. When evaluating patients in the field (e.g., at a sports event), many physicians perform the reduction before obtaining radiographs. This step provides prompt, dramatic relief from pain. Early reduction also requires less force, which should reduce the risk of iatrogenic injury. These benefits should offset the disadvantages of delaying radiographs. This is particularly true for recurrent dislocations, which are much less likely to have associated fractures. Radiographs should be obtained after reduction to rule out associated fractures that might demand specific treatment. In patients with chronic dislocations, radiographs may not be necessary.
Numerous reduction techniques are available.10 There is the self-reduction technique,11 in which the patient interlaces his or her fingers and places them around the flexed ipsilateral knee. The patient then leans backward, and the reduction occurs. This technique is similar mechanically to the traction against countertraction method (called the hippocratic method if done by a single physician). In this method, the physician grasps the patient’s affected arm by the wrist and applies traction at a 45-degree angle, while providing countertraction by placing a foot on the patient’s chest wall or by having an assistant wrap a sheet around the patient’s torso.
In the gravity method (modified Stimson’s method), the patient is placed prone with the affected shoulder supported and the arm hanging over the examination table with a weight attached to the hand. A weight of 2.25 kg (5 lb) usually is sufficient, but greater weight may be needed for larger or more muscular patients. Gravity slowly stretches the spasmed muscles, and the reduction occurs. Unless performed immediately after injury (on the field or by using self-reduction), all of these methods require significant analgesia, such as a narcotic plus benzodiazepine, with appropriate monitoring. Failure to reduce the dislocation successfully using these methods necessitates reduction under general anesthesia.
Treatment after reduction traditionally includes immobilization of the shoulder for four weeks followed by rehabilitation. The possible drawback of conservative treatment is a high incidence of recurrent dislocation (67 to 97 percent), with patients younger than 30 years being at highest risk.11 Multiple studies have shown that early surgical intervention, rather than conservative treatment (especially in the younger athletic population), provides a better outcome.12–15 Unfortunately, anterior shoulder dislocations generate a variety of injuries, making any general intervention recommendations inappropriate. The choice of surgical treatment depends on the soft tissue injury. Referral to an orthopedist is probably a good choice, particularly for younger or athletic patients. If conservative therapy is pursued, range of motion should be addressed in the same way as it is in patients with a humerus fracture. As range of motion improves, strengthening exercises should be added.
Acromioclavicular Sprains
The AC joint is a common site of injury in athletes and active persons. The classic cause of an AC joint injury is a direct blow to the acromion with the humerus in an adducted position. This force drives the acromion medially and inferiorly. AC joint injuries also may be caused by indirect trauma, such as falling on an outstretched arm or elbow. The ligaments that hold the AC joint together include the acromioclavicular, coracoclavicular, and deltotrapezial fascia. These ligaments can be sprained, disrupted, detached, or separated, depending on the severity of the injury.
The first ligament to be damaged is the AC ligament. With enough force, the coracoclavicular ligaments also will be torn, and the deltotrapezial fascia injured or detached. When all support is disrupted or detached, the distal clavicle loses all ligamentous connection, allowing it to rise superiorly. Associated fractures of the coracoid process or the lateral one third of the clavicle may occur, necessitating an orthopedic referral. The Rockwood classification for AC joint injuries includes six categories (Figure 6), depending on which ligaments were damaged and the position of the clavicle.
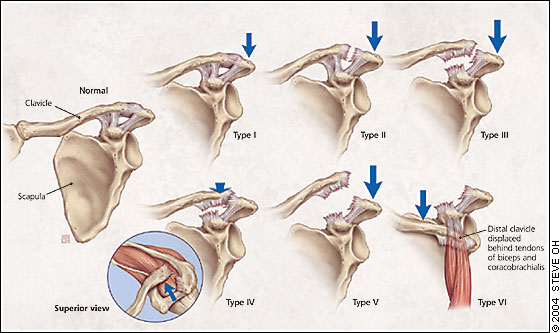
Patients with AC joint injuries should receive a physical examination and evaluation for radiographs. As with all acute injuries, neurovascular assessment should be included. If radiographs are obtained, the uninjured side also should be imaged for comparison. Weighted radiographs have been a part of the traditional work-up, but the need for them has come into question.16 Patients with type I injuries should have tenderness over the AC joint, no visible deformity of the distal clavicle, and normal radiographs. They also will have a positive cross-arm test (sharp pain at the AC joint if the patient holds the arm out straight and brings it across the chest).
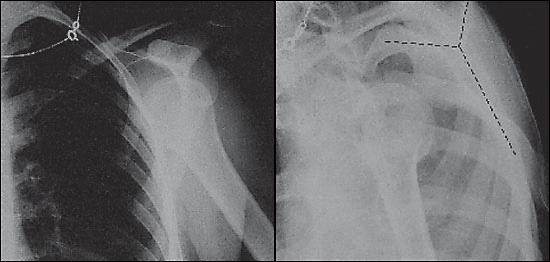
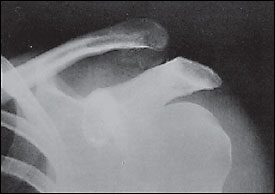
With type II injuries, the distal clavicle may be slightly more prominent on inspection, and the patient may have pain at the distal end of the clavicle from the sprained coracoclavicular ligament. Radiographs usually are normal but may show slight widening of the AC joint. Type III injuries usually present with obvious visible prominence of the distal clavicle. Radiographs (Figure 7) should demonstrate an obvious separation of the AC joint and an increase of the coracoclavicular distance of 25 to 100 percent compared with the normal side.17 Types IV to VI have grossly abnormal radiographs, and patients with these injuries should be referred immediately.
There is a general consensus that type I and II injuries are treated nonoperatively, while types IV, V, and VI are treated surgically. The treatment of type III injuries remains an area of controversy, and patients with these injuries should be referred to an orthopedist for evaluation and possible surgical repair.
Treatment of type I and II injuries focuses on symptomatic relief and includes use of a sling for one to three weeks, ice, and nonsteroidal anti-inflammatory drugs (NSAIDs). Once the acute pain is alleviated, range-of-motion and general strengthening exercises are started. Athletes may return to sports when pain-free function is restored. Most patients are able to return to sports, but some may complain of nuisance symptoms such as clicking and pain with push-ups. Nonoperative treatment of type III injuries is similar to that for types I and II, except that rest in a sling lasts for two to four weeks.
Rotator Cuff Tears
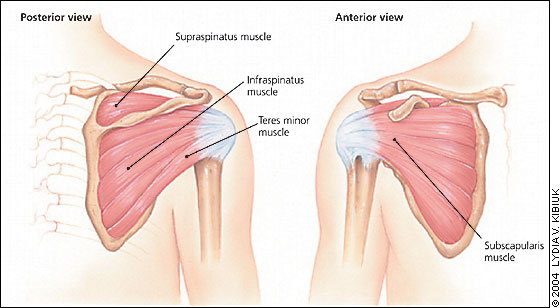
The rotator cuff comprises four muscles, the supraspinatus, infraspinatus, teres minor, and subscapularis (Figure 8). The rotator cuff is the support structure for the shoulder joint. When a tear occurs, patients experience pain and difficulty in abducting or rotating the arm. The incidence of traumatic rotator cuff tears is unknown,18 but it is thought to be relatively uncommon in younger athletes. Rotator cuff tears cover a spectrum of injuries. At one end are relatively high-force injuries to a healthy rotator cuff that typically produce full-thickness tears. The degree of trauma usually required would be a significant fall, motor vehicle crash, or shoulder dislocation. Lesser degrees of trauma can cause partial-thickness tears. On the other end of the spectrum are overuse injuries. Traumatic rotator cuff tears can occur at any age. Overuse injuries generally occur in athletes and increase in frequency with advancing age.19
The diagnosis of an acute rotator cuff tear is based on the history of the injury, the physical examination findings, and magnetic resonance imaging (MRI) when necessary. Pain and weakness usually are made worse by overhead activities and are present in abduction and external rotation. MRI has become the imaging standard and has replaced invasive arthrography to assist in the diagnosis.20
Initially, conservative treatment is acceptable for rotator cuff tears, but the choice of an operative versus nonoperative approach should be individualized for each patient. Young active patients with acute full-thickness tears and severe functional deficit should be referred quickly to an orthopedic surgeon for possible operative repair.19 Competitive athletes generally do better with surgical repair, no matter what their age.13 A Cochrane review of surgical, nonsurgical, conservative, and pharmaceutical (oral and injection) treatments for rotator cuff tears provides little evidence to suggest which treatment course is best.21
Conservative treatment starts with relative rest and restriction of movement; in particular, limiting the possibility of repeating the initial injury by avoiding athletic activities. The rest period should be short (one to two weeks) and should include icing, NSAIDs, and, occasionally, subacromial steroid injections. Gentle range-of-motion exercises (pendulum exercises) should be initiated during the rest period to retard loss of range of motion. After the pain has subsided, a monitored stretching and strengthening program should be started. This program should be supervised by a physical therapist or a sports trainer with substantial knowledge of shoulder rehabilitation.
The success rate of conservative treatment is unclear.19 There are concerns that delaying repair of a rotator cuff tear makes the eventual repair harder or impossible, and that initial surgical repair generally has a good outcome.13 The choice of treatment is clearly an individual one that should be made with input from the patient.