A complete urinalysis includes physical, chemical, and microscopic examinations. Midstream clean collection is acceptable in most situations, but the specimen should be examined within two hours of collection. Cloudy urine often is a result of precipitated phosphate crystals in alkaline urine, but pyuria also can be the cause. A strong odor may be the result of a concentrated specimen rather than a urinary tract infection. Dipstick urinalysis is convenient, but false-positive and false-negative results can occur. Specific gravity provides a reliable assessment of the patient’s hydration status. Microhematuria has a range of causes, from benign to life threatening. Glomerular, renal, and urologic causes of microhematuria often can be differentiated by other elements of the urinalysis. Although transient proteinuria typically is a benign condition, persistent proteinuria requires further work-up. Uncomplicated urinary tract infections diagnosed by positive leukocyte esterase and nitrite tests can be treated without culture.
Urinalysis is invaluable in the diagnosis of urologic conditions such as calculi, urinary tract infection (UTI), and malignancy. It also can alert the physician to the presence of systemic disease affecting the kidneys. Although urinalysis is not recommended as a routine screening tool except in women who may be pregnant, physicians should know how to interpret urinalysis results correctly. This article reviews the correct method for performing urinalysis and the differential diagnosis for several abnormal results.
Strength of Recommendations
| Key clinical recommendation | Label | References |
|---|---|---|
| Patients with dipstick results of 3+ or greater may have significant proteinuria; further work-up is indicated. | B | 5 |
| Patients with microscopic hematuria (i.e., at least three red blood cells per high-power field in two of three specimens) should be evaluated to exclude renal and urinary tract disease. | C | 19,20 |
| Exercise-induced hematuria is a relatively common, self-limited, and benign condition. Because results of repeat urinalysis after 48 to 72 hours should be negative in patients with this condition, extended testing is not warranted. | C | 30 |
A = consistent, good-quality patient-oriented evidence; B = inconsistent or limited-quality patient-oriented evidence; C = consensus, disease-oriented evidence, usual practice, opinion, or case series. See page 1046 for more information.
Specimen Collection
A midstream clean-catch technique usually is adequate in men and women. Although prior cleansing of the external genitalia often is recommended in women, it has no proven benefit. In fact, a recent study1 found that contamination rates were similar in specimens obtained with and without prior cleansing (32 versus 29 percent). Urine must be refrigerated if it cannot be examined promptly; delays of more than two hours between collection and examination often cause unreliable results.2
Physical Properties: Color and Odor
Foods, medications, metabolic products, and infection can cause abnormal urine colors (Table 1).3 Cloudy urine often is a result of precipitated phosphate crystals in alkaline urine, but pyuria also can be the cause.
The normal odor of urine is described as urinoid; this odor can be strong in concentrated specimens but does not imply infection. Diabetic ketoacidosis can cause urine to have a fruity or sweet odor, and alkaline fermentation can cause an ammoniacal odor after prolonged bladder retention. Persons with UTIs often have urine with a pungent odor. Other causes of abnormal odors include gastrointestinal-bladder fistulas (associated with a fecal smell), cystine decomposition (associated with a sulfuric smell), and medications and diet (e.g., asparagus).
TABLE 1 Common Causes of Abnormal Urine Coloration
| Color | Pathologic causes | Food and drug causes |
|---|---|---|
| Cloudy | Phosphaturia, pyuria, chyluria, lipiduria, hyperoxaluria | Diet high in purine-rich foods (hyperuricosuria) |
| Brown | Bile pigments, myoglobin | Fava beans |
| Levodopa (Larodopa), metronidazole (Flagyl), nitrofurantoin (Furadantin), some antimalarial agents | ||
| Brownish-black | Bile pigments, melanin, methemoglobin | Cascara, levodopa, methyldopa (Aldomet), senna |
| Green or blue | Pseudomonal UTI, biliverdin | Amitriptyline (Elavil), indigo carmine, IV cimetidine (Tagamet), IV promethazine (Phenergan), methylene blue, triamterene (Dyrenium) |
| Orange | Bile pigments | Phenothiazines, phenazopyridine (Pyridium) |
| Red | Hematuria, hemoglobinuria, myoglobinuria, porphyria | Beets, blackberries, rhubarb |
| Phenolphthalein, rifampin (Rifadin) | ||
| Yellow | Concentrated urine | Carrots |
| Cascara |
UTI = urinary tract infection; IV = intravenous.
Adapted with permission from Hanno PM, Wein AJ, Malkowicz SB. Clinical manual of urology. 3d ed. New York: McGraw-Hill, 2001:75.
Dipstick Urinalysis
False-positive and false-negative results are not unusual in dipstick urinalysis (Table 2). The accuracy of this test in detecting microscopic hematuria, significant proteinuria, and UTI is summarized in Table 3.4–13
TABLE 2 Causes of False-Positive and False-Negative Urinalysis Results
| Dipstick test | False positive | False negative |
|---|---|---|
| Bilirubin | Phenazopyridine (Pyridium) | Chlorpromazine (Thorazine), selenium |
| Blood | Dehydration, exercise, hemoglobinuria, menstrual blood, myoglobinuria | Captopril (Capoten), elevated specific gravity, pH < 5.1, proteinuria, vitamin C |
| Glucose | Ketones, levodopa (Larodopa) | Elevated specific gravity, uric acid, vitamin C |
| Ketones | Acidic urine, elevated specific gravity, mesna (Mesnex), phenolphthalein, some drug metabolites (e.g., levodopa) | Delay in examination of urine |
| Leukocyte esterase | Contamination | Elevated specific gravity, glycosuria, ketonuria, proteinuria, some oxidizing drugs (cephalexin [Keflex], nitrofurantoin [Furadantin], tetracycline, gentamicin), vitamin C |
| Nitrites | Contamination, exposure of dipstick to air, phenazopyridine | Elevated specific gravity, elevated urobilinogen levels, nitrate reductase-negative bacteria, pH < 6.0, vitamin C |
| Protein | Alkaline or concentrated urine, phenazopyridine, quaternary ammonia compounds | Acidic or dilute urine, primary protein is not albumin |
| Specific gravity* | Dextran solutions, IV radiopaque dyes, proteinuria | Alkaline urine |
| Urobilinogen | Elevated nitrite levels, phenazopyridine | — |
IV = intravenous.
*—False-positive results are caused by false elevation; false-negative results are caused by false depression.
TABLE 3 Accuracy of Urinalysis for Disease Detection
| Condition | Test | Results | Sensitivity (%) | Specificity (%) | PPV | NPV |
|---|---|---|---|---|---|---|
| Microscopic hematuria4 | Dipstick | ≥ 1+ blood | 91 to 100 | 65 to 99 | NA | NA |
| Significant proteinuria5 | Dipstick | ≥ 3+ protein | 96 | 87 | NA | NA |
| Culture-confirmed UTI6–13 | Dipstick | Abnormal leukocyte esterase | 72 to 97 | 41 to 86 | 43 to 56 | 82 to 91 |
| Abnormal nitrites | 19 to 48 | 92 to 100 | 50 to 83 | 70 to 88 | ||
| Abnormal leukocyte esterase or nitrites | 46 to 100 | 42 to 98 | 52 to 68 | 78 to 98 | ||
| ≥ 3+ protein | 63 to 83 | 50 to 53 | 53 | 82 | ||
| ≥ 1+ blood | 68 to 92 | 42 to 46 | 51 | 88 | ||
| Any of the above abnormalities | 94 to 100 | 14 to 26 | 44 | 100 | ||
| Microscopy | > 5 WBCs per HPF | 90 to 96 | 47 to 50 | 56 to 59 | 83 to 95 | |
| > 5 RBCs per HPF | 18 to 44 | 88 to 89 | 27 | 82 | ||
| Bacteria (any amount) | 46 to 58 | 89 to 94 | 54 to 88 | 77 to 86 |
PPV = positive predictive value; NPV = negative predictive value; NA = not applicable; UTI = urinary tract infection; WBCs = white blood cells; HPF = high-powered field; RBCs = red blood cells.
SPECIFIC GRAVITY
Urinary specific gravity (USG) correlates with urine osmolality and gives important insight into the patient’s hydration status. It also reflects the concentrating ability of the kidneys. Normal USG can range from 1.003 to 1.030; a value of less than 1.010 indicates relative hydration, and a value greater than 1.020 indicates relative dehydration.14 Increased USG is associated with glycosuria and the syndrome of inappropriate antidiuretic hormone; decreased USG is associated with diuretic use, diabetes insipidus, adrenal insufficiency, aldosteronism, and impaired renal function.14 In patients with intrinsic renal insufficiency, USG is fixed at 1.010—the specific gravity of the glomerular filtrate.
URINARY PH
Urinary pH can range from 4.5 to 8 but normally is slightly acidic (i.e., 5.5 to 6.5) because of metabolic activity. Ingestion of proteins and acidic fruits (e.g., cranberries) can cause acidic urine, and diets high in citrate can cause alkaline urine.15–17 Urinary pH generally reflects the serum pH, except in patients with renal tubular acidosis (RTA). The inability to acidify urine to a pH of less than 5.5 despite an overnight fast and administration of an acid load is the hallmark of RTA. In type I (distal) RTA, the serum is acidic but the urine is alkaline, secondary to an inability to secrete protons into the urine. Type II (proximal) RTA is characterized by an inability to reabsorb bicarbonate. This situation initially results in alkaline urine, but as the filtered load of bicarbonate decreases, the urine becomes more acidic.
Determination of urinary pH is useful in the diagnosis and management of UTIs and calculi. Alkaline urine in a patient with a UTI suggests the presence of a urea-splitting organism, which may be associated with magnesium-ammonium phosphate crystals and can form staghorn calculi. Uric acid calculi are associated with acidic urine.
HEMATURIA
According to the American Urological Association, the presence of three or more red blood cells (RBCs) per high-powered field (HPF) in two of three urine samples is the generally accepted definition of hematuria.18–20 The dipstick test for blood detects the peroxidase activity of erythrocytes. However, myoglobin and hemoglobin also will catalyze this reaction, so a positive test result may indicate hematuria, myoglobinuria, or hemoglobinuria. Visualization of intact erythrocytes on microscopic examination of the urinary sediment can distinguish hematuria from other conditions. Microscopic examination also may detect RBC casts or dysmorphic RBCs. Hematuria is divided into glomerular, renal (i.e., nonglomerular), and urologic etiologies (Table 4).21
TABLE 4 Common Causes of Hematuria
| Glomerular causes | ||
| Familial causes | ||
| Fabry’s disease | ||
| Hereditary nephritis (Alport’s syndrome) | ||
| Nail-patella syndrome | ||
| Thin basement-membrane disease | ||
| Primary glomerulonephritis | ||
| Focal segmental glomerulonephritis | ||
| Goodpasture’s disease | ||
| Henoch-Schönlein purpura | ||
| IgA nephropathy (Berger’s disease) | ||
| Mesangioproliferative glomerulonephritis | ||
| Postinfectious glomerulonephritis | ||
| Rapidly progressive glomerulonephritis | ||
| Secondary glomerulonephritis | ||
| Hemolytic-uremic syndrome | ||
| Systemic lupus nephritis | ||
| Thrombotic thrombocytopenic purpura | ||
| Vasculitis | ||
| Renal causes | ||
| Arteriovenous malformation | ||
| Hypercalciuria | ||
| Hyperuricosuria | ||
| Loin pain-hematuria syndrome | ||
| Malignant hypertension | ||
| Medullary sponge kidney | ||
| Metabolic causes | ||
| Papillary necrosis | ||
| Polycystic kidney disease | ||
| Renal artery embolism | ||
| Renal vein thrombosis | ||
| Sickle cell disease or trait | ||
| Tubulointerstitial causes | ||
| Vascular cause | ||
| Urologic causes | ||
| Benign prostatic hyperplasia | ||
| Cancer (kidney, ureteral, bladder, prostate, and urethral) | ||
| Cystitis/pyelonephritis | ||
| Nephrolithiasis | ||
| Prostatitis | ||
| Schistosoma haematobium infection | ||
| Tuberculosis | ||
| Other causes | ||
| Drugs (e.g., NSAIDs, heparin, warfarin [Coumadin], cyclophosphamide [Cytoxan]) | ||
| Trauma (e.g., contact sports, running, Foley catheter) | ||
NSAIDs = nonsteroidal anti-inflammatory drugs.
Adapted with permission from Ahmed Z, Lee J. Asymptomatic urinary abnormalities. Hematuria and proteinuria. Med Clin North Am 1997;81:644.
Glomerular Hematuria
Glomerular hematuria typically is associated with significant proteinuria, erythrocyte casts, and dysmorphic RBCs. However, 20 percent of patients with biopsy-proven glomerulonephritis present with hematuria alone.22 IgA nephropathy (i.e., Berger’s disease) is the most common cause of glomerular hematuria.
Renal (Nonglomerular) Hematuria
Nonglomerular hematuria is secondary to tubulointerstitial, renovascular, or metabolic disorders. Like glomerular hematuria, it often is associated with significant proteinuria; however, there are no associated dysmorphic RBCs or erythrocyte casts. Further evaluation of patients with glomerular and nonglomerular hematuria should include determination of renal function and 24-hour urinary protein or spot urinary protein-creatinine ratio.
Urologic Hematuria
Urologic causes of hematuria include tumors, calculi, and infections. Urologic hematuria is distinguished from other etiologies by the absence of proteinuria, dysmorphic RBCs, and erythrocyte casts. Even significant hematuria will not elevate the protein concentration to the 2+ to 3+ range on the dipstick test.23 Up to 20 percent of patients with gross hematuria have urinary tract malignancy; a full work-up with cystoscopy and upper-tract imaging is indicated in patients with this condition.24 In patients with asymptomatic microscopic hematuria (without proteinuria or pyuria), 5 to 22 percent have serious urologic disease, and 0.5 to 5 percent have a genitourinary malignancy.25–29
Exercise-induced hematuria is a relatively common, benign condition that often is associated with long-distance running. Results of repeat urinalysis after 48 to 72 hours should be negative in patients with this condition.30
PROTEINURIA
In healthy persons, the glomerular capillary wall is permeable only to substances with a molecular weight of less than 20,000 Daltons. Once filtered, low–molecular-weight proteins are reabsorbed and metabolized by the proximal tubule cells. Normal urinary proteins include albumin, serum globulins, and proteins secreted by the nephron. Proteinuria is defined as urinary protein excretion of more than 150 mg per day (10 to 20 mg per dL) and is the hallmark of renal disease. Microalbuminuria is defined as the excretion of 30 to 150 mg of protein per day and is a sign of early renal disease, particularly in diabetic patients.
The reagent on most dipstick tests is sensitive to albumin but may not detect low concentrations of γ-globulins and Bence Jones proteins. Dipstick tests for trace amounts of protein yield positive results at concentrations of 5 to 10 mg per dL—lower than the threshold for clinically significant proteinuria.15 A result of 1+ corresponds to approximately 30 mg of protein per dL and is considered positive; 2+ corresponds to 100 mg per dL, 3+ to 300 mg per dL, and 4+ to 1,000 mg per dL.31,32 Dipstick urinalysis reliably can predict albuminuria with sensitivities and specificities of greater than 99 percent.4 Asymptomatic proteinuria is associated with significant renal disease in less than 1.5 percent of patients.4,33
Proteinuria can be classified as transient or persistent (Table 5).21 In transient proteinuria, a temporary change in glomerular hemodynamics causes the protein excess; these conditions follow a benign, self-limited course.34,35 Orthostatic (postural) proteinuria is a benign condition that can result from prolonged standing; it is confirmed by obtaining a negative urinalysis result after eight hours of recumbency.
TABLE 5 Common Causes of Proteinuria
| Transient proteinuria | ||
| Congestive heart failure | ||
| Dehydration | ||
| Emotional stress | ||
| Exercise | ||
| Fever | ||
| Orthostatic (postural) proteinuria | ||
| Seizures | ||
| Persistent proteinuria | ||
| Primary glomerular causes | ||
| Focal segmental glomerulonephritis | ||
| IgA nephropathy (i.e., Berger’s disease) | ||
| IgM nephropathy | ||
| Membranoproliferative glomerulonephritis | ||
| Membranous nephropathy | ||
| Minimal change disease | ||
| Secondary glomerular causes | ||
| Alport’s syndrome | ||
| Amyloidosis | ||
| Collagen vascular diseases (e.g., systemic lupus erythematosus) | ||
| Diabetes mellitus | ||
| Drugs (e.g., NSAIDs, penicillamine [Cuprimine], gold, ACE inhibitors) | ||
| Fabry’s disease | ||
| Infections (e.g., HIV, syphilis, hepatitis, post-streptococcal infection) | ||
| Malignancies (e.g., lymphoma, solid tumors) | ||
| Sarcoidosis | ||
| Sickle cell disease | ||
| Tubular causes | ||
| Aminoaciduria | ||
| Drugs (e.g., NSAIDs, antibiotics) | ||
| Fanconi syndrome | ||
| Heavy metal ingestion | ||
| Hypertensive nephrosclerosis | ||
| Interstitial nephritis | ||
| Overflow causes | ||
| Hemoglobinuria | ||
| Multiple myeloma | ||
| Myoglobinuria | ||
NSAIDs = nonsteroidal anti-inflammatory drugs; ACE = angiotensin-converting enzyme; HIV = human immunodeficiency virus.
Adapted with permission from Ahmed Z, Lee J. Asymptomatic urinary abnormalities. Hematuria and proteinuria. Med Clin North Am 1997;81:650.
Persistent proteinuria is divided into three general categories: glomerular, tubular, and overflow. In glomerular proteinuria, the most common type, albumin is the primary urinary protein. Tubular proteinuria results when malfunctioning tubule cells no longer metabolize or reabsorb normally filtered protein. In this condition, low–molecular-weight proteins predominate over albumin and rarely exceed 2 g per day. In overflow proteinuria, low–molecular-weight proteins overwhelm the ability of the tubules to reabsorb filtered proteins.
Further evaluation of persistent proteinuria usually includes determination of 24-hour urinary protein excretion or spot urinary protein-creatinine ratio, microscopic examination of the urinary sediment, urinary protein electrophoresis, and assessment of renal function.32
GLYCOSURIA
Glucose normally is filtered by the glomerulus, but it is almost completely reabsorbed in the proximal tubule. Glycosuria occurs when the filtered load of glucose exceeds the ability of the tubule to reabsorb it (i.e., 180 to 200 mg per dL). Etiologies include diabetes mellitus, Cushing’s syndrome, liver and pancreatic disease, and Fanconi’s syndrome.
KETONURIA
Ketones, products of body fat metabolism, normally are not found in urine. Dipstick reagents detect acetic acid through a reaction with sodium nitroprusside or nitro-ferricyanide and glycine. Ketonuria most commonly is associated with uncontrolled diabetes, but it also can occur during pregnancy, carbohydrate-free diets, and starvation.
NITRITES
Nitrites normally are not found in urine but result when bacteria reduce urinary nitrates to nitrites. Many gram-negative and some gram-positive organisms are capable of this conversion, and a positive dipstick nitrite test indicates that these organisms are present in significant numbers (i.e., more than 10,000 per mL). This test is specific but not highly sensitive. Thus, a positive result is helpful, but a negative result does not rule out UTI.6 The nitrite dipstick reagent is sensitive to air exposure, so containers should be closed immediately after removing a strip. After one week of exposure, one third of strips give false-positive results, and after two weeks, three fourths give false-positive results.36 Non-nitrate–reducing organisms also may cause false-negative results, and patients who consume a low-nitrate diet may have false-negative results.
LEUKOCYTE ESTERASE
Leukocyte esterase is produced by neutrophils and may signal pyuria associated with UTI. [ corrected] To detect significant pyuria accurately, 30 seconds to two minutes should be allowed for the dipstick reagent strip to change color, depending in the brand used. Leukocyte casts in the urinary sediment can help localize the area of inflammation to the kidney.
Organisms such as Chlamydia and Ureaplasma urealyticum should be considered in patients with pyuria and negative cultures. Other causes of sterile pyuria include balanitis, urethritis, tuberculosis, bladder tumors, viral infections, nephrolithiasis, foreign bodies, exercise, glomerulonephritis, and corticosteroid and cyclophosphamide (Cytoxan) use.
BILIRUBIN AND UROBILINOGEN
Urine normally does not contain detectable amounts of bilirubin. Unconjugated bilirubin is water insoluble and cannot pass through the glomerulus; conjugated bilirubin is water soluble and indicates further evaluation for liver dysfunction and biliary obstruction when it is detected in the urine.
Normal urine contains only small amounts of urobilinogen, the end product of conjugated bilirubin after it has passed through the bile ducts and been metabolized in the intestine. Urobilinogen is reabsorbed into the portal circulation, and a small amount eventually is filtered by the glomerulus. Hemolysis and hepatocellular disease can elevate urobilinogen levels, and antibiotic use and bile duct obstruction can decrease urobilinogen levels.
Microscopic Urinalysis
Microscopic examination is an indispensable part of urinalysis; the identification of casts, cells, crystals, and bacteria aids in the diagnosis of a variety of conditions. To prepare a urine specimen for microscopic analysis, a fresh sample of 10 to 15 mL of urine should be centrifuged at 1,500 to 3,000 rpm for five minutes. The supernatant then is decanted and the sediment resuspended in the remaining liquid.37 A single drop is transferred to a clean glass slide, and a cover slip is applied.
CELLS
Leukocytes may be seen under low- and high-power magnification (Figure 1). Men normally have fewer than two white blood cells (WBCs) per HPF; women normally have fewer than five WBCs per HPF.
Figure 1.
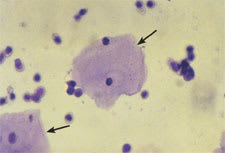
Squamous epithelial cells (arrows) and leukocytes (200 X).
Epithelial cells often are present in the urinary sediment. Squamous epithelial cells are large and irregularly shaped, with a small nucleus and fine granular cytoplasm; their presence suggests contamination. The presence of transitional epithelial cells is normal. These cells are smaller and rounder than squamous cells, and they have larger nuclei. The presence of renal tubule cells indicates significant renal pathology (Figure 2). Erythrocytes are best visualized under high-power magnification. Dysmorphic erythrocytes, which have odd shapes because of their passage through an abnormal glomerulus, suggest glomerular disease.
Figure 2.
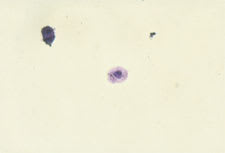
Convoluted renal tubule cells (200 X).
CASTS
Casts in the urinary sediment may be used to localize disease to a specific location in the genitourinary tract (Table 6).38 Casts, which are a coagulum of Tamm-Horsfall mucoprotein and the trapped contents of tubule lumen, originate from the distal convoluted tubule or collecting duct during periods of urinary concentration or stasis, or when urinary pH is very low. Their cylindrical shape reflects the tubule in which they were formed and is retained when the casts are washed away. The predominant cellular elements determine the type of cast: hyaline, erythrocyte, leukocyte, epithelial, granular, waxy, fatty, or broad (Figure 3).
TABLE 6 Urinary Casts and Associated Pathologic Conditions
| Type of cast | Composition | Associated conditions |
|---|---|---|
| Hyaline | Mucoproteins | Pyelonephritis, chronic renal disease |
| May be a normal finding | ||
| Erythrocyte | Red blood cells | Glomerulonephritis |
| May be a normal finding in patients who play contact sports | ||
| Leukocyte | White blood cells | Pyelonephritis, glomerulonephritis, interstitial nephritis, renal inflammatory processes |
| Epithelial | Renal tubule cells | Acute tubular necrosis, interstitial nephritis, eclampsia, nephritic syndrome, allograft rejection, heavy metal ingestion, renal disease |
| Granular | Various cell types | Advanced renal disease |
| Waxy | Various cell types | Advanced renal disease |
| Fatty | Lipid-laden renal tubule cells | Nephrotic syndrome, renal disease, hypothyroidism |
| Broad | Various cell types | End-stage renal disease |
Information from reference 38.
Figure 3.
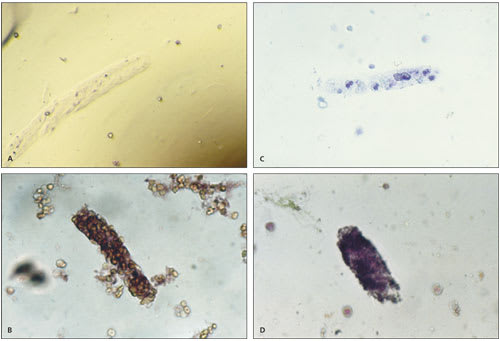
Urinary casts. (A) Hyaline cast (200 X); (B) erythrocyte cast (100 X); (C) leukocyte cast (100 X); (D) granular cast (100 X).
CRYSTALS
Crystals may be seen in the urinary sediment of healthy patients (Figure 4). Calcium oxalate crystals have a refractile square “envelope” shape that can vary in size. Uric acid crystals are yellow to orange-brown and may be diamond- or barrel-shaped. Triple phosphate crystals may be normal but often are associated with alkaline urine and UTI (typically associated with Proteus species). These crystals are colorless and have a characteristic “coffin lid” appearance. Cystine crystals are colorless, have a hexagonal shape, and are present in acidic urine, which is diagnostic of cystinuria.
Figure 4.
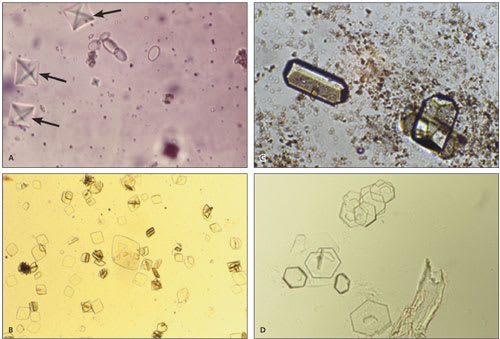
Urinary crystals. (A) Calcium oxalate crystals (arrows; 100 X); (B) uric acid crystals (100 X); (C) triple phosphate crystals with amorphous phosphates (400 X); (D) cystine crystals (100 X).
BACTERIURIA
[ corrected] Under high-powered magnification, gram-negative rods, streptococci, and staphylococci can be distinguished by their characteristic appearance.
Gram staining can help guide antibiotic therapy, but it is not indicated in routine outpatient practice. Clean-catch specimens from female patients frequently are contaminated by vaginal flora. In these patients, five bacteria per HPF represents roughly 100,000 colony-forming units (CFU) per mL, the classic diagnostic criterion for asymptomatic bacteriuria and certainly compatible with a UTI. In symptomatic patients, a colony count as low as 100 CFU per mL suggests UTI, and antibiotics should be considered. The presence of bacteria in a properly collected male urine specimen is suggestive of infection, and a culture should be obtained.

