The term “acute coronary syndrome” encompasses a range of thrombotic coronary artery diseases, including unstable angina and both ST-segment elevation and non–ST-segment elevation myocardial infarction. Diagnosis requires an electrocardiogram and a careful review for signs and symptoms of cardiac ischemia. In acute coronary syndrome, common electrocardiographic abnormalities include T-wave tenting or inversion, ST-segment elevation or depression (including J-point elevation in multiple leads), and pathologic Q waves. Risk stratification allows appropriate referral of patients to a chest pain center or emergency department, where cardiac enzyme levels can be assessed. Most high-risk patients should be hospitalized. Intermediate-risk patients should undergo a structured evaluation, often in a chest pain unit. Many low-risk patients can be discharged with appropriate follow-up. Troponin T or I generally is the most sensitive determinant of acute coronary syndrome, although the MB isoenzyme of creatine kinase also is used. Early markers of acute ischemia include myoglobin and creatine kinase–MB subforms (or isoforms), when available. In the future, advanced diagnostic modalities, such as myocardial perfusion imaging, may have a role in reducing unnecessary hospitalizations.
Acute coronary syndrome encompasses a spectrum of coronary artery diseases, including unstable angina, ST-elevation myocardial infarction (STEMI; often referred to as “Q-wave myocardial infarction”), and non-STEMI (NSTEMI; often referred to as “non–Q-wave myocardial infarction”). The term “acute coronary syndrome” is useful because the initial presentation and early management of unstable angina, STEMI, and NSTEMI frequently are similar.
STRENGTH OF RECOMMENDATIONS
| Key clinical recommendation | Label | References |
|---|---|---|
| The likelihood of acute coronary syndrome (low, intermediate, high) should be determined in all patients who present with chest pain. | C | 9 |
| A 12-lead ECG should be obtained within 10 minutes of presentation in patients with ongoing chest pain. | C | 9 |
| Cardiac markers (troponin T, troponin I, and/or creatine kinase–MB isoenzyme of creatine kinase) should be measured in any patient who has chest pain consistent with acute coronary syndrome. | C | 9 |
| A normal electrocardiogram does not rule out acute coronary syndrome. | B | 12 |
| When used by trained physicians, the Acute Cardiac Ischemia Time-Insensitive Predictive Instrument (a computerized, decision-making program built into the electrocardiogram machine) results in a significant reduction in hospital admissions of patients who do not have acute coronary syndrome. | B | 26,27 |
A = consistent, good-quality patient-oriented evidence; B = inconsistent or limited-quality patient-oriented evidence; C = consensus, disease-oriented evidence, usual practice, opinion, or case series. For information about the SORT evidence rating system, see page 15 orhttps://www.aafp.org/afpsort.xml.
Differentiating acute coronary syndrome from noncardiac chest pain is the primary diagnostic challenge. The initial assessment requires a focused history (including risk factor analysis), a physical examination, an electrocardiogram (ECG) and, frequently, serum cardiac marker determinations (Table 1).1
TABLE 1 Risk Stratification to Determine the Likelihood of Acute Coronary Syndrome
| Assessment | Findings indicating HIGH likelihood of ACS | Findings indicating INTERMEDIATE likelihood of ACS in absence of high-likelihood findings | Findings indicating LOW likelihood of ACS in absence of high- or intermediate-likelihood findings |
|---|---|---|---|
| History | Chest or left arm pain or discomfort as chief symptom |
|
|
| Reproduction of previous documented angina | |||
| Known history of coronary artery disease, including myocardial infarction | |||
| Physical examination | New transient mitral regurgitation, hypotension, diaphoresis, pulmonary edema or rales | Extracardiac vascular disease | Chest discomfort reproduced by palpation |
| ECG | New or presumably new transient ST-segment deviation (> 0.05 mV) or T-wave inversion (> 0.2 mV) with symptoms |
|
|
| Serum cardiacMarkers | Elevated cardiac troponin T or I, or elevated CK-MB | Normal | Normal |
ACS = acute coronary syndrome; ECG = electrocardiogram; CK-MB = MB isoenzyme of creatine kinase.
Adapted from Braunwald E, et al. Unstable angina: diagnosis and management. Rockville, Md.: U.S. Dept. of Health and Human Services, Public Health Service, Agency for Health Care Policy and Research, National Heart, Lung, and Blood Institute, 1994; Clinical practice guideline no. 10; AHCPR publication no. 94–0602.
Clinical Evaluation
Symptoms of acute coronary syndrome include chest pain, referred pain, nausea, vomiting, dyspnea, diaphoresis, and light-headedness. Some patients may present without chest pain; in one review,2 sudden dyspnea was the sole presenting feature in 4 to 14 percent of patients with acute myocardial infarction. Pain may be referred to either arm, the jaw, the neck, the back, or even the abdomen. Pain radiating to the shoulder, left arm, or both arms somewhat increases the likelihood of acute coronary syndrome (likelihood ratio [LR]: 1.6).3
Typical angina is described as pain that is substernal, occurs on exertion, and is relieved with rest. Patients with all three of these features have a greater likelihood of having acute coronary syndrome than patients with none, one, or even two of these features. Chest pain that occurs suddenly at rest or in a young patient may suggest acute coronary vasospasm, which occurs in Prinzmetal’s angina or with the use of cocaine or methamphetamine. Only about 2 percent of patients with cocaine-associated chest pain have acute coronary syndrome.4
Atypical symptoms do not necessarily rule out acute coronary syndrome. One study5 found the syndrome in 22 percent of 596 patients who presented to emergency departments with sharp or stabbing pain. However, a combination of atypical symptoms improves identification of low-risk patients. The same study5 demonstrated that patients presenting with sharp or stabbing pain, pleuritic pain, and positional chest pain had only a 3 percent likelihood of having acute coronary syndrome.
The physical examination in patients with acute coronary syndrome frequently is normal. Ominous physical findings include a new mitral regurgitation murmur, hypotension, pulmonary rales, a new third heart sound (S3 gallop), and new jugular venous distention. Chest-wall tenderness reduces the likelihood of acute coronary syndrome (-LR: 0.2).3
The likelihood of silent ischemia traditionally has been thought to be greater in patients with diabetes. The “silent myocardial infarction” hypothesis is based on the relatively high incidence of ischemic changes noted on screening ECGs in patients with diabetes. However, in a prospective observational study6 of 528 patients with symptoms suggestive of coronary artery disease on presentation to the emergency department of a cardiac referral center, symptoms did not differ significantly in patients with and without diabetes. The increased frequency of ischemic changes noted on screening ECGs in patients with diabetes simply may reflect their greater baseline risk of coronary artery disease.
Any patient with a history suggestive of acute coronary syndrome should be evaluated in a facility that has ECG and cardiac monitoring equipment.7 Patients with suspected acute coronary syndrome who have chest pain at rest for more than 20 minutes, syncope/presyncope, or unstable vital signs should be referred to an emergency department immediately.7 The diagnosis of acute myocardial infarction, which includes both STEMI and NSTEMI, requires at least two of the following: ischemic symptoms, diagnostic ECG changes, and serum cardiac marker elevation.8,9
The likelihood of acute myocardial infarction is extremely low in patients with a normal or nearly normal ECG who are younger than 60 years and do not have pain described as “pressure” or pain radiating to the arm, shoulder, neck, or jaw. The likelihood of acute infarction is 1.1 percent or less with a normal ECG and 2.6 percent or less with nonspecific ECG changes.10
ECG Interpretation
The ECG provides information that assists in stratifying the patient’s risk of having acute coronary syndrome, establishing the diagnosis, and determining the treatment strategy. Accuracy is enhanced when the ECG is obtained in a patient with ongoing chest pain. The characteristics of common ECG abnormalities in specific anatomic locations are presented in Table 2.11
TABLE 2 ECG Findings for the Diagnosis of Acute Coronary Syndrome
| ECG findings | Lesion | Sensitivity (%) | Specificity (%) | Positive predictive value (%) | Negative predictive value (%) | |
|---|---|---|---|---|---|---|
| ST-segment elevation greater in lead III than in lead II plus ST-segment depression of > 1 mm in lead I, lead aVL, or both | Right coronary artery | 90 | 71 | 94 | 70 | |
| Absence of the above findings plus ST-segment elevation in leads I, aVL, V5, and V6 and ST-segment depression in leads V1, V2, and V3 | Left circumflex coronary artery | 83 | 96 | 91 | 93 | |
| ST-segment elevation in leads V1, V2, and V3 plus any of the features below: | ||||||
| ST-segment elevation of > 2.5 mm in lead V1, right bundle branch block with Q wave, or both | Proximal LAD coronary artery | 12 | 100 | 100 | 61 | |
| ST-segment depression of > 1 mm in leads II, III, and aVF | Proximal LAD coronary artery | 34 | 98 | 93 | 68 | |
| ST-segment depression of ≤ 1 mm or ST-segment elevation in leads II, III, and aVF | Distal LAD coronary artery | 66 | 73 | 78 | 62 | |
ECG = electrocardiogram; LAD = left anterior descending.
Information from Zimetbaum PJ, Josephson ME. Use of the electrocardiogram in acute myocardial infarction. N Engl J Med 2003;348:934, 935.
DIAGNOSTIC ACCURACY
The predictive value of the ECG varies markedly, depending on the baseline risk (pretest probability) for coronary artery disease in a given patient. The number and magnitude of ECG abnormalities also affect sensitivity and specificity.
In a study12 of 775 consecutive patients with chest pain who were admitted to a cardiac care unit, acute myocardial infarction was diagnosed in 10 percent of patients with normal ECG findings (11 of 107 patients) in the emergency department, 8 percent of patients with “minimal changes” (six of 73 patients), and 41 percent of patients with “frankly abnormal” ECG findings (245 of 595 patients).
The magnitude of an ECG abnormality affects diagnostic accuracy. One group of investigators13 found that the diagnosis of NSTEMI is greater than three times more likely in patients with chest pain whose ECG showed ST-segment depression in three or more leads or ST-segment depressions that were greater than or equal to 0.2 mV.
DIAGNOSTIC GUIDELINES
Subendocardial ischemia classically results in ST-segment depression and T-wave inversion.14 Approximately 25 percent of patients with ST-segment depression and elevated creatine kinase–MB isoenzyme (CK-MB) levels eventually develop STEMI, and 75 percent have NSTEMI. Transmural myocardial ischemia results in ST-segment elevation with the vector shifted toward the involved epicardial layer, and without treatment typically results in STEMI. Occasionally, “reciprocal” ST-segment depression occurs in leads that are electrically opposite to the area of injury.
Based on Marriott’s criteria,15 epicardial injury is diagnosed when the J point (origin of the ST segment at its junction with the QRS complex) is (1) elevated by 1 mm or more in two or more limb leads or precordial leads V4 to V6 or by 2 mm or more in two or more precordial leads V1 to V3; or is (2) depressed by 1 mm or more in two or more precordial leads V1 to V3. Serial cardiac marker determinations confirm myocardial injury or infarction in more than 90 percent of patients with J-point elevation in the limb leads.9
Significant Q waves (greater than 0.04 seconds in duration and at least one quarter of the height of the corresponding R wave) suggest myocardial infarction. Isolated small Q waves in leads II, III, and aVF (in the electrically vertical heart) and leads I and aVL (in the electrically horizontal heart) frequently are normal. These small Q waves are known as “septal Q waves” because of the origin of the initial vector in ventricular depolarization.
Although the ECG may be completely normal in a patient with myocardial ischemia and evolving infarction, classic ECG changes occur in STEMI.14 Within minutes, there is J-point elevation, and tall, peaked, “hyperacute” T waves develop; ST-segment elevation and reciprocal-lead ST-segment depression also occur. Abnormal Q waves usually develop within the first day, and T-wave inversion and normalization of ST segments occur within hours to days.
SERUM CARDIAC MARKERS
Serum cardiac marker determinations play a vital role in the diagnosis of acute myocardial infarction. Serum markers such as aspartate transaminase, lactate dehydrogenase, and lactate dehydrogenase subforms no longer are used because they lack cardiac specificity and their delayed elevation precludes early diagnosis.9 Characteristics of the most important serum cardiac markers are summarized in Table 3.16–19
TABLE 3 Characteristics of Serum Cardiac Markers for the Diagnosis of Acute Myocardial Infarction*
| Serum cardiac marker | Test first becomes positive (hours) | Peak level (hours) | Sensitivity (%) | Specificity (%) | Positive predictive value (%) † | Negative predictive value (%) † |
|---|---|---|---|---|---|---|
| CK | ||||||
| Single assay | 3 to 8 | 12 to 24 | 35 | 80 | 20 | 90 |
| Serial assays | 95 | 68 | 30 | 99 | ||
| CK-MB | ||||||
| Single assay | 4 to 6 | 12 to 24 | 35 | 85 | 25 | 90 |
| Serial assays | 95 | 95 | 73 | 99 | ||
| Troponin I and T | ||||||
| Measured 4 hours after onset of chest pain | 4 to 10 | 35 | 96 | 56 | 91 | |
| Measured 10 hours after onset of chest pain | 8 to 28 | 89 | 95 | 72 | 98 | |
CK = creatine kinase; CK-MB = MB isoenzyme of CK.
*—ST-segment elevation myocardial infarction or non-ST-segment elevation in patients presenting to emergency departments with chest pain.
†—Given a 12.5 percent overall likelihood of acute myocardial infarction.
CREATINE KINASE
Creatine kinase (CK) is an enzyme that is found in striated muscle and tissues of the brain, kidney, lung, and gastrointestinal tract. This widely available marker has low sensitivity and specificity for cardiac damage. Furthermore, CK levels may be elevated in a number of noncardiac conditions, including trauma, seizures, renal insufficiency, hyperthermia, and hyperthyroidism.
The serum CK level rises within three to eight hours after myocardial injury, peaks by 12 to 24 hours, and returns to baseline within three to four days.16 A serum CK level may be used as a screening test to determine the need for more specific testing. Although CK commonly was measured serially (along with CK-MB) at the time of hospital admission and six to 12 hours after admission, this marker largely has been replaced by cardiac troponins and CK-MB.9,16
CK-MB ISOENZYME
CK-MB is much more cardiac specific than CK alone, and is useful for the early diagnosis of acute myocardial infarction.9 CK-MB typically is detectable in the serum four to six hours after the onset of ischemia, peaks in 12 to 24 hours, and normalizes in two to three days. The CK-MB mass assay is more sensitive than the CK-MB activity assay.20
Like the CK level, the peak CK-MB level does not predict infarct size; however, it can be used to detect early reinfarction.16 Serial CK-MB levels commonly are obtained at admission to the emergency department and are repeated in six to 12 hours, depending on the assay that is used.20
CK-MB SUBFORMS
CK-MB may be further characterized into subforms (or isoforms). CK-MB2 is found in myocardial tissue, and CK-MB1 is found in plasma. The CK-MB subform assay takes about 25 minutes to perform.21 A CK-MB2 level greater than 1 U per L in combination with a subform ratio greater than 1.5 suggests myocardial injury.9,22 One large study23 involving 1,110 patients with chest pain found that CK-MB subform analysis is 96 percent sensitive and 94 percent specific when the marker is measured six hours after symptom onset. However, the CK-MB subform assay is not yet widely available.
CARDIAC TROPONINS
Troponins (T, I, C) are found in striated and cardiac muscle. Because the cardiac and skeletal muscle isoforms of troponin T and I differ, they are known as the “cardiac troponins.” They are the preferred markers for the diagnosis of myocardial injury.24 Troponin T and I generally have similar sensitivity and specificity for the detection of myocardial injury. Unlike troponin I levels, troponin T levels may be elevated in patients with renal disease, polymyositis, or dermatomyositis.
The cardiac troponins typically are measured at emergency department admission and repeated in six to 12 hours.20 Patients with a normal CK-MB level but elevated troponin levels are considered to have sustained minor myocardial damage or microinfarction, whereas patients with elevations of both CK-MB and troponins are considered to have had acute myocardial infarction. The cardiac troponins may remain elevated up to two weeks after symptom onset, which makes them useful as late markers of recent acute myocardial infarction.9
An elevated troponin T or I level is helpful in identifying patients at increased risk for death or the development of acute myocardial infarction.16 Increased risk is related quantitatively to the serum troponin level. The troponins also can help identify low-risk patients who may be sent home with close follow-up.17 In a study17 of 773 patients presenting to an emergency department with acute chest pain, those with a normal or nearly normal ECG and a normal troponin I test six hours after admission had a very low risk of major cardiac events (0.3 percent) during the next 30 days. Bedside troponin assays are being developed.
MYOGLOBIN
Myoglobin is a low-molecular-weight protein that is present in both cardiac and skeletal muscle. It can be detected in the serum as early as two hours after myocardial necrosis begins. Myoglobin has low cardiac specificity but high sensitivity, which makes it most useful for ruling out myocardial infarction if the level is normal in the first four to eight hours after the onset of symptoms.9
Time changes in the myoglobin value also can be extremely helpful. Combining a doubling of the baseline myoglobin level at two hours after symptom onset with an abnormal myoglobin test at six hours after symptom onset increases the sensitivity to 95 percent at six hours.25
Myoglobin should be used in conjunction with other serum markers, because its level peaks and falls rapidly in patients with ischemia.
A Practical Plan of Action
No assessment protocol or constellation of tests is totally accurate in diagnosing acute coronary syndrome. From 1 to 4 percent of patients ultimately proven to have acute coronary syndrome are sent home from the emergency department.24 Patients with acute coronary syndrome who are sent home without further evaluation are more likely to be women, to be nonwhite, to present without chest pain, or to have ECGs that are normal or show nonspecific changes.18
A suggested approach to the evaluation of patients with chest pain or symptoms consistent with acute coronary syndrome is provided in Figure 1. When a patient presents with chest pain or symptoms suggestive of acute coronary syndrome, vital signs should be obtained, the patient should be monitored, and a focused but careful history should be obtained. A 12-lead ECG should be obtained within 10 minutes of presentation.7
Figure 1. Evaluation of Patients with Chest Pain or Symptoms Suggesting ACS
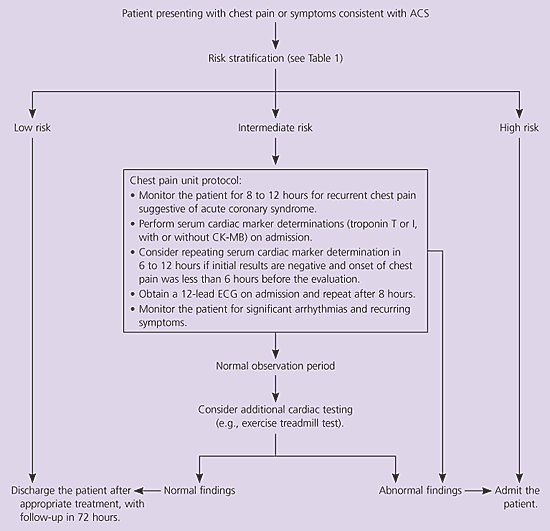
Suggested approach to the evaluation of patients with chest pain or symptoms suggestive of ACS. (ACS = acute coronary syndrome; CK-MB = MB isoenzyme of creatine kinase; ECG = electrocardiogram.)
Risk stratification then should be performed using the criteria in Table 1.1 Alternatively, the Acute Cardiac Ischemia Time-Insensitive Predictive Instrument can be used.26 This is a computerized decision-making program that is built into the ECG machine. Use of this instrument in an emergency department resulted in no change in appropriate admission of patients who had acute coronary syndrome. The benefit of its use was a significant reduction in hospital admissions of patients who did not have acute coronary syndrome.26 However, a subsequent study27 suggested that this benefit is not seen unless physicians have been trained in the use of the instrument.
Patients who are at high risk for acute coronary syndrome should be admitted to a coronary care unit. Patients at intermediate risk may be monitored in a telemetry bed in an inpatient setting or a chest pain unit. A chest pain unit is a specialized unit within an emergency department or a medical center; the unit is dedicated to careful monitoring and aggressive implementation of diagnostic protocols (clinical guidelines) for the evaluation of acute coronary syndrome. Most low-risk patients may undergo early exercise testing or can be discharged with careful outpatient follow-up.
Although protocols for chest pain units may vary somewhat, one protocol28 that has been shown to be safe and cost-effective in an intermediate-risk population consists of the following:
1. Event monitoring and continuous ST-segment monitoring;
2. Measurement of troponins I and T and/or CK-MB at admission and six to eight hours after admission;
3. Four patients staffed by one full-time nurse;
4. Admission to the cardiac care unit or a telemetry bed on the cardiology service for patients with elevated cardiac enzyme levels, recurrent chest pain consistent with unstable angina, or significant ventricular arrhythmias;
5. An exercise treadmill test for patients without abnormal findings on the initial tests, or a nuclear stress test or echocardiographic stress test;
6. Admission of patients with an equivocal or positive result.
Use of this type of systematic approach has the potential to improve the ability of physicians to care for patients with possible acute coronary syndrome, as well as reduce the likelihood of medical error. In the future, advanced diagnostic modalities, such as myocardial perfusion imaging, may have a role in reducing unnecessary hospitalizations.

